PCOS (Polycystic Ovary Syndrome)
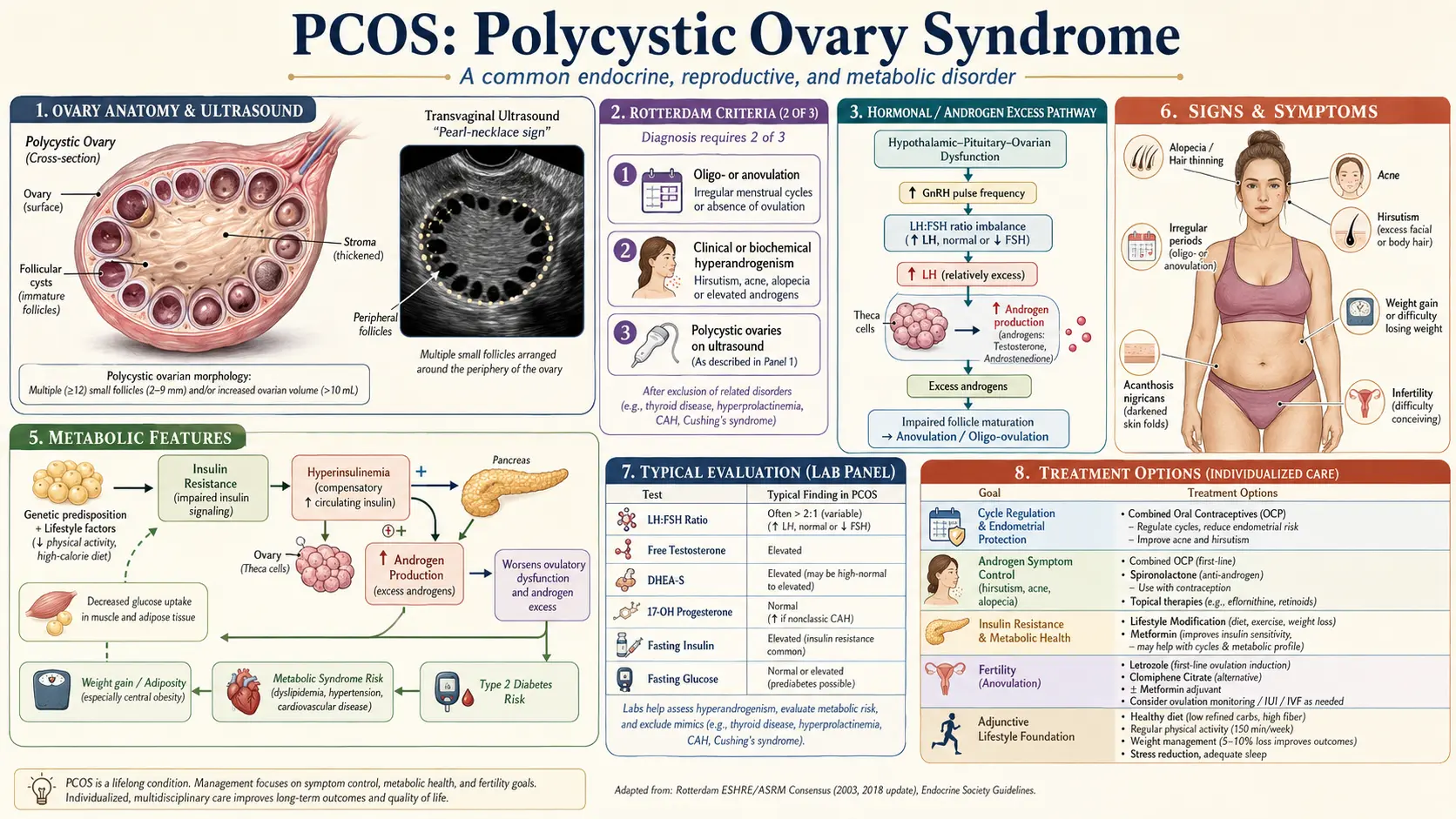
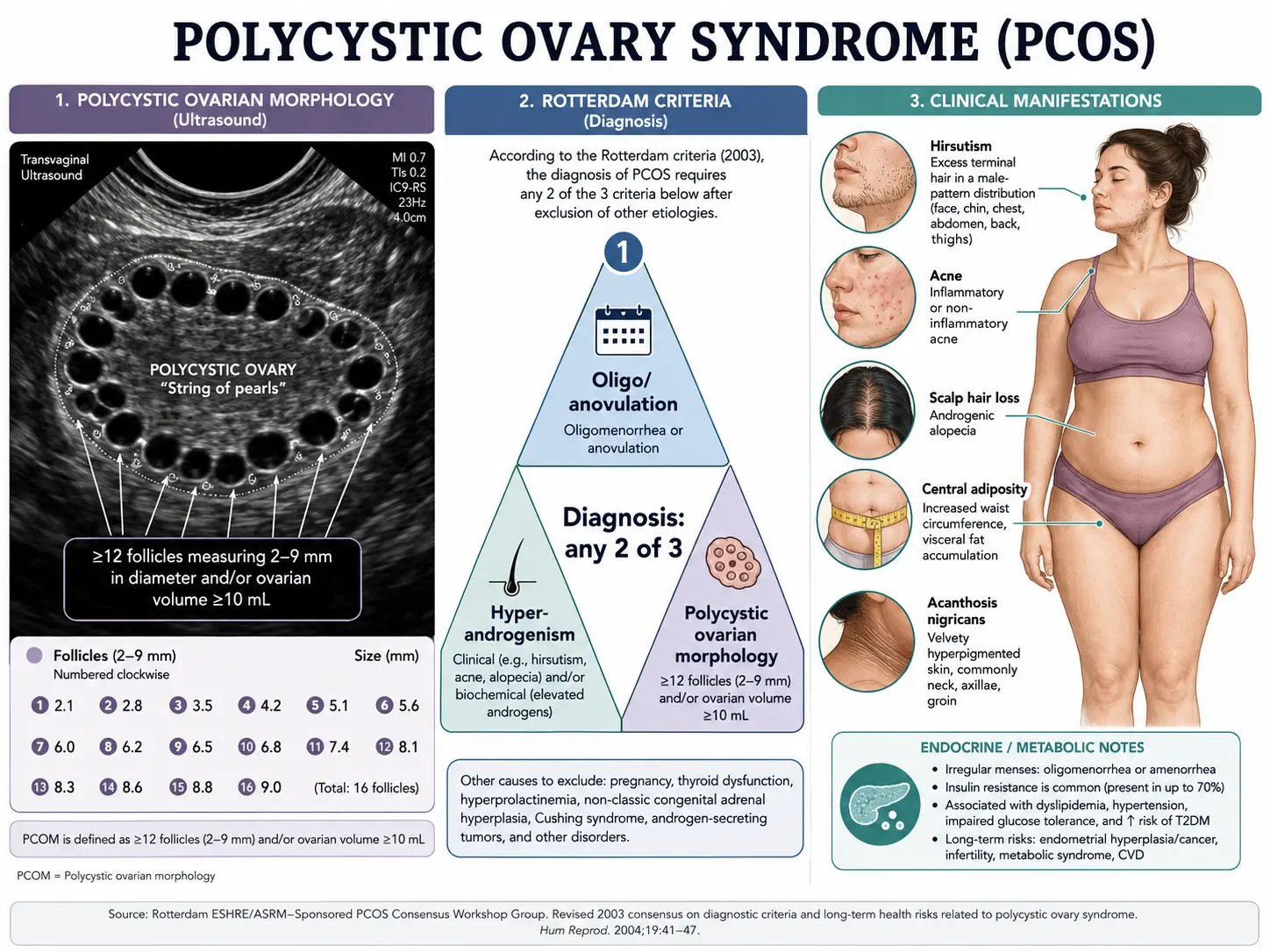
Table of Contents
- What is PCOS?
- Rotterdam Criteria
- Insulin Resistance — The Driver
- Symptoms and Manifestations
- Diagnosis
- Treatment Options
- Long-term Health Risks
- Research Papers
- Connections
- Featured Videos
What is PCOS?
Polycystic ovary syndrome (PCOS) is the most common endocrine disorder in reproductive-age women, affecting an estimated 6–12% of women worldwide depending on the diagnostic criteria used. It is the leading cause of anovulatory infertility and a significant contributor to lifelong metabolic, cardiovascular, and reproductive complications.
