Bladder & Bowel Endometriosis
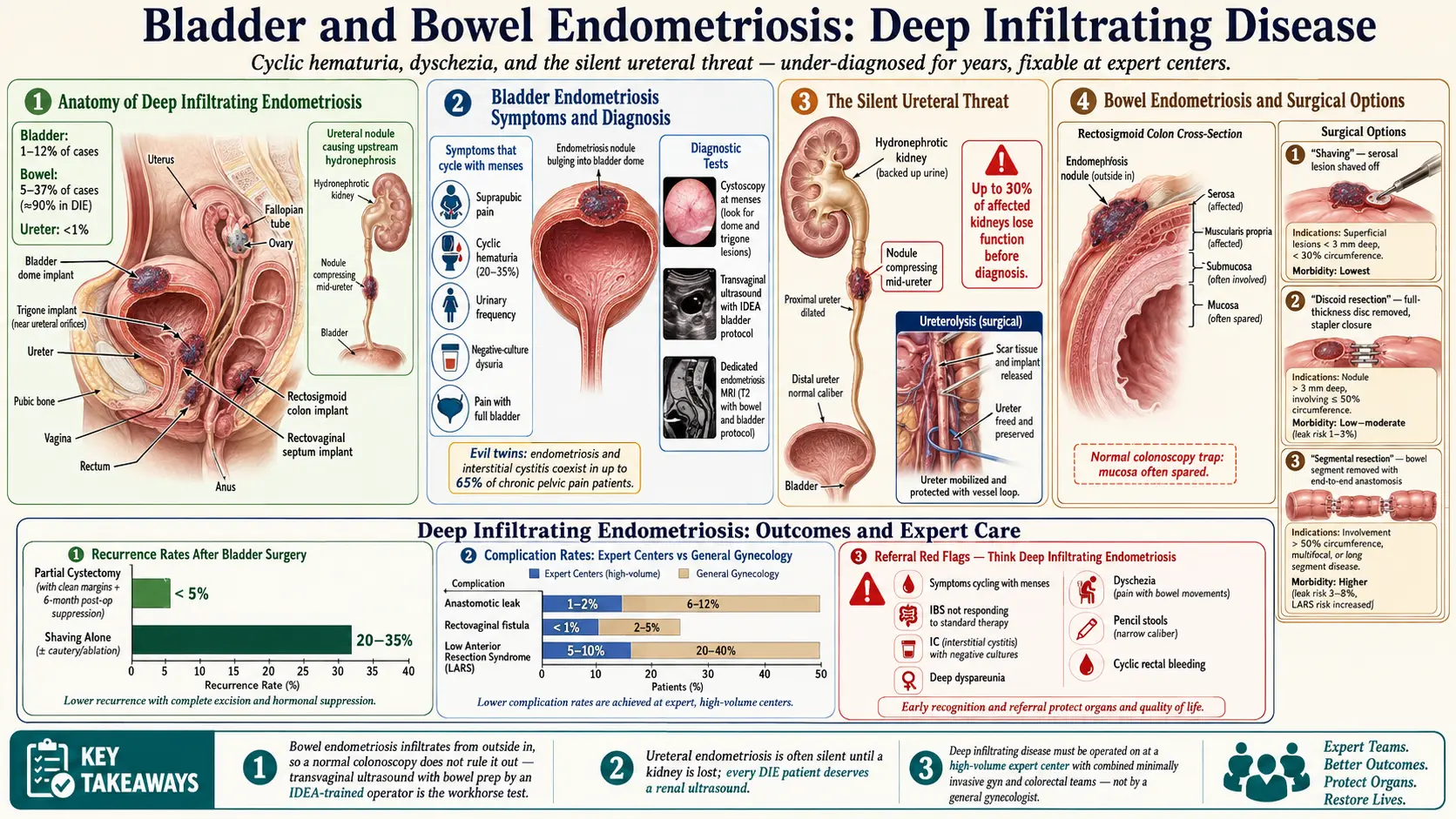
Endometriosis is not confined to the ovaries and peritoneum. In a meaningful minority of patients, the disease invades the wall of the bladder, the ureter, or the bowel — a form called deep infiltrating endometriosis (DIE). These lesions are under-recognized, often misdiagnosed for years as interstitial cystitis, irritable bowel syndrome, or "just bad periods," and they can do real structural damage. The good news: when the right imaging is done and the right surgeon operates, outcomes are generally excellent.
This article is for patients who suspect urinary or bowel involvement, have already been told they "just have IBS" or "just have IC," or are preparing for surgery and want to know what the options are and what to ask for.
Table of Contents
- How Common Is It?
