Acesulfame Potassium (Ace-K): The Hidden Sweetener
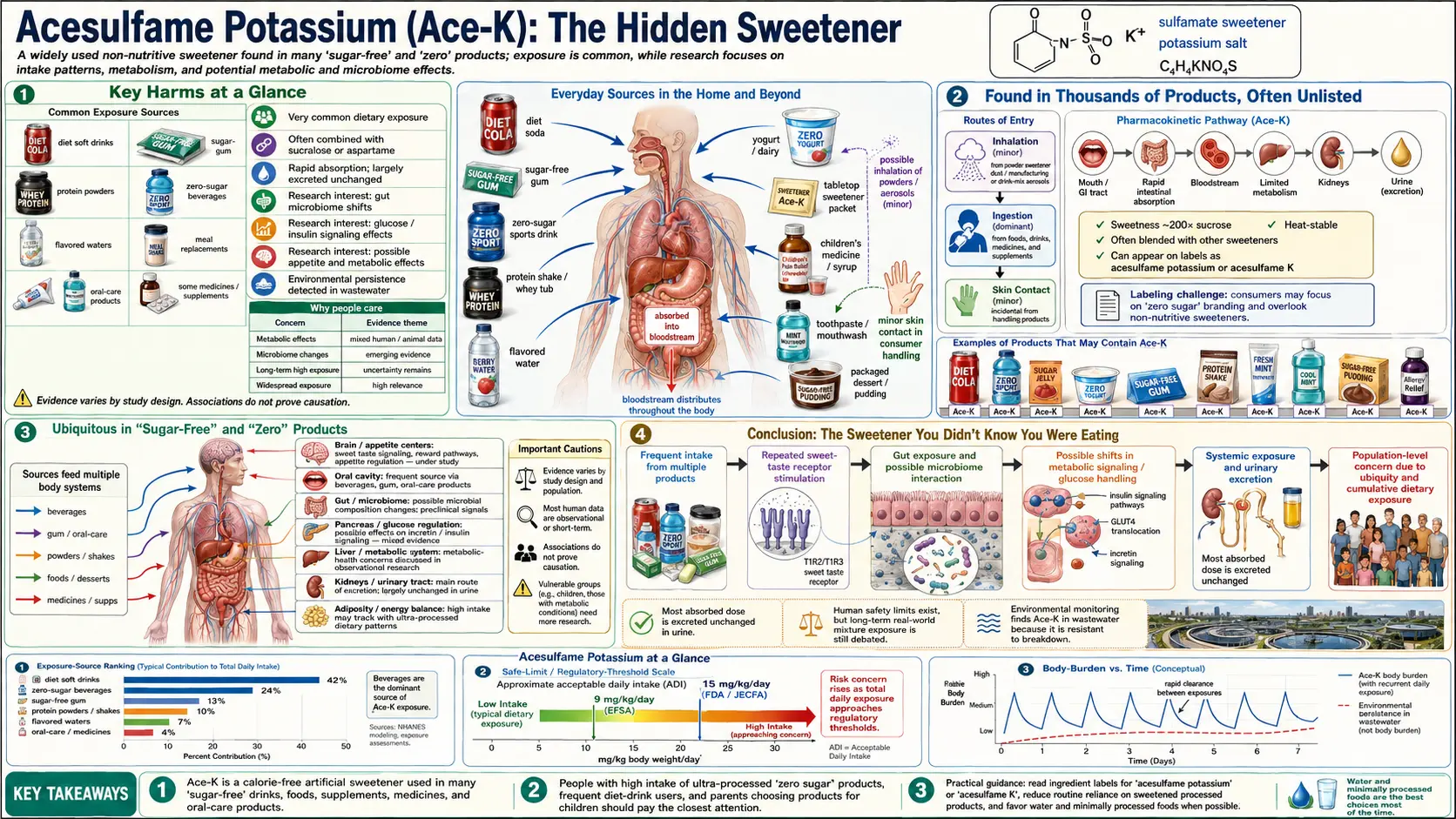
Acesulfame potassium, commonly abbreviated as Ace-K and sold under the brand names Sunett and Sweet One, is one of the most widely used yet least recognized artificial sweeteners in the modern food supply. Approximately 200 times sweeter than sugar, Ace-K is found in thousands of products worldwide, yet most consumers have never heard of it. This obscurity is by design: Ace-K is frequently blended with other sweeteners and often listed deep in ingredient panels or obscured under generic terms like "artificial flavors." Its approval was based on studies widely criticized as inadequate, it contains a known carcinogen (methylene chloride), and it has received virtually no long-term human safety testing.
