Thiamine and Brain Health
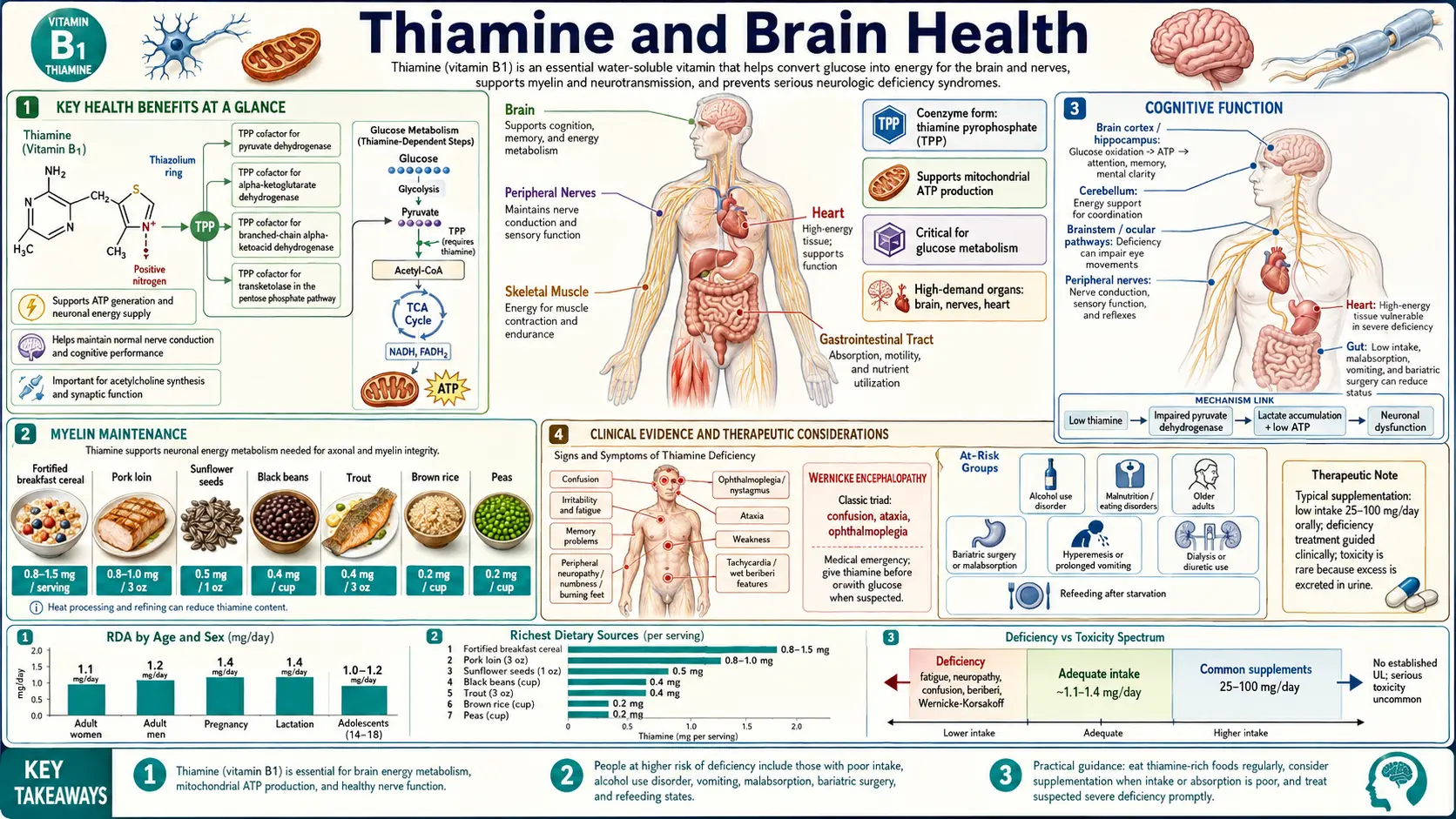
The human brain, despite constituting only 2% of total body weight, consumes approximately 20% of the body’s total energy production. This extraordinary metabolic demand makes the brain uniquely vulnerable to disruptions in energy metabolism. Thiamine (vitamin B1), in its active form thiamine pyrophosphate (TPP), is an indispensable cofactor for three TPP-dependent enzyme complexes — pyruvate dehydrogenase, alpha-ketoglutarate dehydrogenase, and transketolase — each of which sits at a chokepoint of cerebral glucose metabolism. Emerging research has revealed that thiamine’s role in brain health extends far beyond basic energy production, encompassing neurotransmitter synthesis, myelin maintenance, blood-brain barrier integrity, and neuroprotection against age-related cognitive decline. Notably, alpha-ketoglutarate dehydrogenase activity is reduced by 30–75% in post-mortem Alzheimer’s brain tissue, and benfotiamine (a lipid-soluble thiamine prodrug) is under active trial for mild cognitive impairment.