Despite the name, PCOS is not primarily a disease of cysts. The classic ultrasound finding — multiple small follicles arrayed around the periphery of an enlarged ovary (the so-called "string of pearls") — reflects arrested follicular development, not true ovarian cysts. PCOS is best understood as a systemic hormonal and metabolic syndrome in which insulin resistance, ovarian and adrenal androgen excess, and disordered ovulation interact to produce the clinical picture.
PCOS typically emerges around puberty or in the late teens and twenties, but it often goes undiagnosed for years because its symptoms — irregular periods, acne, weight gain, unwanted hair growth — are dismissed as cosmetic or attributed to lifestyle. Early diagnosis matters: women with PCOS face a roughly fourfold increased risk of type 2 diabetes, elevated risk of cardiovascular disease, and higher rates of endometrial cancer, sleep apnea, non-alcoholic fatty liver disease, and mood disorders.
Rotterdam Criteria
The Rotterdam consensus criteria (2003, reaffirmed by international guidelines through 2023) remain the dominant diagnostic framework. A patient must meet at least two of three features after other causes have been excluded:
- Oligo- or anovulation — menstrual cycles longer than 35 days, fewer than eight cycles per year, or absent periods (amenorrhea) for three months or more.
- Clinical or biochemical hyperandrogenism — clinical signs include hirsutism (male-pattern terminal hair on the face, chest, abdomen, or back), persistent acne, or androgenic scalp hair loss; biochemical markers include elevated total or free testosterone, elevated free androgen index, or elevated DHEA-S.
- Polycystic ovarian morphology on ultrasound — classically defined as ≥12 follicles measuring 2–9 mm in either ovary, or ovarian volume >10 mL. With the higher resolution of modern transvaginal probes, the 2018 international guideline raised the follicle threshold to ≥20 per ovary when high-frequency transducers (≥8 MHz) are used.
Exclusion of mimics is mandatory. Several conditions produce overlapping features and must be ruled out before a PCOS diagnosis is made:
- Thyroid dysfunction — check TSH (and free T4 if abnormal).
- Hyperprolactinemia — measure prolactin; elevated levels suppress ovulation.
- Non-classical congenital adrenal hyperplasia (NCAH) — measure 17-hydroxyprogesterone (early-morning, follicular phase).
- Cushing's syndrome — consider when there is rapid weight gain, central obesity with thin extremities, purple striae, or proximal muscle weakness; screen with overnight dexamethasone suppression or 24-hour urinary free cortisol.
- Androgen-secreting tumors — suspect with rapid-onset virilization or markedly elevated testosterone (typically >200 ng/dL) or DHEA-S (>700 µg/dL).
- Premature ovarian insufficiency — elevated FSH points away from PCOS.
The Rotterdam criteria define four PCOS phenotypes based on which features are present (A: all three; B: hyperandrogenism + anovulation; C: hyperandrogenism + polycystic morphology; D: anovulation + polycystic morphology). Phenotypes A and B (the "classic" hyperandrogenic phenotypes) carry the highest metabolic risk.
Insulin Resistance — The Driver
Approximately 70% of women with PCOS have measurable insulin resistance, and the proportion climbs higher among women with the classic hyperandrogenic phenotype or with overweight/obesity. Insulin resistance is not merely an associated feature — it is, for most patients, the central pathophysiologic driver of the syndrome.
The mechanism unfolds in three steps:
- Compensatory hyperinsulinemia. Peripheral tissues (muscle, liver, adipose) become insulin-resistant, but the pancreas responds by pumping out more insulin to maintain normoglycemia. Fasting and post-prandial insulin levels rise.
- Insulin acts directly on the ovaries and adrenals. Unlike skeletal muscle, the ovarian theca cells and the adrenal cortex remain insulin-sensitive. High insulin amplifies LH-driven androgen production by theca cells (testosterone, androstenedione) and also stimulates adrenal androgen output (DHEA-S).
- Insulin suppresses sex hormone-binding globulin (SHBG). Hyperinsulinemia inhibits hepatic SHBG synthesis. Less SHBG means a larger fraction of circulating testosterone is unbound and bioactive — even when total testosterone is only mildly elevated, free testosterone can be substantially higher.
The downstream effect is a self-reinforcing cycle: insulin resistance → hyperinsulinemia → ovarian androgen excess → visceral adiposity and inflammation → worsening insulin resistance.
How insulin resistance is assessed. No single test is definitive, but useful measures include:
- Fasting insulin — values >10–12 µIU/mL after an overnight fast suggest hyperinsulinemia (some practitioners use a tighter target of <7).
