Endometriosis and Adenomyosis — the Cousin Condition
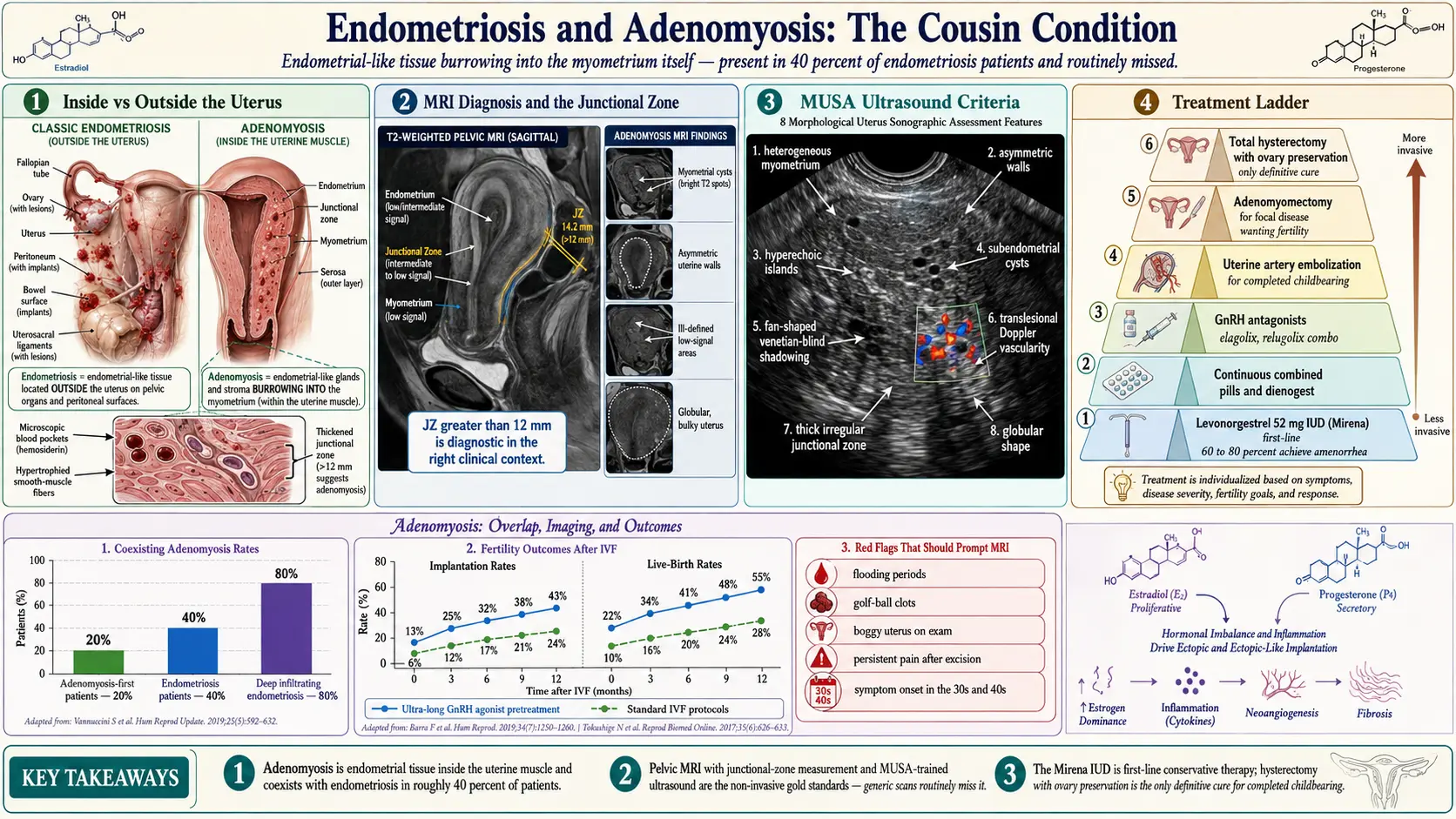
Table of Contents
- What Adenomyosis Is
- How Often Endo and Adeno Overlap
- Symptoms — and Why Attribution Is Hard
- Why the Overlap Matters
- Diagnosis — MRI as the Non-Invasive Gold Standard
- Sonographic Features and the MUSA Criteria
- Clinical Signs Your Doctor Can Feel
- Subtypes: Diffuse vs Focal Adenomyoma
- Treatment Differences from Endometriosis
- Hysterectomy — the Only Definitive Cure
- Fertility and IVF Considerations
- Why an Excision Surgeon Alone Isn't Enough
- Pregnancy with Adenomyosis
- Choosing Medical vs Surgical Management
- When to Suspect Adenomyosis If You Have Endo
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
What Adenomyosis Is
Adenomyosis is endometrial tissue — the same glands and stroma that line the inside of your uterus and shed every month as a period — growing inside the muscular wall of the uterus itself (the myometrium). That is the entire distinction from endometriosis in one sentence. Endometriosis: endometrial-like tissue outside the uterus, seeded on ovaries, peritoneum, bowel, bladder, diaphragm. Adenomyosis: the same kind of tissue burrowing into the uterine muscle.