- Bladder Endometriosis — Symptoms
- Bladder Endometriosis — Diagnosis
- Bladder Endometriosis — Surgery
- The Silent Danger: Ureteral Endometriosis
- Bowel Endometriosis — Symptoms
- Bowel Endometriosis — Diagnosis
- Bowel Endometriosis — Surgical Options
- Hormonal Suppression, Nutrition, and Expert-Center Referral
- Key Research Papers
- PubMed Topic Searches
- Connections
- Featured Videos
How Common Is It?
The numbers vary because the disease is chronically under-diagnosed, but published series give a consistent range:
- Bladder endometriosis: roughly 1–12% of endometriosis cases. It is by far the most common urinary-tract site — about 85% of all urinary endometriosis.
- Bowel endometriosis: roughly 5–37% of endometriosis cases depending on the cohort (specialist-referral centers report higher numbers than general gynecology clinics). In deep infiltrating endometriosis specifically, the bowel is involved in up to 90% of patients.
- Ureteral endometriosis: less than 1% of cases, but disproportionately dangerous because it can quietly destroy a kidney.
Two facts worth internalizing: first, most urinary and bowel endometriosis is never diagnosed, because nobody images for it. Second, the average delay from first symptom to correct diagnosis for any form of endometriosis is 7–10 years — and the delay is often longer when the main complaint is urinary or gastrointestinal rather than pelvic pain, because patients get routed to urology or gastroenterology and the cyclic pattern gets missed.
Bladder Endometriosis — Symptoms
The single most important clue is that symptoms cycle with menses. A flare that lines up with your period almost every month is the signature of endometriosis anywhere in the body.
- Suprapubic pain (right above the pubic bone) that worsens in the days leading up to and during your period.
- Cyclic hematuria — visible or microscopic blood in the urine, appearing only around menstruation. This is the most specific sign, but it is present in only about 20–35% of cases. Its absence does not rule out bladder endometriosis.
- Urinary frequency and urgency, often severe enough to wake you at night.
- Dysuria (burning with urination) that is not explained by infection — urine cultures come back negative.
- Pain with a full bladder that eases after emptying.
This symptom cluster is almost identical to interstitial cystitis/bladder pain syndrome (IC/BPS), which is how bladder endometriosis so often gets missed — and why so many women accumulate years of failed IC treatments before anyone images the bladder for endometrial implants. To complicate matters further, the two conditions genuinely coexist: in a classic study, endometriosis and IC together were dubbed the "evil twins" because up to 65% of women with chronic pelvic pain had both. If your IC is not responding to standard therapy, or if flares track your cycle, push for endometriosis workup.
Bladder Endometriosis — Diagnosis
Getting the diagnosis right requires the right test, done by the right person, at the right time in your cycle.
Cystoscopy. A urologist threads a small camera into the bladder. In deep infiltrating bladder endometriosis, they may see a bluish or reddish nodule bulging into the bladder dome. But here is the trap: cystoscopy is often completely normal, because most bladder endometriosis sits in the outer muscle layer (detrusor) and has not yet eroded through the mucosa. A "normal cystoscopy" does not rule out the disease. Ideally cystoscopy is performed in the premenstrual or menstrual window when lesions are most engorged and visible.
Transvaginal ultrasound with bladder protocol. This is the workhorse test, but only in expert hands. The bladder must be partially filled (about 50–100 mL), and the sonographer must be specifically trained in the IDEA consensus protocol for deep endometriosis. A routine "pelvic ultrasound" is not the same thing. Sensitivity in expert centers exceeds 80% for bladder DIE.
MRI of the pelvis. A dedicated endometriosis MRI protocol (with bowel and bladder preparation) is the other mainstay, and it maps not just the bladder but the uterus, ovaries, ureters, bowel, and pelvic fascia in one scan. If surgery is on the table, most expert centers want an MRI for the operative map.
Urinalysis around menses. A simple urine dipstick or microscopy during your period can catch microscopic hematuria that is absent the rest of the month.
Bladder Endometriosis — Surgery
When a bladder nodule is symptomatic and confirmed on imaging, the gold-standard treatment is laparoscopic or robotic partial cystectomy — the surgeon cuts out the full-thickness disc of bladder wall containing the nodule and closes the bladder in two layers. A Foley catheter stays in place for 7–14 days post-op to let the suture line heal. Most patients go home the same day or the next morning and return to normal voiding within two weeks.
Location on the bladder matters a great deal:
- Bladder dome (the top) — the easy zone. Far from the ureters, far from the trigone. Partial cystectomy here is routine for experienced minimally invasive gyn surgeons.
- Trigone (the triangle at the base where the ureters enter) — far more technically demanding. Surgery risks ureteral injury, requires ureteral stenting, and should only be done at a tertiary expert center. Reimplantation of a ureter may be needed.