- HOMA-IR (fasting insulin × fasting glucose / 405) — values >2.0–2.5 indicate insulin resistance; >3.5 is significant.
- Two-hour oral glucose tolerance test (OGTT) with paired insulin measurements — the most sensitive test for early dysglycemia and reactive hyperinsulinemia. Recommended at diagnosis and then every 1–3 years.
- HbA1c — less sensitive than OGTT in PCOS but useful for tracking.
- Lipid panel and waist circumference — PCOS overlaps heavily with metabolic syndrome (low HDL, elevated triglycerides, central adiposity, hypertension, impaired glucose).
Recognizing insulin resistance reframes treatment. Many of the most effective PCOS interventions — weight loss, metformin, inositol, GLP-1 agonists, low-glycemic diet, resistance training — work primarily by improving insulin sensitivity, with the reproductive and androgenic improvements following downstream.
Symptoms and Manifestations
PCOS presents along a wide spectrum. Two women with the same diagnosis may look entirely different clinically. Common manifestations include:
Reproductive
- Irregular or absent periods — oligomenorrhea (cycles >35 days) or amenorrhea; some women have unpredictable, heavy menstrual bleeding when periods do occur.
- Anovulatory infertility — difficulty conceiving because ovulation is inconsistent or absent.
- Increased miscarriage risk — especially when insulin resistance is uncontrolled.
Androgenic (skin and hair)
- Hirsutism — coarse, dark, terminal hair growth in male-pattern distribution (upper lip, chin, sideburns, chest, lower abdomen, inner thighs, lower back). Severity is graded with the modified Ferriman-Gallwey score (≥4–8 considered abnormal depending on ethnicity).
- Persistent or cystic acne — especially along the jawline, chin, and upper back, frequently flaring premenstrually.
- Androgenic alopecia — thinning of the scalp hair, particularly along the crown and central parting.
Metabolic
- Weight gain with central (visceral) adiposity — even women with PCOS who are normal weight often carry disproportionate fat around the waist.
- Acanthosis nigricans — velvety, dark, thickened skin patches in the neck, armpits, groin, or under the breasts; a clinical fingerprint of insulin resistance.
- Skin tags — another insulin-resistance marker.
- Sugar and carbohydrate cravings, post-meal energy crashes, and reactive hypoglycemia.
Other
- Mood symptoms — depression and anxiety are roughly twice as common in PCOS as in age-matched controls; eating-disorder prevalence is also elevated.
- Sleep apnea — under-recognized and disproportionately common, even in lean PCOS patients.
- Fatigue, brain fog, and reduced exercise tolerance.
The presentation often shifts across the lifespan. Adolescents may show acne and irregular cycles; women in their twenties and thirties more commonly seek care for fertility concerns; older women with long-standing PCOS frequently present with metabolic complications — type 2 diabetes, hypertension, fatty liver.
Diagnosis
A complete PCOS workup combines clinical history, physical examination, targeted laboratory tests, and pelvic ultrasound.
History
- Menstrual history from menarche onward — cycle length, regularity, flow, age of first irregularity.
- Onset and progression of acne, hirsutism, scalp hair changes.
- Weight trajectory and dietary patterns; family history of PCOS, type 2 diabetes, cardiovascular disease.
- Current and prior contraceptive use (hormonal contraception masks both menstrual irregularity and androgen levels).
- Reproductive plans — influences treatment selection.
Physical examination
- BMI, waist circumference, blood pressure.
- Hirsutism scoring (modified Ferriman-Gallwey).
- Inspection for acne distribution, scalp hair density, acanthosis nigricans, skin tags, abdominal striae (rule out Cushing's).
- Thyroid palpation.
Laboratory tests
Hormones should ideally be drawn in the early follicular phase (cycle days 2–5) for menstruating women, or any time for amenorrheic women. Hormonal contraception should be discontinued for at least three months for accurate androgen assessment when feasible.
- Total and free testosterone — total typically mildly elevated; free testosterone is the more sensitive measure.
- Sex hormone-binding globulin (SHBG) — usually low; allows calculation of free androgen index.
- DHEA-S — reflects adrenal androgen production; markedly elevated values raise suspicion of adrenal tumor or NCAH.
- 17-hydroxyprogesterone — rules out non-classical CAH.