The practical consequence is dramatic. In endometriosis, the implants bleed into the pelvis and trigger scarring, adhesions, and nerve inflammation in places they should not be. In adenomyosis, the implants bleed inside the uterine muscle, which has nowhere to drain. Each cycle, microscopic pockets of trapped blood swell, the surrounding muscle hypertrophies in response, and the uterus becomes progressively bulky, boggy, and hypercontractile. The resulting periods are not just painful — they are heavy, flooding, and accompanied by cramping that feels deeper and more internal than classic endo pain.
The two diseases share a suspected origin in the junction between endometrium and myometrium (the so-called junctional zone), share estrogen dependence, share response to hormonal suppression, and very often share a patient. They are close enough to be called cousins, and different enough that treating one and ignoring the other leaves half the problem behind.
How Often Endo and Adeno Overlap
The numbers that matter, because they reframe how you should think about your own diagnosis:
- Roughly 40% of women with endometriosis also have adenomyosis when carefully evaluated by MRI or expert ultrasound. Older estimates hovered near 20%; better imaging has pushed the number up.
- Roughly 20% of women diagnosed with adenomyosis also have endometriosis lesions somewhere in the pelvis.
- In women with deep infiltrating endometriosis, coexisting adenomyosis rates climb to 80% or higher in some imaging series.
Read those numbers again. Nearly half of all endometriosis patients have a second, structurally different disease sitting inside the uterus. If you have been told you "just" have endo and your uterus still hurts, those statistics are the first clue that the picture may be incomplete.
Symptoms — and Why Attribution Is Hard
Classic adenomyosis symptoms cluster into four areas:
- Heavy menstrual bleeding (menorrhagia). Soaking a pad or tampon every hour, flooding overnight, passing golf-ball-sized clots, bleeding that lasts more than seven days. This is the most reliable adenomyosis red flag and the one least typical of pure endometriosis.
- Severe cramping during and just before the period, often described as deep, pulling, whole-pelvis pain rather than the sharp, stabbing, position-dependent pain of pelvic endo lesions.
- Dyspareunia (pain with deep intercourse) caused by the bulky uterus itself being hit on deep penetration, not by a retrocervical lesion.
- Chronic pelvic pressure or a feeling of heaviness between periods — patients often describe "feeling my uterus all the time" or a sensation that something is swollen inside.
The attribution problem is obvious: every one of those symptoms overlaps with endometriosis. A patient with both diseases cannot, by symptom history alone, tell her doctor which disease is doing what. This is exactly where imaging earns its keep.
Why the Overlap Matters
Here is the scenario that plays out in gynecology clinics every week. A patient with textbook endometriosis undergoes a skilled excision surgery. Every lesion is mapped, excised, and pathologically confirmed. She recovers, and for a few months things are better. Then the heavy bleeding returns. The deep cramping returns. She returns to her excision surgeon in tears, certain the endo has grown back.
In many of those cases, the endo did not grow back. It was never what was causing the uterine pain and flooding periods in the first place. The second disease — adenomyosis — was untouched by the excision surgery because it lives inside the uterine wall, which the excision surgeon (correctly) did not cut into. The only way to prevent this outcome is to know before surgery that both diseases are present, so both can be planned for.
This is why every endometriosis patient deserves a dedicated uterine imaging evaluation. Not a quick pelvic ultrasound at the end of a routine visit, but a focused look at the uterine wall by someone who knows what adenomyosis looks like.