Table of Contents
- Key Harms at a Glance
- What Is Acesulfame Potassium?
- Methylene Chloride: A Carcinogen in Your Sweetener
- Inadequate Original Safety Testing
- Where It Is Found (Exposure Routes)
- Potential Thyroid Disruption
- Pregnancy Concerns
- Neurological Effects
- Virtually No Long-Term Human Studies
- Ubiquitous in Sugar-Free and Zero Products
- The Gut Microbiome Connection
- Safety Thresholds & ADI
- How to Avoid Acesulfame K
- Conclusion
- Research Papers
- Connections
- Featured Videos
Key Harms at a Glance
- Methylene chloride residue – EPA Group B2 probable human carcinogen used as a solvent in Ace-K synthesis.
- Inadequate original safety testing – 80-week studies, small sample sizes, industry-funded; CSPI petitions for better testing were denied.
- Thyroid disruption signal – Animal evidence of TSH-axis interference.
- Placental and breast-milk transfer – Fetal and infant exposure documented.
- Gut microbiome disruption – Increases Bacteroides and reduces beneficial genera in mice; associated weight gain.
- NutriNet-Santé cohort – Associations with increased cancer and cardiovascular disease risk.
- No long-term RCTs in humans after 35+ years of widespread consumption.
What Is Acesulfame Potassium?
Acesulfame potassium is a synthetic sweetener discovered in 1967 by German chemist Karl Clauss at Hoechst AG (now part of Nutrinova). Chemically, it is the potassium salt of 6-methyl-1,2,3-oxathiazine-4(3H)-one 2,2-dioxide. It was approved by the FDA in 1988 for use in specific food categories and received general-purpose approval in 2003.
Ace-K has a clean, quickly-perceived sweet taste that is often described as having a slightly bitter or metallic aftertaste, particularly at higher concentrations. To mask this bitterness, it is almost always combined with other sweeteners, most commonly aspartame or sucralose. This blending strategy means that consumers of "sugar-free" or "zero-calorie" products are typically ingesting multiple artificial sweeteners simultaneously, with unknown interactive effects.
Methylene Chloride: A Carcinogen in Your Sweetener
One of the most concerning aspects of acesulfame potassium is that its manufacturing process involves methylene chloride (dichloromethane, DCM), a volatile organic solvent that is classified as a probable human carcinogen. Methylene chloride is used as a solvent during the synthesis of Ace-K, and trace residues may remain in the final product.
- Cancer risk — Methylene chloride has been linked to liver cancer, lung cancer, and other malignancies in animal studies. The U.S. EPA classifies it as a "probable human carcinogen" (Group B2). Chronic exposure has been associated with increased cancer mortality in occupational studies of workers exposed to the chemical.
- Neurological toxicity — Methylene chloride exposure is associated with headaches, dizziness, nausea, memory loss, numbness and tingling in the extremities, and impaired cognitive function. These symptoms overlap with many of the complaints reported by consumers of products containing Ace-K.
- Liver and kidney damage — As a volatile organic compound, methylene chloride is metabolized by the liver and excreted by the kidneys, placing stress on both organs with chronic exposure.
- No safe threshold established — For genotoxic carcinogens, many toxicologists argue that no safe level of exposure exists, as even minimal doses can initiate DNA damage that may lead to cancer over time.
While the food industry argues that only trace amounts of methylene chloride remain in finished Ace-K products, the concept of "acceptable" levels of a carcinogen in a food additive consumed daily by millions of people — including children and pregnant women — raises fundamental ethical and public health questions.
Inadequate Original Safety Testing
The 1988 FDA approval of acesulfame potassium has been widely criticized by independent scientists and consumer advocacy groups as being based on seriously flawed and inadequate studies:
- Short study duration — The longest animal studies submitted for approval lasted only 80 weeks, far shorter than the lifetime bioassays now considered the standard for cancer assessment. Since many cancers have latency periods exceeding 80 weeks in rodents, these abbreviated studies may have missed carcinogenic effects that would have appeared with longer observation.
