United States vs European Union: Food Safety Regulation Compared
The United States and the European Union represent two fundamentally different approaches to food safety regulation. While both systems aim to protect public health, they are built on opposing philosophical foundations that produce dramatically different outcomes for consumers. The EU bans or restricts over 1,300 chemicals in food and personal care products, while the US bans or restricts a small fraction of that number. American food companies routinely sell different — and safer — versions of the same products in Europe than they sell at home. Understanding why this gap exists, and how it affects your health, is essential for any informed consumer.
Table of Contents
- Key Effects at a Glance
- The Philosophical Divide
- The GRAS Loophole
- Same Company, Different Products: The Two-Tier System
- FDA Funding and Industry Influence
- How the EU System Works in Practice
- What These Differences Mean for Your Health
- How Consumers Can Protect Themselves
- Signs of Progress
- The Bottom Line
- Research Papers
- Connections
- Featured Videos
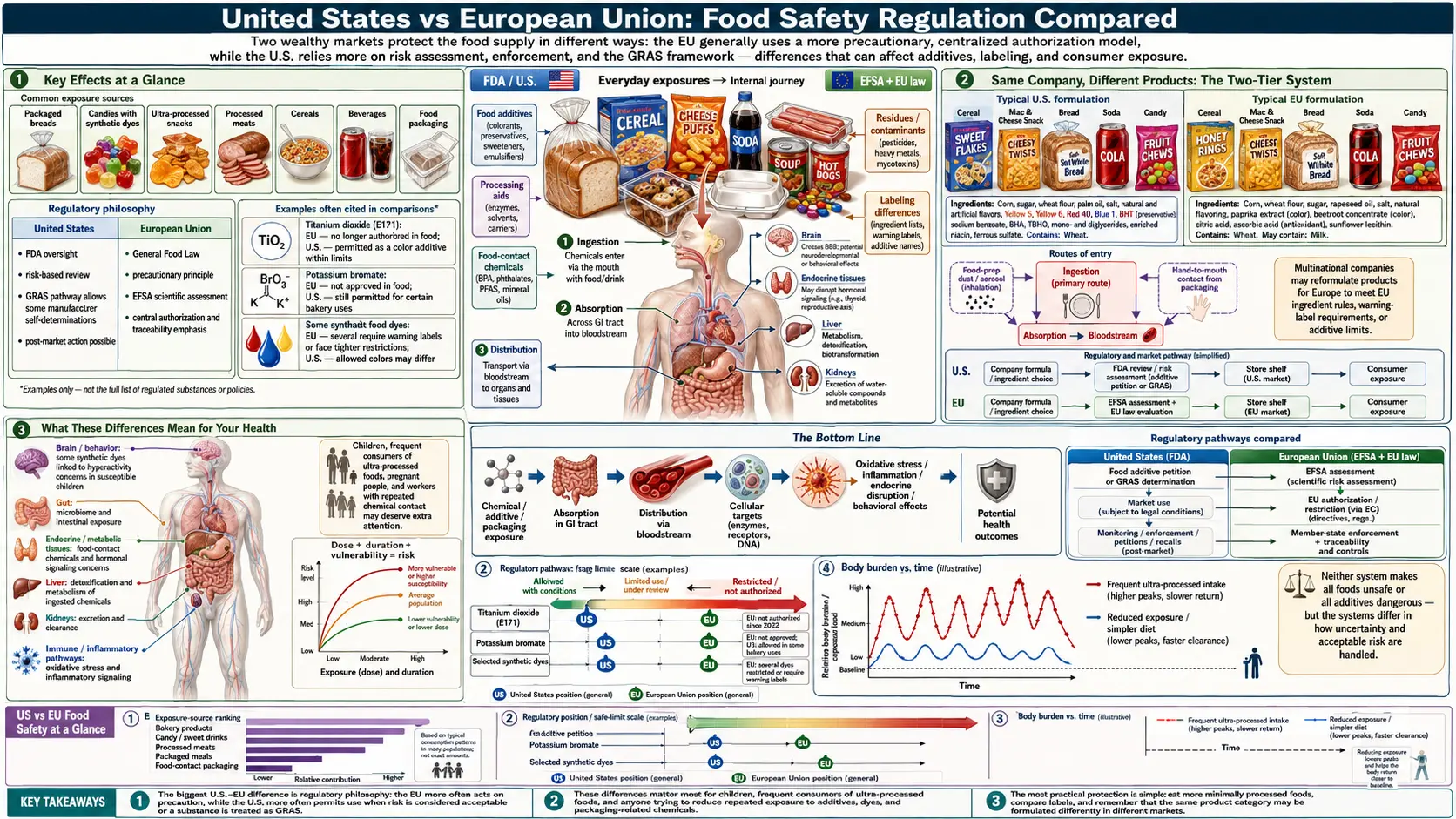
Key Effects at a Glance
- US permits ~10,000+ food additives, many through self-affirmed GRAS with no FDA review.