Table of Contents
- Key Health Benefits at a Glance
- ATP Production in Neurons
- Acetylcholine Synthesis
- Myelin Maintenance
- Blood-Brain Barrier Integrity
- Alzheimer’s Disease Research
- Cognitive Function
- Mood and Depression
- Thiamine and Glucose Metabolism in the Brain
- Clinical Evidence and Therapeutic Considerations
- Research Papers and References
- Connections
- Featured Videos
Key Health Benefits at a Glance
The following is a high-level summary of the evidence-backed brain-health benefits of adequate thiamine status. Each is explored in depth below, and the supporting papers are listed in the Research Papers section.
- Powers cerebral ATP production – TPP is required by pyruvate dehydrogenase and alpha-ketoglutarate dehydrogenase, the two rate-limiting enzymes that link glycolysis to the citric acid cycle in neurons.
- Fuels antioxidant defense via transketolase – TPP-dependent transketolase drives the pentose phosphate pathway, which generates NADPH for glutathione recycling and protects neurons from oxidative stress.
- Supports acetylcholine synthesis – Acetyl-CoA from pyruvate dehydrogenase is the direct substrate for choline acetyltransferase; thiamine deficiency reduces ChAT activity in the hippocampus and basal forebrain.
- Preserves myelin integrity – Adequate thiamine supports the energetic demands of oligodendrocytes and Schwann cells and supplies acetyl-CoA/NADPH for myelin lipid synthesis.
- Protects the blood-brain barrier – Deficiency downregulates tight-junction proteins (claudin-5, occludin) and triggers microglial cytokine release, a pattern visible on MRI as periventricular edema in Wernicke’s.
- Links to Alzheimer’s pathology – Post-mortem AD brain tissue shows 30–75% reductions in alpha-ketoglutarate dehydrogenase activity; benfotiamine (a lipid-soluble prodrug) is in trial for MCI/early AD.
- Prevents Wernicke-Korsakoff – Prophylactic thiamine in alcoholic, post-bariatric, hyperemesis, or dialysis patients prevents the irreversible mammillary-body amnesia of Korsakoff syndrome.
- May reduce diabetic cognitive decline – Type 2 diabetics show increased urinary thiamine loss and lower plasma levels; repletion is a plausible modifiable risk factor.
- Benfotiamine bioavailability advantage – Benfotiamine achieves plasma thiamine levels ~5× higher than equivalent oral thiamine HCl, aiding CNS entry.
ATP Production in Neurons
Neurons depend almost exclusively on oxidative metabolism of glucose for their energy supply. Thiamine pyrophosphate serves as an essential cofactor for three critical enzyme complexes in this metabolic pathway:
- Pyruvate dehydrogenase complex (PDC) – This mitochondrial enzyme complex catalyzes the irreversible oxidative decarboxylation of pyruvate to acetyl-CoA, linking glycolysis to the citric acid cycle. Without adequate TPP, pyruvate accumulates and is shunted to lactate via anaerobic glycolysis, producing far less ATP per glucose molecule (2 versus 36–38 ATP) and generating lactic acidosis.
- Alpha-ketoglutarate dehydrogenase complex (KGDHC) – This rate-limiting enzyme of the citric acid cycle converts alpha-ketoglutarate to succinyl-CoA. KGDHC activity is particularly sensitive to thiamine deficiency, and its impairment directly reduces the flux through the citric acid cycle, diminishing NADH and FADH2 production for the electron transport chain. Reduced KGDHC activity has been consistently documented in post-mortem brain tissue from patients with Alzheimer’s disease.
- Branched-chain alpha-keto acid dehydrogenase – This TPP-dependent enzyme is involved in the catabolism of branched-chain amino acids (leucine, isoleucine, valine), which serve as alternative energy substrates for neurons during metabolic stress and contribute to neurotransmitter synthesis.