- Small sample sizes — Several key studies used inadequately small numbers of animals, reducing statistical power and making it difficult to detect meaningful increases in tumor rates.
- Poor study design — The Center for Science in the Public Interest (CSPI) and other independent reviewers have identified numerous methodological flaws in the submitted studies, including inadequate controls, poor pathological examination, and incomplete reporting of results.
- Industry-conducted research — All studies submitted for FDA approval were conducted or commissioned by the manufacturer, Hoechst AG. No independent safety studies were required or conducted prior to approval.
- Ignored tumor findings — Some of the submitted studies did show increases in tumors in exposed animals, but these were dismissed as "not statistically significant" or "within the historical range" — judgments that independent statisticians have questioned.
The Center for Science in the Public Interest (CSPI) formally petitioned the FDA in 1988 and again in 1996 to require better testing before allowing widespread use of Ace-K. These petitions were denied. CSPI has consistently rated acesulfame potassium as one of the food additives to "avoid," noting that "the safety tests of acesulfame-K that were conducted in the 1970s were of mediocre quality."
Found in Thousands of Products, Often Unlisted
Acesulfame potassium has become one of the most ubiquitous sweeteners in the processed food supply, yet its presence is often not prominently disclosed:
- Beverages — Coke Zero Sugar, Pepsi Zero Sugar, Diet Mountain Dew, Powerade Zero, Gatorade Zero, flavored waters, energy drinks, protein shakes, and thousands of other beverages use Ace-K, typically in combination with aspartame or sucralose.
- Sugar-free and "zero" products — Sugar-free gum, candies, mints, ice cream, yogurt, gelatin desserts, and puddings frequently contain Ace-K.
- Baked goods and cereals — Many "light," "reduced sugar," and "sugar-free" baked goods include Ace-K.
- Pharmaceuticals and supplements — Chewable tablets, liquid medications, cough drops, toothpaste, and mouthwash commonly contain Ace-K as a sweetening agent.
- Hidden under generic labels — In some products, particularly pharmaceuticals and flavored supplements, Ace-K may be included under umbrella terms such as "artificial flavors," "sweetening agents," or simply not listed at all if it falls below certain threshold concentrations, making consumer avoidance nearly impossible.
Potential Thyroid Disruption
Animal studies have raised concerns about acesulfame potassium's effects on thyroid function:
- Thyroid hormone disruption — Studies in rats have shown that chronic Ace-K consumption can alter thyroid hormone levels, potentially affecting metabolism, energy regulation, growth, and development. The thyroid gland is exquisitely sensitive to chemical disruption, and even small perturbations in thyroid hormone levels can have significant downstream health effects.
- Thyroid stimulation — Early animal studies submitted to the FDA showed evidence of thyroid gland stimulation in rats fed Ace-K, raising concerns about potential thyroid hyperplasia (abnormal growth) with chronic exposure.
- Endocrine disruption mechanism — As a potassium salt with structural similarities to certain thyroid-active compounds, Ace-K may interfere with iodine uptake or thyroid hormone synthesis, though the precise mechanism has not been fully elucidated due to the lack of modern studies investigating this endpoint.
Given that thyroid disorders affect approximately 20 million Americans and that subclinical thyroid dysfunction is even more prevalent, the potential for a widely consumed food additive to contribute to thyroid disruption warrants serious investigation. Yet no comprehensive modern studies on Ace-K and thyroid function have been conducted.
Pregnancy Concerns
The safety of acesulfame potassium during pregnancy is of particular concern due to several factors:
- Placental transfer — Studies have demonstrated that Ace-K crosses the placental barrier and reaches the developing fetus. Unlike many substances that are filtered or metabolized before reaching the fetus, Ace-K appears to pass through relatively unchanged.
- Breast milk presence — Ace-K has been detected in human breast milk, meaning that nursing infants are exposed to this synthetic compound during a critical period of development.
- Taste preference development — Prenatal and early postnatal exposure to sweet-tasting compounds can influence the development of taste preferences, potentially programming a preference for intensely sweet foods that persists throughout life.