- LH and FSH — many PCOS patients show an LH:FSH ratio >2:1, but this is not required for diagnosis and is not part of the Rotterdam criteria.
- Anti-Müllerian hormone (AMH) — commonly elevated in PCOS, reflecting the increased follicle pool. Helpful when ultrasound is inconclusive or unavailable, though not yet a formal Rotterdam criterion.
- Estradiol, prolactin, TSH — rule out alternative causes of menstrual irregularity.
- Fasting glucose, fasting insulin, HbA1c, 2-hour OGTT — assess insulin resistance and dysglycemia.
- Lipid panel — total cholesterol, LDL, HDL, triglycerides.
- Liver enzymes (AST/ALT) and ferritin — screen for non-alcoholic fatty liver disease.
- Cortisol screening if Cushing's features are present.
Imaging
- Transvaginal pelvic ultrasound is the standard. Transabdominal ultrasound is acceptable in adolescents or patients who have never been sexually active, although it is less sensitive.
- Ultrasound is not recommended for diagnosis within 8 years of menarche because polycystic ovarian morphology is common in healthy adolescents.
- Look for follicle count per ovary, ovarian volume, endometrial thickness, and any focal lesion.
Treatment Options
There is no "cure" for PCOS, but the syndrome responds well to a layered approach. Treatment is guided by the patient's primary concern — menstrual regulation, hirsutism, acne, fertility, or metabolic risk — while always addressing the underlying insulin resistance.
Lifestyle — the foundation
- Weight loss of 5–10% in women with overweight/obesity restores ovulatory cycles in roughly half of patients, lowers free testosterone, improves insulin sensitivity, and reduces miscarriage risk. Even modest loss has outsized benefit.
- Diet — lower-glycemic, Mediterranean-style, or anti-inflammatory patterns outperform low-fat diets in PCOS. Adequate protein, fiber, and healthy fats; limit refined carbohydrates and added sugars. There is no single "PCOS diet," but consistency and quality matter more than precise macronutrient ratios.
- Exercise — combined aerobic and resistance training is most effective. Resistance training is particularly valuable for muscle insulin sensitivity. Aim for 150 minutes of moderate aerobic activity plus 2–3 strength sessions per week.
- Sleep and stress — 7–9 hours nightly; treat sleep apnea aggressively when present. Chronic cortisol elevation worsens insulin resistance.
Metformin
- An insulin-sensitizing biguanide; the most extensively studied pharmacologic agent in PCOS.
- Improves insulin sensitivity, lowers fasting insulin and androgens, restores ovulation in many patients, and reduces progression to type 2 diabetes.
- Typical dose 1500–2000 mg/day in divided doses; extended-release formulation reduces gastrointestinal side effects.
- First-line for PCOS-associated dysglycemia or impaired glucose tolerance.
Inositol (myo-inositol and D-chiro-inositol)
- Naturally occurring insulin-sensitizing carbocyclic sugars. The 40:1 ratio of myo-inositol to D-chiro-inositol approximates the physiologic ratio in healthy ovaries and is the most-studied formulation.
- Typical dose: 4 g myo-inositol + 100 mg D-chiro-inositol daily, divided.
- Improves ovulation rates, oocyte quality, and insulin sensitivity with a side-effect profile often better-tolerated than metformin.
- Frequently combined with metformin or used as a metformin alternative when GI tolerance is an issue.
Combined oral contraceptives (COCs)
- First-line for menstrual regulation and androgenic skin/hair symptoms in women not seeking pregnancy.
- Suppress ovarian androgen production, increase SHBG (lowering free testosterone), and provide endometrial protection against unopposed estrogen.
- Pills containing low-androgenicity or anti-androgenic progestins (drospirenone, cyproterone, dienogest) are typically preferred. Avoid or minimize progestins with high androgenic activity.
- Choice should weigh thrombotic risk, blood pressure, migraine history, and metabolic profile.
Anti-androgens
- Spironolactone 50–200 mg/day — the most common anti-androgen used for hirsutism and androgenic acne. Effects build slowly (3–6 months for hair, faster for acne). Always pair with reliable contraception in reproductive-age women because spironolactone is teratogenic.
- Other options include cyproterone acetate (not available in the US), finasteride, and bicalutamide; these are less commonly first-line.
Ovulation induction (for fertility)
- Letrozole — an aromatase inhibitor; now first-line ovulation-induction agent for PCOS based on superior live-birth rates compared with clomiphene (PPCOS II trial). Typical dose 2.5–7.5 mg on cycle days 3–7.