Additionally, thiamine pyrophosphate is a cofactor for transketolase, a key enzyme in the pentose phosphate pathway. This pathway generates NADPH (required for reductive biosynthesis and antioxidant defense via glutathione recycling) and ribose-5-phosphate (needed for nucleotide and nucleic acid synthesis). Impaired transketolase activity compromises both the neuron’s antioxidant capacity and its ability to synthesize and repair DNA and RNA.
Acetylcholine Synthesis
Acetylcholine is a critical neurotransmitter involved in memory formation, attention, learning, and arousal. Thiamine’s influence on cholinergic neurotransmission operates through several mechanisms:
- Acetyl-CoA provision – The synthesis of acetylcholine requires acetyl-CoA as a substrate. Since TPP-dependent pyruvate dehydrogenase is the primary source of acetyl-CoA in neurons, thiamine deficiency directly reduces the availability of this precursor, leading to diminished acetylcholine production.
- Choline acetyltransferase activity – Studies in animal models of thiamine deficiency have demonstrated reduced choline acetyltransferase (ChAT) activity in the hippocampus, basal forebrain, and cortex – brain regions critical for memory and executive function.
- Cholinergic neuron vulnerability – The basal forebrain cholinergic neurons that project to the hippocampus and cerebral cortex (the nucleus basalis of Meynert) are among the most metabolically active neurons in the brain and are disproportionately affected by thiamine deficiency. Damage to these neurons mirrors the cholinergic deficit observed in Alzheimer’s disease.
- Synaptic vesicle loading – Adequate ATP is required for the vesicular acetylcholine transporter (VAChT) to load acetylcholine into synaptic vesicles. Energy depletion from thiamine deficiency impairs this process, reducing the amount of neurotransmitter available for release at cholinergic synapses.
Myelin Maintenance
Myelin, the lipid-rich insulating sheath surrounding axons, is essential for rapid and efficient nerve impulse conduction. Thiamine contributes to myelin integrity through several pathways:
- Lipid synthesis – Myelin is composed predominantly of lipids (approximately 70–80% of its dry weight), including cholesterol, cerebrosides, and phospholipids. The synthesis of these lipid components requires acetyl-CoA (from TPP-dependent pyruvate dehydrogenase) and NADPH (from the TPP-dependent pentose phosphate pathway).
- Oligodendrocyte metabolism – Oligodendrocytes, the glial cells responsible for producing and maintaining myelin in the central nervous system, have exceptionally high metabolic rates. They are among the most metabolically active cells in the brain and are particularly vulnerable to energy depletion resulting from thiamine deficiency.
- Demyelination in deficiency – Both clinical observations and animal studies have documented demyelination in thiamine-deficient states, particularly affecting the mammillary bodies, thalamus, and periventricular white matter. This demyelination contributes to the cognitive and motor symptoms seen in Wernicke’s encephalopathy.
- Schwann cell function – In the peripheral nervous system, Schwann cells perform the myelinating function. Thiamine deficiency-induced Schwann cell dysfunction contributes to the peripheral neuropathy characteristic of dry beriberi, where segmental demyelination and axonal degeneration both occur.
Blood-Brain Barrier Integrity
The blood-brain barrier (BBB) is a specialized system of endothelial cells, pericytes, astrocyte foot processes, and basement membrane that tightly regulates the passage of substances between the bloodstream and the brain parenchyma. Thiamine plays important roles in maintaining BBB function:
- Endothelial tight junctions – Brain capillary endothelial cells are connected by tight junctions that are far more restrictive than those in peripheral vasculature. Thiamine deficiency has been shown to downregulate tight junction protein expression (claudin-5, occludin), increasing paracellular permeability.
- Oxidative stress and BBB damage – Impaired antioxidant defense (through reduced NADPH production from the pentose phosphate pathway) leads to accumulation of reactive oxygen species that damage endothelial cell membranes and basement membrane components.
- Inflammatory cytokine release – Thiamine deficiency activates microglia and astrocytes, which release pro-inflammatory cytokines (TNF-alpha, IL-1beta, IL-6) that further compromise BBB integrity. This inflammatory response creates a feedforward cycle of BBB breakdown and neuroinflammation.
- Vasogenic edema – BBB disruption in thiamine deficiency permits extravasation of plasma proteins and fluid into the brain parenchyma, producing vasogenic edema. This is particularly evident in the medial thalami and periaqueductal regions, as demonstrated by MRI findings in Wernicke’s encephalopathy showing characteristic signal abnormalities in these areas.