- Developmental vulnerability — The developing fetal brain, endocrine system, and gut microbiome are particularly vulnerable to chemical disruption. The effects of chronic Ace-K exposure during these critical developmental windows have never been adequately studied in humans.
- No adequate pregnancy safety studies — Despite its widespread presence in the food supply, no randomized controlled trials have evaluated the safety of Ace-K consumption during human pregnancy. Current safety assurances are based entirely on limited animal data.
Neurological Effects
Emerging research suggests that acesulfame potassium may have adverse effects on the nervous system:
- Cognitive impairment in animal models — Studies have shown that chronic Ace-K consumption can impair learning and memory in mice. A study published in PLOS ONE found that mice exposed to Ace-K through their mothers' consumption during pregnancy showed impaired cognitive function, suggesting developmental neurotoxicity.
- Neurotransmitter disruption — Ace-K has been shown to affect levels of key neurotransmitters including serotonin, dopamine, and GABA in the brains of experimental animals. These neurotransmitters are critical for mood regulation, reward processing, and cognitive function.
- Metabolic effects on the brain — Sweet taste receptors in the brain respond to Ace-K, and the dissociation between sweet taste signaling and caloric content can disrupt brain reward pathways, potentially contributing to overeating, addiction-like behavior patterns, and metabolic dysregulation.
- Methylene chloride residue effects — The neurological symptoms associated with methylene chloride exposure (headaches, dizziness, cognitive impairment) may be relevant even at the trace levels present in Ace-K, particularly with chronic daily consumption over years or decades.
Virtually No Long-Term Human Studies
Perhaps the most troubling aspect of acesulfame potassium's regulatory status is the near-total absence of long-term human safety data:
- No long-term human clinical trials — Despite being consumed by millions of people daily for over 35 years, no randomized controlled trials have evaluated the long-term health effects of Ace-K in humans. The current acceptable daily intake (ADI) is based entirely on animal studies conducted in the 1970s and 1980s.
- Limited epidemiological data — Most epidemiological studies on artificial sweeteners do not disaggregate the effects of individual sweeteners, making it difficult to isolate Ace-K's specific contribution to health outcomes. The few studies that have looked at Ace-K specifically (such as the NutriNet-Santé cohort) have found associations with increased cancer and cardiovascular disease risk.
- Interaction effects unknown — Because Ace-K is almost always consumed in combination with other sweeteners, the potential for synergistic or additive toxic effects is entirely uninvestigated. Consumers of diet beverages typically ingest Ace-K along with aspartame, sucralose, or both — combinations that have never been tested for long-term safety.
- Dose creep — The proliferation of "zero-calorie" and "sugar-free" products means that actual daily intake of Ace-K may significantly exceed the levels anticipated when the ADI was established. A person who drinks diet soda, chews sugar-free gum, uses sugar-free condiments, and takes sweetened medications may consume far more Ace-K than the original safety assessments contemplated.
Ubiquitous in "Sugar-Free" and "Zero" Products
The marketing of "sugar-free" and "zero" products has exploded in recent decades, driven by consumer demand for lower-calorie options and the food industry's desire to maintain the sweet taste profiles that drive sales. Acesulfame potassium has been a major beneficiary of this trend, largely because of its practical advantages as a sweetener ingredient:
- Heat stability — Unlike aspartame, Ace-K is relatively stable at high temperatures, making it suitable for baked goods and processed foods that undergo heat treatment during manufacturing.
- Synergistic sweetening — When combined with other sweeteners, Ace-K enhances overall sweetness perception while masking the off-flavors of other sweeteners. This makes it invaluable to food formulators seeking a sugar-like taste profile.
- Long shelf life — Ace-K is chemically stable in aqueous solutions and does not degrade over time like aspartame, giving products a longer shelf life.
- Cost-effective — Its intense sweetness (200 times that of sugar) and chemical stability make it one of the most economical sweetening options for manufacturers.
These practical advantages explain why Ace-K appears in an ever-expanding range of products. However, the consumer health implications of this expanding exposure have received woefully inadequate scientific attention.