- Clomiphene citrate — selective estrogen receptor modulator; second-line. Typical dose 50–150 mg on cycle days 3–7 or 5–9.
- Metformin may be added when insulin resistance is prominent or for combination protocols.
- Gonadotropins and IVF — reserved for patients who fail oral agents; carry higher risk of multiple pregnancy and ovarian hyperstimulation syndrome (OHSS), to which PCOS patients are particularly susceptible.
GLP-1 receptor agonists
- Semaglutide, liraglutide, tirzepatide (a dual GIP/GLP-1) — produce substantial weight loss and improve insulin sensitivity. Increasing real-world use in PCOS, especially when obesity is the dominant problem and lifestyle alone has plateaued.
- Improvements in ovulation, androgens, and metabolic parameters are mediated largely through weight loss and direct metabolic effects.
- Not approved for use in pregnancy — require effective contraception and a clear off-ramp before conception attempts.
Berberine
- A plant alkaloid with insulin-sensitizing and AMPK-activating effects comparable to metformin in head-to-head PCOS studies.
- Typical dose 500 mg three times daily with meals.
- May improve cycle regularity, lipids, and insulin sensitivity. A reasonable alternative or adjunct when metformin is poorly tolerated. See Berberine.
Long-term Health Risks
PCOS is a lifelong condition with cumulative health implications. Long-term surveillance and proactive management of these risks should be part of every PCOS care plan:
- Type 2 diabetes — women with PCOS face roughly a 4-fold increased risk of developing T2D compared with age-matched controls. Onset is often a decade earlier than in the general population. Periodic OGTT is recommended even for normal-weight patients.
- Cardiovascular disease — PCOS is associated with elevated rates of hypertension, dyslipidemia (low HDL, high triglycerides, atherogenic small-dense LDL), endothelial dysfunction, and metabolic syndrome. Cardiovascular risk should be assessed and managed early.
- Endometrial hyperplasia and endometrial cancer — chronic anovulation produces unopposed estrogen exposure to the uterine lining. Women who go more than three months without a period need either cyclic progestin, a combined oral contraceptive, or a progestin-releasing IUD to protect the endometrium. Risk of endometrial cancer is increased roughly 3-fold.
- Non-alcoholic fatty liver disease (NAFLD/MASLD) — up to half of PCOS patients have hepatic steatosis on imaging; insulin resistance is the shared driver.
- Obstructive sleep apnea — substantially more common in PCOS, even in lean patients; should be screened by symptom history (snoring, daytime sleepiness, witnessed apneas) and treated when present.
- Depression and anxiety — roughly double the prevalence in age-matched controls. Eating disorders, body-image concerns, and reduced quality of life are also more common. Mental health should be part of routine PCOS care.
- Adverse pregnancy outcomes — gestational diabetes (3–4× risk), pregnancy-induced hypertension, preeclampsia, preterm birth, and miscarriage are all elevated. Pre-conception optimization (weight, glucose control, blood pressure) and early gestational diabetes screening (often at first prenatal visit, with repeat 24–28 weeks) are recommended.
- Vitamin D deficiency — common in PCOS and linked to worsened insulin resistance and ovulatory dysfunction; testing and supplementation are appropriate.
The good news is that the same interventions that address the day-to-day symptoms — weight management, insulin sensitization, anti-inflammatory diet, regular exercise, sleep optimization — also reduce these long-term risks. PCOS rewards consistency.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- PCOS Rotterdam criteria
- PCOS insulin resistance
- PCOS metformin
- PCOS inositol myo-inositol
- PCOS letrozole fertility
- PCOS spironolactone hirsutism
- PCOS lifestyle weight loss
- PCOS GLP-1 agonist
- PCOS cardiovascular risk
- PCOS endometrial cancer
- PCOS pregnancy outcomes
- PCOS phenotypes hyperandrogenism
Connections
- Berberine
- Insulin Resistance
- Polycystic Ovary Syndrome
- Fasting Insulin
- Anti-Inflammatory Diet
- Diabetes
- Thyroid Disorders
- Magnesium
- Hormone Panel
- Vitamin D3
- Omega-3 Fatty Acids
- NAC
- Testosterone
- Inositol
- Acne
- Obesity
- Infertility
- DHEA-S