- EU bans or restricts 1,300+ chemicals in food and cosmetics not restricted in the US.
- Identical products are reformulated for European markets (Kraft Mac & Cheese, Fanta, Starburst, Froot Loops, McDonald’s fries, Subway bread).
- 100% of GRAS notifications in one JAMA Internal Medicine analysis had industry-connected reviewers.
- Precautionary principle vs. prove-harm – EU shifts burden of proof to manufacturers; US shifts it to regulators and the public.
- EFSA conducts systematic re-evaluation of authorized additives; FDA has no analogous program.
The Philosophical Divide
The EU Precautionary Principle
The European Union's approach to food safety is governed by the precautionary principle, which is codified in Article 191 of the Treaty on the Functioning of the European Union and further elaborated in EU Regulation 178/2002 (the General Food Law). The precautionary principle states that when scientific evidence suggests a substance may pose a risk to human health, protective action should be taken even if the evidence is not yet conclusive.
In practice, this means:
- Pre-market approval is required — New food additives must undergo comprehensive safety testing and receive formal authorization from the European Commission, based on a scientific risk assessment by the European Food Safety Authority (EFSA), before they can be used in food.
- Safety must be demonstrated by the applicant — The company seeking to use a food additive must provide extensive toxicological data proving the substance is safe. The burden of proof falls on industry, not on regulators or the public.
- Approved additives are regularly re-evaluated — EFSA systematically re-evaluates all previously authorized food additives, incorporating the latest scientific evidence. This process has led to the revocation of authorizations for substances like titanium dioxide (E171), which was banned as a food additive in 2022 after a re-evaluation found it could no longer be considered safe.
- Reasonable suspicion is grounds for action — Regulators do not need to wait for definitive proof of harm in humans. Evidence from animal studies, in vitro studies, or mechanistic data can be sufficient to restrict or ban a substance.
- The precautionary principle acknowledges uncertainty — Rather than treating the absence of evidence as evidence of safety, the EU recognizes that toxicological science is inherently limited and that some risks may not be detectable until after widespread, long-term human exposure.
The US "Substantial Equivalence" and "Prove Harm" Approach
The United States regulatory framework for food safety, administered primarily by the FDA (Food and Drug Administration) and the USDA (United States Department of Agriculture), operates on a fundamentally different set of assumptions:
- Substances are innocent until proven guilty — The default position is that a substance is safe unless there is compelling evidence of harm. The burden of proof falls on regulators, consumers, or independent scientists to demonstrate that a substance is dangerous — not on manufacturers to prove it is safe.
- The GRAS system allows substances to bypass FDA review entirely — Under the GRAS (Generally Recognized as Safe) framework, substances can be used in food without any FDA review if they are "generally recognized" as safe by qualified experts. This determination can be made by the manufacturer itself without even notifying the FDA (self-affirmed GRAS).