Diagnosis — MRI as the Non-Invasive Gold Standard
Pelvic MRI is the most reliable non-invasive test for adenomyosis. A good radiology protocol uses T2-weighted sequences to visualize the three uterine layers: the inner endometrium, the thin junctional zone (the inner myometrium), and the outer myometrium. In a healthy uterus, the junctional zone is a thin dark band measuring under 8 mm.
The MRI findings that establish adenomyosis:
- Junctional zone thickening greater than 12 mm — the single most specific sign. A junctional zone between 8 and 12 mm is suggestive; above 12 mm is diagnostic in the right clinical context.
- Globular, enlarged uterus without a focal fibroid to account for the size.
- Myometrial cysts — bright T2 spots within the muscle, representing trapped blood and glandular fluid.
- Ill-defined low-signal areas in the myometrium, indicating smooth-muscle hyperplasia around ectopic endometrium.
- Asymmetric myometrial walls, where one wall is noticeably thicker than the other.
MRI also stages any coexisting deep infiltrating endometriosis — uterosacral nodules, bladder and bowel involvement, endometriomas — in the same scan. If you are paying out of pocket for one imaging study before major surgery, pelvic MRI with an endo-adeno protocol is the one to choose. Insist on a radiologist who reads pelvic MRIs routinely; this is a pattern-recognition specialty.
Sonographic Features and the MUSA Criteria
High-resolution transvaginal ultrasound (TVUS) in expert hands rivals MRI for adenomyosis diagnosis and costs a fraction of the price. In 2015 an international group published the Morphological Uterus Sonographic Assessment (MUSA) consensus criteria, which standardize what sonographers should look for and how to report it. The MUSA features:
- Heterogeneous myometrium — a patchy, disorganized texture instead of the uniform smooth-muscle pattern of a healthy uterus.
- Asymmetric uterine walls — anterior wall visibly thicker than posterior, or vice versa.
- Myometrial or subendometrial cysts — small round anechoic (black) pockets inside the muscle.
- Hyperechoic islands — bright spots within the myometrium representing ectopic endometrial glands.
- Fan-shaped or "venetian-blind" shadowing — a distinctive striped acoustic shadow caused by the alternating densities of muscle and gland.
- Translesional vascularity on color Doppler, with vessels crossing the affected area rather than branching around a discrete mass.
- Thickened, irregular junctional zone on 3D reconstruction.
- Globular uterine shape with an indistinct endomyometrial interface.
The catch is the operator. An average OB/GYN ultrasound performed quickly at the end of a visit will routinely miss adenomyosis. You need a sonographer or sonologist trained in MUSA reporting — typically at an academic center, a dedicated endometriosis imaging practice, or a reproductive imaging clinic. Ask explicitly, "Do you screen for adenomyosis using MUSA criteria?" before booking.
Clinical Signs Your Doctor Can Feel
On a bimanual pelvic exam, adenomyosis often produces a classic finding: a symmetrically enlarged, boggy, tender uterus. "Boggy" is the word experienced clinicians use for a uterus that feels softer and more yielding than normal, as if the muscle is waterlogged — which, in a sense, it is. Compare this with a fibroid uterus, which feels firm and knobby, or a normal uterus, which feels firm and walnut-shaped.
Most adenomyosis shows up in the 30s and 40s, often after childbirth, with symptoms peaking in the decade before menopause. This is an important generational shift from endometriosis, which classically begins in the teens and twenties. An endo patient who had manageable disease in her twenties and now, at 38, faces newly catastrophic periods has not necessarily had her endo worsen — she may have developed adenomyosis on top of it. That said, adolescent and early-twenties adenomyosis is now recognized more often with better imaging, so age alone never excludes the diagnosis.
Subtypes: Diffuse vs Focal Adenomyoma
Adenomyosis comes in two structural patterns, which matter because they respond differently to surgery:
- Diffuse adenomyosis — endometrial tissue scattered throughout the myometrium, producing a globally enlarged, heterogeneous uterus. This is the more common pattern and the one that responds best to hormonal suppression or, ultimately, hysterectomy.
- Focal adenomyosis (adenomyoma) — a discrete nodule of adenomyotic tissue, sometimes mistaken for a fibroid on ultrasound. Focal lesions can occasionally be excised surgically while preserving the uterus (adenomyomectomy).