Alzheimer’s Disease Research
A growing body of evidence links thiamine status to the pathogenesis and progression of Alzheimer’s disease (AD). While thiamine deficiency alone does not cause AD, several converging lines of evidence suggest that impaired thiamine-dependent metabolism contributes to the neurodegenerative process:
- Reduced KGDHC activity – Activity of the alpha-ketoglutarate dehydrogenase complex is consistently reduced by 30–75% in post-mortem brain tissue from AD patients compared to age-matched controls. This reduction correlates with clinical dementia severity and precedes significant neuronal loss.
- Thiamine-dependent enzyme deficits – Beyond KGDHC, transketolase and pyruvate dehydrogenase activities are also reduced in AD brains. These reductions are not simply a consequence of neuronal loss, as they occur in brain regions with minimal neuron loss and are detectable early in the disease course.
- Amyloid-beta connection – Thiamine deficiency in animal models increases amyloid precursor protein (APP) expression and promotes amyloidogenic processing, leading to increased amyloid-beta production. Conversely, thiamine supplementation has been shown to reduce amyloid plaque burden in transgenic mouse models of AD.
- Tau hyperphosphorylation – Impaired glucose metabolism and oxidative stress resulting from thiamine deficiency promote abnormal hyperphosphorylation of tau protein, a key pathological feature of AD. Hyperphosphorylated tau forms neurofibrillary tangles that disrupt microtubule stability and axonal transport.
- Clinical trials – Benfotiamine, a lipid-soluble thiamine derivative with superior bioavailability, has shown promise in clinical trials for mild cognitive impairment and early AD. A pilot study demonstrated improvements in cognitive test scores and reductions in AD biomarkers in cerebrospinal fluid after 12 months of benfotiamine supplementation.
Cognitive Function
Even in the absence of overt deficiency, suboptimal thiamine status can influence cognitive performance across the lifespan:
- Attention and processing speed – Marginal thiamine depletion (insufficient to produce clinical signs) has been associated with impaired attention, slower reaction times, and reduced working memory capacity in controlled human studies.
- Learning and memory – Animal studies consistently demonstrate that thiamine deficiency impairs spatial learning and memory, as measured by maze performance and fear conditioning tasks. These deficits correlate with reduced long-term potentiation (LTP) in the hippocampus, the cellular mechanism underlying memory formation.
- Age-related cognitive decline – Elderly populations frequently exhibit suboptimal thiamine status due to reduced dietary intake, impaired absorption, medication interactions (particularly loop diuretics), and age-related changes in thiamine metabolism. Supplementation studies in elderly cohorts have shown modest improvements in cognitive test scores and subjective well-being.
- Diabetes and cognition – Patients with type 2 diabetes exhibit increased urinary thiamine excretion and frequently have low plasma thiamine levels. Given the established link between diabetes and cognitive decline, thiamine depletion may represent a modifiable risk factor in this population.
Mood and Depression
Emerging evidence suggests that thiamine status influences mood regulation and may contribute to depressive symptomatology:
- Serotonin metabolism – Thiamine deficiency affects tryptophan metabolism through the kynurenine pathway, potentially reducing serotonin availability. Since tryptophan is the precursor to serotonin, diversion of tryptophan toward kynurenine pathway metabolites reduces substrate availability for serotonin synthesis.
- Supplementation studies – Several controlled trials have reported improvements in mood, energy, and overall well-being following thiamine supplementation, even in participants without clinical deficiency. Doses of 50–100 mg daily have shown benefits in young adult women and elderly populations.
- Fatigue and apathy – Generalized fatigue, irritability, and apathy are among the earliest nonspecific symptoms of thiamine depletion. These symptoms overlap substantially with those of major depressive disorder and may be misdiagnosed as primary psychiatric illness rather than nutritional deficiency.
- Neuroinflammation – Thiamine deficiency-induced microglial activation and pro-inflammatory cytokine release may contribute to neuroinflammatory processes that have been increasingly recognized as contributing factors in the pathophysiology of depression.
Thiamine and Glucose Metabolism in the Brain
The brain’s near-total dependence on glucose as its primary energy substrate makes the relationship between thiamine and cerebral glucose metabolism critically important:
- Glucose utilization – Positron emission tomography (PET) studies using fluorodeoxyglucose (FDG) have demonstrated widespread reductions in cerebral glucose metabolic rate in thiamine-deficient states, with the most pronounced decreases in the thalamus, mammillary bodies, and cerebellum.