- Risk-based rather than hazard-based assessment — While the EU considers whether a substance has the inherent ability to cause harm (hazard-based assessment), the US focuses on whether the expected level of exposure poses an unacceptable risk (risk-based assessment). This approach assumes that exposure levels can be accurately predicted and controlled, which is often not the case.
- Regulatory action is slow and reactive — The US system is designed to respond to demonstrated harm rather than to prevent potential harm. This means that harmful substances often remain in the food supply for years or decades after evidence of their danger emerges.
The GRAS Loophole: The Biggest Hole in US Food Safety
The GRAS (Generally Recognized as Safe) system is the single most significant structural weakness in US food safety regulation. Originally intended for common food substances with a long history of safe use (like vinegar, salt, and baking soda), the GRAS framework has been exploited to allow thousands of novel chemicals into the food supply with little or no independent safety review.
How GRAS Works (and Fails)
- Self-affirmed GRAS — A company can determine that a substance is GRAS based on its own internal review and the opinion of its own hired experts. The company is not required to inform the FDA of this determination, and the FDA may never know the substance is being used in food. An estimated 1,000 or more food chemicals have entered the food supply through self-affirmed GRAS without any FDA review.
- Voluntary GRAS notification — Companies can voluntarily notify the FDA of a GRAS determination. The FDA reviews the notification and issues a "no objection" letter — but this is not the same as an approval. The FDA's review is limited to the information submitted by the company, and the agency does not conduct its own independent testing.
- Conflict of interest — A 2013 study published in JAMA Internal Medicine found that in the voluntary GRAS notification program, 100% of the safety determinations submitted to the FDA were made by experts with financial ties to the companies seeking approval. Not a single independent scientist was involved in any of the 451 notifications examined.
- No systematic re-evaluation — Unlike the EU system, there is no mechanism for the systematic re-evaluation of GRAS substances as new science emerges. Substances that were declared GRAS decades ago, based on the limited science of the time, remain in that category indefinitely unless the FDA takes affirmative action to remove them.
- Historical GRAS substances — Some substances, including BHA and BHT, received GRAS status before modern toxicology methods were available. Despite subsequent evidence of carcinogenicity (in BHA's case, classification as a "reasonably anticipated human carcinogen" by the NTP), their GRAS status has never been revoked.
The Numbers Tell the Story
The disparity between the US and EU systems is stark when measured by outcomes:
- The EU has banned or restricted over 1,300 chemicals in food and cosmetics. The US has banned or restricted far fewer — estimates vary, but the US cosmetics industry alone uses approximately 1,100 chemicals that are banned in the EU.
- The EU requires pre-market safety assessment for all food additives. The US GRAS system allows an estimated 10,000+ substances in food, many of which have never been reviewed by the FDA.
- EFSA employs hundreds of scientists dedicated to food safety assessment. The FDA's food safety functions are chronically underfunded and understaffed relative to the scope of the agency's responsibilities, which also include drugs, medical devices, tobacco, and more.
Same Company, Different Products: The Two-Tier System
Perhaps the most damning indictment of the US food safety system is the fact that American food companies routinely sell different versions of the same products in different countries — with the safer version going to European consumers and the inferior version going to Americans. These companies have already developed the safer formulations; they simply choose not to sell them in the US because they are not required to.
Kraft Mac & Cheese
In the United States, Kraft Macaroni & Cheese historically contained Yellow 5 (tartrazine) and Yellow 6 (sunset yellow) — artificial petroleum-derived dyes linked to hyperactivity in children and requiring warning labels in the EU. In the United Kingdom, the same product (sold as "Kraft Macaroni Cheese" or under other Kraft brands) used paprika extract and beta-carotene for coloring — natural, plant-derived colorants with no associated health risks. After sustained consumer pressure, Kraft reformulated its US product in 2016 to remove artificial dyes, demonstrating that the safer formulation was feasible all along.
Fanta Orange
Fanta Orange sold in the US contains Red 40 and Yellow 6, both synthetic dyes. Fanta Orange sold in the UK and EU uses pumpkin and carrot extracts for coloring. The taste and appearance are similar; the ingredients are dramatically different.