Recurrence after complete excision is low — under 5% in most series — provided margins are clean and hormonal suppression is used post-operatively for at least 6 months. Simply shaving off the visible nodule without full-thickness excision carries a much higher recurrence rate and is not recommended.
The Silent Danger: Ureteral Endometriosis
Of all the urinary-tract manifestations, ureteral endometriosis is the one that terrifies specialists, because it is often completely silent until a kidney is already lost. Implants on or around the ureter slowly compress or infiltrate it, urine backs up, the kidney swells (hydronephrosis), and over months to years renal function fades. Some women present with vague flank pain; many have no symptoms at all until imaging for another reason picks up a hydronephrotic kidney. By the time it is found, up to 30% of affected kidneys have irreversibly lost function.
This is why any woman with deep infiltrating endometriosis — especially with endometriomas, bowel involvement, or a palpable nodule — deserves a look at the ureters and kidneys. A renal ultrasound is cheap, non-invasive, and can be done at the same visit as the pelvic scan. Dedicated endometriosis MRI protocols include the ureters. If hydronephrosis is found, ureterolysis (freeing the ureter from scar tissue) or, in severe cases, segmental resection with reimplantation is performed — and the sooner, the better, because renal function lost is not recovered.
Bowel Endometriosis — Symptoms
The rectosigmoid colon (the S-shaped last stretch of colon before the rectum) is the most common bowel site by a wide margin, followed by the rectum itself, then the appendix, cecum, and terminal ileum. Because the rectosigmoid sits right behind the uterus and cervix, endometriosis implants from the pouch of Douglas can invade it directly.
The symptom list overlaps heavily with irritable bowel syndrome, which is why women with bowel endometriosis are frequently told for years that they have IBS:
- Dyschezia — painful bowel movements, especially during menstruation. Often described as a stabbing or "knife-in-the-rectum" sensation.
- Cyclic rectal bleeding — blood in or on the stool, only around your period. Less common than dyschezia but highly specific.
- Alternating constipation and diarrhea that cycles with menses — constipated in the luteal phase, loose during bleeding, for example.
- Small-caliber or "pencil" stools — a narrowed rectosigmoid lumen from an extrinsic nodule.
- Severe bloating — the abdomen distends dramatically, sometimes looking several months pregnant by evening. Commonly called "endo belly." See the Endo Belly article.
- Tenesmus — the constant feeling of needing to defecate even after you just have.
- Deep dyspareunia — pain with deep vaginal penetration, because the nodule sits in the rectovaginal septum.
Two things to note. First, IBS and endometriosis coexist at far above chance rates — estimates run 30–50% overlap — so having one does not protect you from the other. Second, if "IBS" symptoms cycle with your period, it is endometriosis until proven otherwise. See the IBS page for how the two distinguish from each other.
Bowel Endometriosis — Diagnosis
Colonoscopy is usually normal, and that is a trap. Bowel endometriosis typically infiltrates from the outside in — it starts on the serosa and burrows into the muscularis propria, usually stopping before it reaches the mucosa. Because colonoscopy only sees the mucosa, the implants are invisible from inside the bowel. A completely clean colonoscopy does not rule out bowel endometriosis. Colonoscopy is still worth doing to rule out other diseases (inflammatory bowel disease, cancer), but it is not the diagnostic test.
The real tests are:
- Transvaginal ultrasound with bowel prep performed by a sonographer trained in deep endometriosis imaging. Sensitivity for rectosigmoid disease in expert centers is 90% or higher. This is non-invasive, inexpensive, and should be the first step.
- MRI of the pelvis with bowel and rectal preparation, which maps the depth of infiltration (serosa, muscularis, submucosa) and the length of bowel involved — both critical for surgical planning.
- Rectal endoscopic ultrasound in select cases where depth of infiltration is unclear.
The same principle applies as with the bladder: a "routine" pelvic ultrasound at a general imaging center is not the same test as a deep-endometriosis protocol done by a trained operator. If your local sonographer does not know what the IDEA protocol is, go elsewhere.
Bowel Endometriosis — Surgical Options
Three surgical techniques exist, and morbidity rises stepwise with each. The choice depends on the depth of infiltration, size of the nodule, and percentage of bowel circumference involved.
- Shaving. The surgeon carefully shaves the nodule off the bowel wall without entering the lumen. Suitable for superficial disease limited to the serosa and outer muscularis. Lowest morbidity; preserves the bowel; recurrence risk is higher if the nodule extends deeper than it appeared.
- Discoid resection. A full-thickness disc of bowel wall containing the nodule is cut out and the defect is closed, usually with a circular stapler. Appropriate for nodules under about 3 cm involving less than one-third of the bowel circumference. Middle of the road for morbidity.