- Lactate accumulation – When pyruvate dehydrogenase activity is compromised by thiamine deficiency, pyruvate is diverted to lactate rather than entering the citric acid cycle. Elevated brain lactate levels, detectable by magnetic resonance spectroscopy, indicate a shift toward anaerobic metabolism and impending energy failure.
- Selective vulnerability – Not all brain regions are equally affected by thiamine deficiency. Structures with the highest metabolic rates and thiamine turnover, including the medial thalamus, mammillary bodies, inferior colliculus, and cerebellar vermis, are preferentially damaged. This selective vulnerability explains the characteristic clinical and radiological patterns of Wernicke’s encephalopathy.
- Metabolic crisis threshold – The brain has very limited glycogen reserves (sufficient for only a few minutes of normal metabolic activity) and cannot efficiently utilize fatty acids as an alternative fuel. Thiamine depletion therefore creates an energy crisis more rapidly in the brain than in most other organs.
Clinical Evidence and Therapeutic Considerations
The clinical evidence supporting thiamine’s importance in brain health has several practical implications:
- High-risk screening – Patients with alcohol use disorder, bariatric surgery, chronic kidney disease on dialysis, cancer with prolonged vomiting, and hyperemesis gravidarum should be routinely assessed for thiamine deficiency and considered for prophylactic supplementation.
- Benfotiamine advantages – This lipid-soluble thiamine prodrug achieves plasma thiamine levels approximately 5-fold higher than equivalent doses of water-soluble thiamine hydrochloride. Its superior bioavailability and ability to cross cell membranes make it a promising agent for neuroprotective applications.
- Combination approaches – Thiamine works synergistically with other B vitamins (B6, B12, folate) in supporting brain metabolism. Deficiency of multiple B vitamins is common in at-risk populations, and combined supplementation may provide greater cognitive benefit than single-nutrient approaches.
- Recommended intakes – The current Recommended Dietary Allowance for thiamine is 1.1–1.2 mg per day for adults. However, this level is set to prevent overt deficiency and may not be optimal for neuroprotection. Many brain health researchers advocate for higher intakes, particularly in older adults and those with increased metabolic demands.
Thiamine’s fundamental role in cerebral energy metabolism, neurotransmitter synthesis, and neuroprotection makes it one of the most important micronutrients for brain health across the lifespan. Ensuring adequate thiamine intake through diet (whole grains, legumes, pork, fortified cereals) or supplementation is a simple yet powerful strategy for supporting cognitive function and potentially reducing the risk of neurodegenerative disease.
Research Papers and References
The following are landmark and frequently cited research papers underpinning the claims on this page. Links resolve to the publisher DOI or PubMed record.
Foundational Reviews
- Lonsdale D. A review of the biochemistry, metabolism and clinical benefits of thiamin(e) and its derivatives. Evid Based Complement Alternat Med. 2006;3(1):49-59.
- PubMed — Thiamine pyrophosphate and cerebral energy metabolism reviews
TPP-Dependent Enzymes and Alzheimer’s Disease
- PubMed — Alpha-ketoglutarate dehydrogenase activity in Alzheimer’s disease
- PubMed — Transketolase, pentose phosphate pathway and Alzheimer’s
- PubMed — Thiamine deficiency, amyloid-beta and tau hyperphosphorylation
Benfotiamine and Clinical Trials in MCI/AD
- PubMed — Benfotiamine in mild cognitive impairment and early Alzheimer’s
- PubMed — Benfotiamine versus thiamine HCl bioavailability
Cognition, Mood, and Diabetes
- PubMed — Thiamine supplementation and cognition in elderly cohorts
- PubMed — Thiamine status, mood and depression
- PubMed — Type 2 diabetes and urinary thiamine loss
Blood-Brain Barrier and Wernicke Imaging
- PubMed — Thiamine deficiency and blood-brain barrier tight-junction breakdown
- PubMed — Wernicke encephalopathy MRI findings
External Authoritative Resources
- NIH Office of Dietary Supplements — Thiamin Fact Sheet for Health Professionals
- Linus Pauling Institute Micronutrient Information Center — Thiamin
- Harvard T.H. Chan School — Nutrition Source: B Vitamins
- PubMed — Thiamine and brain/cognition research search
Connections
- Vitamin B1
- Thiamine and Beriberi
- Vitamin B6
- Vitamin B12
- Vitamin B9
- Vitamin D3
- Magnesium
- Alzheimer's Disease
- Parkinson's Disease
- Depression
- Diabetes
- Sleep Hygiene
- Choline
- Peripheral Neuropathy
- Brain Fog