Starburst and Skittles
In the US, these candies contain multiple artificial dyes including Red 40, Yellow 5, Yellow 6, and Blue 1. In the UK, the same brands use natural colorants derived from plants, fruits, and vegetables, including spirulina extract, black carrot concentrate, and safflower extract.
Kellogg's Cereals
Several Kellogg's cereals sold in the US contain BHT and artificial colors that are not present in the versions sold in the EU. Froot Loops in the US contains Red 40, Blue 1, Yellow 6, and Blue 2. In the EU, Froot Loops (where available) uses natural colorants.
McDonald's
McDonald's french fries in the US contain 19 ingredients, including TBHQ (a preservative derived from butane) and dimethylpolysiloxane (an anti-foaming agent also used in silicone caulk). McDonald's french fries in the UK contain just 4 ingredients: potatoes, vegetable oil, dextrose, and salt.
Subway Bread
Subway's bread in the US contained azodicarbonamide (the "yoga mat chemical") until 2014, when public outcry led the company to remove it. Azodicarbonamide was never permitted in Subway's European bread because it is banned in the EU.
These examples demonstrate a critical point: the safer formulations are not more expensive or technologically challenging to produce. American food companies could sell the same safer products in the US that they sell in Europe. They simply choose not to, because US regulations do not require them to.
FDA Funding and Industry Influence
The structural inadequacies of US food safety regulation are compounded by the FDA's funding model and the pervasive influence of the food and chemical industries on regulatory decision-making.
Industry User Fees
A significant and growing portion of the FDA's budget comes from user fees paid by the industries the agency regulates. In the pharmaceutical sector, industry user fees (under PDUFA, the Prescription Drug User Fee Act) now fund the majority of the drug review process. While the food sector's user fee contribution is smaller, the broader pattern creates an institutional culture in which the FDA views regulated industries as "clients" and "stakeholders" rather than as entities to be regulated in the public interest.
The Revolving Door
The movement of personnel between the FDA, the USDA, and the food/chemical industry is well-documented and profoundly undermines regulatory independence:
- Industry executives become regulators — Former executives and lobbyists from companies like Monsanto, DuPont, Kraft, and Coca-Cola have held senior positions at the FDA and USDA, where they make decisions affecting the products of their former (and often future) employers.
- Regulators become industry employees — FDA and USDA officials frequently leave government service for lucrative positions in the industries they previously regulated. This dynamic creates an incentive for regulators to maintain favorable relationships with industry during their government tenure.
- Advisory committee conflicts — The FDA's scientific advisory committees, which provide expert recommendations on food safety issues, frequently include members with financial ties to affected industries. While disclosure requirements exist, they do not prevent conflicted individuals from participating in and voting on decisions that affect their financial interests.
Lobbying
The food and beverage industry spends hundreds of millions of dollars annually on lobbying at the federal and state levels. According to the Center for Responsive Politics, the food and beverage sector has consistently been among the top lobbying spenders in Washington. This spending buys access, influence, and, critically, the ability to delay or prevent regulatory action on harmful substances.
- The sugar industry funded research in the 1960s to deflect blame for heart disease from sugar to fat, shaping dietary guidelines for decades.
- The trans fat industry lobbied for years to delay mandatory labeling and bans, even as evidence of cardiovascular harm accumulated.
- The artificial dye industry has lobbied against warning labels similar to those required in the EU.
- The pesticide industry has lobbied to prevent the banning of atrazine, chlorpyrifos, and other chemicals banned in the EU.
How the EU System Works in Practice
The European Food Safety Authority (EFSA)
EFSA is an independent scientific agency established in 2002 in response to food safety crises including the BSE (mad cow disease) outbreak. EFSA provides scientific risk assessments that form the basis for regulatory decisions by the European Commission and member states. Key features of EFSA include:
- Independence — EFSA is funded by the EU budget, not by industry fees. Its scientific panels are composed of independent experts who must declare and manage conflicts of interest.