- Segmental resection. A length of bowel containing the nodule is removed entirely and the two ends are rejoined (anastomosis). Reserved for large nodules (>3 cm), multifocal disease, or involvement of more than a third of the circumference. Highest morbidity: anastomotic leak (1–3%), rectovaginal fistula, low anterior resection syndrome (bowel-habit disturbance that can persist), and a small risk of needing a temporary or permanent ostomy.
Functional outcomes after expert bowel endometriosis surgery are generally excellent — most patients report major improvement in pain, dyschezia, and quality of life — but complications are real and the surgery should never be done by a general gynecologist or general surgeon in isolation. A combined team of a minimally invasive gynecologic surgeon and a colorectal surgeon working together at a high-volume center is the standard of care.
Hormonal Suppression, Nutrition, and Expert-Center Referral
Pre-operative hormonal suppression. Many surgeons place patients on 2–3 months of a GnRH antagonist (elagolix, relugolix-combo) or continuous progestin (dienogest, norethindrone acetate) before surgery. This shrinks lesions, reduces inflammation, and makes the operation technically easier. See the hormonal therapy page for how each class works.
Post-operative suppression. After excision of deep infiltrating disease, continuous progestin or a GnRH antagonist for at least 6–12 months is standard to reduce recurrence. Combined oral contraceptives used cyclically are generally not adequate for DIE — continuous or progestin-only regimens suppress more completely.
Nutrition around surgery. Bowel surgery in particular benefits from prehabilitation: adequate protein (1.2–1.5 g/kg/day), iron repletion if deficient, vitamin D optimization, and reducing inflammatory dietary triggers for the 4–8 weeks before the operation. Post-operatively, a low-residue diet for the first 2–4 weeks gives the anastomosis time to heal. See the diet and lifestyle page for longer-term nutritional support.
Expert-center referral. This cannot be said strongly enough: do not have your deep infiltrating bladder, ureteral, or bowel endometriosis surgery done by a general gyn. Outcomes at high-volume endometriosis centers — places doing hundreds of these operations a year, with dedicated radiology, colorectal, urology, and pelvic floor PT teams — are dramatically better than community outcomes. The right question is not "can you do this surgery?" It is "how many of these do you do a year, what is your complication rate, and can I see your data?" A surgeon who resents that question is not the right surgeon.
In the U.S., useful starting points include the AAGL-accredited Centers of Excellence in Minimally Invasive Gynecology (COEMIG) list and patient advocacy networks like Endometriosis.org and Nancy's Nook. Insurance coverage of out-of-network expert centers is negotiable, particularly when you can document local treatment failures — be prepared to appeal.
Key Research Papers
- Chapron C, et al. Anatomical distribution of deeply infiltrating endometriosis: surgical implications and proposition for a classification. Hum Reprod. 2003.
- Abrao MS, et al. Deep endometriosis infiltrating the recto-sigmoid: critical factors to consider before management. Hum Reprod Update. 2015.
- Maccagnano C, et al. Diagnosis and treatment of bladder endometriosis: state of the art. Urol Int. 2013.
- Guerriero S, et al. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: a consensus opinion from the IDEA group. Ultrasound Obstet Gynecol. 2016.
- Nezhat C, et al. Urinary tract endometriosis: review of 40 years of literature. Fertil Steril. 2012.
- Chiu CL, Hoffman MK. The "evil twins" of chronic pelvic pain syndrome: endometriosis and interstitial cystitis. JSLS. 2012.
PubMed Topic Searches
For deeper reading, the following PubMed searches return current peer-reviewed literature on urinary- and bowel-tract endometriosis:
- Bladder endometriosis
- Ureteral endometriosis and hydronephrosis
- Bowel endometriosis and rectosigmoid disease
- Deep infiltrating endometriosis and MRI imaging
- Transvaginal ultrasound and the IDEA protocol
- Segmental resection outcomes for bowel endometriosis
- Shaving versus discoid resection for bowel endometriosis
- Interstitial cystitis and endometriosis overlap
- Irritable bowel syndrome and endometriosis
- Partial cystectomy for bladder endometriosis
Connections
- Diet and Lifestyle
- Irritable Bowel Syndrome
- Hormonal Therapy Options
- Endo Belly and Gut Connection
- Endometriosis
- Laparoscopy: Excision vs Ablation
- Diagnosis Delay and Imaging
- Pelvic Floor Therapy and Central Sensitization
- Fertility and Endometriosis
- Adenomyosis Overlap
- Interstitial Cystitis
- Full Body MRI
- Urinalysis
- Constipation
- Bloating
- Inflammatory Bowel Disease
- Iron