Some patients have both: a diffuse background with a dominant focal nodule. An expert MRI report will describe which pattern is present, which is critical pre-operative information.
Treatment Differences from Endometriosis
Adenomyosis and endometriosis share some therapies and differ sharply on others. The overlap and the divergences:
Levonorgestrel IUD (Mirena) — First-Line
The 52 mg levonorgestrel intrauterine system (Mirena, Liletta) is the single most effective conservative therapy for adenomyosis and in most guidelines is the first-line recommendation. Local progestin thins and atrophies the ectopic endometrium inside the muscle. Studies show the majority of patients experience substantial pain reduction and roughly 60–80% progress to amenorrhea (no periods) within a year. The IUD lasts up to eight years. For many patients, a Mirena avoids hysterectomy entirely.
Continuous Hormonal Suppression
Same as endometriosis: continuous combined oral contraceptives, norethindrone acetate, dienogest, or DMPA injections. Stopping ovulation and the monthly hormonal cycle quiets both diseases simultaneously. This is an attractive option when a patient has both conditions and wants one therapy that treats both.
GnRH Antagonists
Elagolix (Orilissa) and the relugolix combination (Myfembree, approved for uterine fibroids and endometriosis-associated pain) can be used off-label for adenomyosis with good symptomatic response. They suppress ovarian estrogen production and typically include low-dose add-back hormones to protect bone. Treatment is often limited to two years.
Uterine Artery Embolization (UAE)
Interventional radiologists thread a catheter into the uterine arteries and inject particles that cut off blood flow to the diseased muscle. The ectopic endometrium shrinks. UAE works for both diffuse and focal adenomyosis and avoids surgery. Fertility implications are mixed — pregnancies after UAE are possible but carry increased risks of placentation abnormalities and preterm delivery, so UAE is generally recommended for patients who have completed childbearing.
Adenomyomectomy
Surgical excision of a focal adenomyoma, preserving the uterus. This is a difficult operation with no clean surgical plane (unlike a fibroid, which shells out of a capsule). Only a handful of surgeons worldwide do it routinely. Post-operative pregnancy is possible but carries a meaningful risk of uterine rupture, and most centers mandate cesarean delivery. Reserved for patients with focal disease who want to preserve fertility.
Hysterectomy
The one definitive cure. Discussed in its own section below.
Hysterectomy — the Only Definitive Cure
Because adenomyosis lives inside the uterine muscle itself, removing the uterus is the only way to guarantee the disease is gone. For women who have completed childbearing and whose adenomyosis remains severe despite medical therapy, hysterectomy is often the right answer and the one that restores quality of life most reliably.
Key choices within the operation:
- Total vs supracervical. Total hysterectomy removes the uterus and cervix. Supracervical keeps the cervix. For adenomyosis, total is usually preferred because adenomyosis can extend into the cervical stroma, and retained cervical tissue occasionally continues to produce symptoms.
- Ovary preservation. The ovaries should almost always be kept if they are healthy. Ovaries produce estrogen, testosterone, and protective cardiovascular and bone effects for years after hysterectomy. Removing them before natural menopause is a separate surgical decision that should not be made casually.
- Approach. Laparoscopic, robotic, or vaginal hysterectomy is standard; open abdominal hysterectomy is reserved for very large uteri or complex anatomy. Recovery is typically two to six weeks depending on approach.
- Simultaneous endo excision. If coexisting endometriosis is suspected, schedule with a surgeon who can excise it in the same operation. A hysterectomy that leaves endo lesions behind will not eliminate all pelvic pain.
Hysterectomy for adenomyosis, done well and with ovaries preserved, has one of the highest patient-satisfaction rates of any gynecologic procedure.
Fertility and IVF Considerations
Adenomyosis reduces fertility by two mechanisms: distorted uterine contractility (abnormal peristalsis that interferes with sperm transport and embryo implantation) and a hostile junctional-zone environment. IVF outcomes in untreated adenomyosis are meaningfully lower than in matched controls.
The best-studied rescue protocol is ultra-long GnRH agonist pre-treatment: three to six months of monthly leuprolide injections before embryo transfer to shrink adenomyotic lesions, reduce uterine inflammation, and normalize the junctional zone. Studies consistently show improved implantation and live-birth rates with this approach compared with standard IVF protocols. If you have adenomyosis and are planning IVF, ask your reproductive endocrinologist about the ultra-long protocol specifically.