- Transparency — Under the 2019 Transparency Regulation, EFSA is required to proactively publish all studies and data submitted as part of food safety applications. This allows independent scientists and the public to scrutinize the evidence.
- Systematic re-evaluation — EFSA is conducting a systematic re-evaluation of all food additives that were authorized before 2009. This program, scheduled for completion by the end of 2024, applies current scientific standards to substances that were approved under older, less rigorous criteria. This re-evaluation led to the ban on titanium dioxide (E171) in 2022.
- Comprehensive scope — EFSA assesses food additives, pesticides, GMOs, contaminants, animal feed, nutrition, and food-borne diseases, providing a holistic view of food safety.
The EU Authorization Process
Before a food additive can be used in the EU, it must go through a rigorous authorization process:
- Application submission — The company must submit a comprehensive dossier including toxicological studies, exposure assessments, and proposed conditions of use.
- EFSA scientific assessment — EFSA's Panel on Food Additives and Flavourings (FAF) evaluates the scientific evidence and issues an opinion on the safety of the substance.
- Risk management decision — The European Commission, in consultation with member states, makes the final regulatory decision, considering EFSA's scientific opinion along with other legitimate factors such as consumer concerns and socioeconomic impacts.
- Inclusion in the Union List — Authorized food additives are placed on the Union List (EU Regulation 1333/2008), which specifies the conditions under which each additive may be used, including which food categories, maximum levels, and labeling requirements.
- Ongoing monitoring — After authorization, EFSA may require post-market monitoring and can initiate a re-evaluation if new safety concerns emerge.
What These Differences Mean for Your Health
The practical consequences of these regulatory differences are significant:
- Chemical exposure — Americans are exposed to hundreds of chemicals in their food and personal care products that Europeans are not. This includes potential carcinogens, endocrine disruptors, neurotoxins, and immune system disruptors.
- Chronic disease rates — While many factors contribute to chronic disease, the US has higher rates of many diet-related conditions than most EU countries, including obesity, type 2 diabetes, certain cancers, and autoimmune diseases. The role of food additives in these disparities is difficult to isolate but plausible.
- Children's health — American children are exposed to higher levels of food dyes, preservatives, and hormones than their European counterparts. Rates of ADHD diagnosis, food allergies, and early puberty are higher in the US than in most EU countries.
- Consumer trust — European consumers generally have higher trust in their food safety system than American consumers. The EU's precautionary approach provides a level of assurance that the US system does not.
How Consumers Can Protect Themselves
Until the US regulatory system undergoes fundamental reform, American consumers must take their own protective measures:
- Educate yourself — Learn which additives are banned in other countries and check for them on labels. Resources like the Environmental Working Group's (EWG) Food Scores database and the Center for Science in the Public Interest's (CSPI) Chemical Cuisine guide can help.
- Choose organic — USDA Organic certification prohibits most synthetic additives, artificial colors, preservatives, growth hormones, and many pesticides. While organic certification is not perfect, it significantly reduces exposure to banned-elsewhere chemicals.
- Eat whole foods — Unprocessed fruits, vegetables, whole grains, legumes, nuts, and seeds contain no added chemicals. The more of your diet that comes from whole foods, the less chemical exposure you will have.
- Seek EU-formulated products — Some specialty and import stores carry European versions of products, which meet stricter EU standards. Some US brands now offer "clean label" lines that meet EU standards voluntarily.
- Cook at home — Preparing food from scratch gives you complete control over ingredients. Homemade bread does not need azodicarbonamide. Homemade mac and cheese does not need Yellow 5.
- Filter your water — Activated carbon filters (such as Brita or Berkey) and reverse osmosis systems can remove many pesticide residues, including atrazine, from drinking water.
- Support advocacy organizations — Groups like the Environmental Working Group (EWG), Center for Science in the Public Interest (CSPI), Consumer Reports, and the Center for Food Safety advocate for stronger food safety regulations and provide consumer guidance.