The Gut Microbiome Connection
Like other artificial sweeteners, acesulfame potassium has been shown to disrupt the gut microbiome. A 2017 study published in PLOS ONE found that Ace-K consumption altered the composition of gut bacteria in mice after just four weeks, with changes in key bacterial populations linked to body weight regulation and metabolic function. The study found increases in Bacteroides species and decreases in Clostridium and other beneficial genera.
A separate study found that Ace-K exposure altered gut bacterial gene expression, particularly genes involved in energy metabolism and inflammation. These microbiome changes were associated with increased body weight gain in male mice, consistent with the broader pattern of artificial sweeteners promoting rather than preventing obesity through microbiome-mediated mechanisms.
Safety Thresholds & ADI
- FDA ADI – 15 mg/kg body weight/day.
- JECFA ADI – 0–15 mg/kg body weight/day.
- EFSA ADI – 9 mg/kg body weight/day (more conservative).
- WHO 2023 guideline – Advises against using non-sugar sweeteners for weight control in adults or children (JAMA summary).
- NTP review (2023) – Ongoing systematic review of long-term safety evidence for non-nutritive sweeteners.
How to Avoid Acesulfame K
- Read ingredient labels for “acesulfame potassium,” “acesulfame K,” “Ace-K,” “Sunett,” or “Sweet One,” and the EU code E950.
- Avoid “zero” and “sugar-free” beverages, gums, and protein powders, where Ace-K is nearly universal.
- Check medications and children’s vitamins – ask the pharmacist for dye- and Ace-K-free alternatives.
- Check toothpaste and mouthwash, which commonly contain Ace-K.
- Prefer whole foods and unsweetened beverages (water, plain tea, black coffee).
- If craving sweetness, use small amounts of whole-food sources such as fruit, or physiologically neutral options like allulose or monk fruit (without added Ace-K blend).
Conclusion: The Sweetener You Didn't Know You Were Eating
Acesulfame potassium exemplifies the failures of the modern food additive regulatory system: a synthetic chemical approved based on inadequate, industry-conducted studies in the 1970s and 1980s, manufactured using a known carcinogen, never subjected to long-term human safety testing, and now consumed by millions of people daily in an ever-growing number of products. Its deliberate pairing with other sweeteners makes it nearly impossible for consumers to track their total exposure, and its frequent omission from prominent labeling means that many people consume it without their knowledge or consent.
Until comprehensive, independent, long-term human studies are conducted, the safety of acesulfame potassium remains an open question — one that should concern anyone who consumes "sugar-free," "zero-calorie," or "diet" products on a regular basis. The precautionary principle demands that we not wait for definitive proof of harm before exercising caution with a chemical whose safety was never adequately established in the first place.
Research Papers
- Bian X, Chi L, Gao B, Tu P, Ru H, Lu K. The artificial sweetener acesulfame potassium affects the gut microbiome and body weight gain in CD-1 mice. PLOS ONE. 2017;12(6):e0178426.
- Debras C, Chazelas E, Srour B, et al. Artificial sweeteners and cancer risk: results from the NutriNet-Santé population-based cohort study. PLOS Medicine. 2022;19(3):e1003950.
- Debras C, Chazelas E, Sellem L, et al. Artificial sweeteners and risk of cardiovascular diseases in the prospective NutriNet-Santé cohort. BMJ. 2022;378:e071204.
- WHO. Use of non-sugar sweeteners: WHO guideline. 2023.
- PubMed topic search: acesulfame potassium safety
- PubMed topic search: acesulfame K gut microbiome
- PubMed topic search: acesulfame placental transfer
- PubMed topic search: acesulfame breast milk
- PubMed topic search: methylene chloride carcinogen occupational
- PubMed topic search: non-nutritive sweetener glucose tolerance
- PubMed topic search: artificial sweetener thyroid function
- PubMed topic search: artificial sweetener cognitive impairment mice
Connections
- Artificial Sweeteners
- Aspartame
- Sucralose
- US vs EU Food Safety
- MSG
- Preservatives
- Food Dyes
- Food Additives
- Gut Microbiome
- Type 2 Diabetes
- Cancer
- Gut Brain Axis
- Cardiovascular Disease
- Potassium
- Dizziness
- Sodium Nitrite
- Yellow 5 Tartrazine
- Potassium Sorbate