- Contact your elected officials — Express support for food safety legislation and FDA reform. The California Food Safety Act of 2023 demonstrated that state-level action is possible and can drive national change.
- Demand transparency from brands — Use social media, customer service channels, and consumer campaigns to ask food companies why they sell different (safer) formulations in Europe than in the US. Consumer pressure has already driven several major reformulations.
- Share this information — Many consumers are unaware of the regulatory gap between the US and the EU. Sharing knowledge about banned-elsewhere additives empowers others to make informed choices and builds the consumer demand that drives industry change.
Signs of Progress
Despite the significant structural problems in the US food safety system, there are reasons for cautious optimism:
- California Food Safety Act (2023) — This landmark state law banned Red No. 3, potassium bromate, brominated vegetable oil, and propylparaben from food sold in California, effective 2027. As the world's fifth-largest economy, California's food safety standards often influence national and international practices.
- FDA ban on BVO (2024) — After decades of allowing brominated vegetable oil despite international bans, the FDA finally banned BVO in 2024, demonstrating that federal action is possible, even if slow.
- FDA ban on Red No. 3 (2025/2027) — The FDA revoked the authorization for Red No. 3 in food, ending a decades-long debate about a dye that was shown to cause thyroid tumors in rats.
- Corporate reformulations — Companies including Kraft, General Mills, Nestle, and Mars have voluntarily removed some artificial dyes and preservatives from US products, often citing consumer demand.
- Growing consumer awareness — Social media, documentary films, and consumer advocacy have dramatically increased public awareness of food safety issues, creating market pressure for cleaner food.
- State-level action — Multiple states are considering food safety legislation modeled on California's act, potentially creating a patchwork of state regulations that could pressure federal action.
The Bottom Line
The difference between the US and EU food safety systems is not a matter of differing scientific opinions — both systems have access to the same science. It is a matter of values, priorities, and political will. The EU has decided that when science suggests a food additive may harm people, the additive should be restricted until its safety is proven. The US has decided that when science suggests a food additive may harm people, the additive should remain available until its danger is conclusively proven — a standard that can take decades to meet while millions of people are exposed.
American consumers deserve the same protections as European consumers. The same companies already make the safer products; they simply sell them overseas. Until the US system changes, every American must be their own food safety advocate — reading labels, choosing wisely, demanding better, and refusing to accept that the land of the free should also be the land of the most chemically contaminated food supply in the developed world.
Research Papers
- Neltner TG, Alger HM, O’Reilly JT, Krimsky S, Bero LA, Maffini MV. Conflicts of interest in approvals of additives to food determined to be generally recognized as safe. JAMA Internal Medicine. 2013;173(22):2032-2036.
- Regulation (EC) No 1333/2008 on food additives (consolidated). Official Journal of the European Union.
- Regulation (EC) No 178/2002 (General Food Law) establishing EFSA and the precautionary principle.
- EFSA Panel on Food Additives. Safety assessment of titanium dioxide (E171) as a food additive. EFSA Journal. 2021;19(5):6585.
- PubMed topic search: GRAS food additive conflict of interest
- PubMed topic search: precautionary principle food safety regulation
- PubMed topic search: food additive regulation EU US comparison
- PubMed topic search: titanium dioxide E171 genotoxicity
- PubMed topic search: azodicarbonamide flour improver toxicity
- PubMed topic search: brominated vegetable oil BVO thyroid
- PubMed topic search: potassium bromate flour carcinogen
- PubMed topic search: ultra-processed food chronic disease
Connections
- Banned Additives
- Preservatives
- BHA and BHT
- Sodium Nitrite
- Pesticides
- Food Dyes Banned Worldwide
- Red 3 Erythrosine
- Red 40 Allura Red
- Yellow 5 Tartrazine
- Acesulfame Potassium
- Food Additives
- Titanium Dioxide
- Azodicarbonamide
- Brominated Vegetable Oil
- Potassium Bromate
- ADHD
- Allergies
- Food Dyes