Focal adenomyoma may be a candidate for pre-pregnancy excision if it distorts the uterine cavity or is very large. Diffuse disease is treated medically rather than surgically before IVF.
Why an Excision Surgeon Alone Isn't Enough
Excision surgery is transformative for endometriosis. It is also, by itself, insufficient for a patient who also has adenomyosis. The mental model to carry into your consultations: excision handles the disease outside the uterus, and a separate decision — medical suppression, IUD, UAE, or hysterectomy — handles the disease inside the uterus.
A careful pre-operative workup should therefore always include a look at the uterine wall itself. If the imaging shows adenomyosis, the surgical plan should address both conditions: either by combining excision with a uterus-sparing adenomyosis therapy (Mirena placed at surgery, post-op suppression) or, if adenomyosis is severe and childbearing is complete, by combining excision with hysterectomy in a single operation. Many excision surgeons are comfortable doing both. Others partner with a second gynecologic surgeon. Either way, the plan should name the adenomyosis out loud before you go to sleep.
Pregnancy with Adenomyosis
Adenomyosis is compatible with pregnancy but carries measurable obstetric risks. Published data point to:
- Increased miscarriage risk, particularly in the first trimester, in the range of 1.5 to 2 times the baseline rate.
- Preterm labor and preterm birth, likely driven by abnormal uterine contractility.
- Placental abnormalities, including placenta previa and a modest increase in placenta accreta spectrum disorders.
- Postpartum hemorrhage, because the diseased myometrium contracts less effectively after delivery.
- Hypertensive disorders of pregnancy, including preeclampsia, at slightly elevated rates.
None of these risks should frighten patients out of pregnancy. They should prompt co-management with a maternal-fetal medicine specialist, extra attention to cervical length surveillance in the second trimester, a delivery plan at a hospital equipped for postpartum hemorrhage, and careful third-stage management (active management of the placenta with uterotonics on hand).
Choosing Medical vs Surgical Management
The decision framework every patient should be walked through:
- Age. Younger patients lean medical; adenomyosis often becomes manageable after menopause when estrogen falls.
- Fertility plans. Desired future pregnancy pushes toward Mirena-free suppression, ultra-long GnRH protocols, or adenomyomectomy rather than UAE or hysterectomy.
- Severity. Mild symptoms on a hormonal IUD may never need surgery. Flooding periods causing anemia, missed work, and emergency-room visits push the balance toward definitive treatment.
- Response to first-line therapy. Three to six months on Mirena or continuous pills is a reasonable trial. Failure of that trial is a meaningful signal that structural treatment is needed.
- Coexisting disease. Large fibroids, severe endometriosis, or pre-cancerous endometrial findings can tip the balance toward surgery.
When to Suspect Adenomyosis If You Have Endo
A practical checklist. If you already carry an endometriosis diagnosis and any of the following apply, ask your doctor specifically about adenomyosis and request dedicated imaging:
- Heavy bleeding with flooding, clots, or cycles longer than seven days — uncommon in pure endo.
- Palpably enlarged uterus on exam, or a uterus your clinician has called "bulky" or "boggy."
- Persistent pelvic pain after a thorough excision surgery — especially pain that is deep, central, and period-related rather than localized.
- MRI report noting a junctional zone over 8 mm, myometrial cysts, or asymmetric uterine walls, even if adenomyosis was not the stated focus.
- Deep dyspareunia on penetration in the absence of retrocervical lesions on exam.
- Anemia from heavy periods, especially if ferritin is low and transfusion has been discussed.
- Symptom onset or worsening in your 30s or 40s, after years of more stable endometriosis.
If one of these rings true, the next step is specific: a pelvic MRI with adenomyosis protocol, or a transvaginal ultrasound by an operator trained in MUSA criteria. Not a generic scan. Ask by name.
Key Research Papers
- Van den Bosch T, et al. Terms, definitions and measurements to describe sonographic features of myometrium and uterine masses: a consensus opinion from the Morphological Uterus Sonographic Assessment (MUSA) group. Ultrasound Obstet Gynecol. 2015.
- Chapron C, et al. Diagnosing adenomyosis: an integrated clinical and imaging approach. Hum Reprod Update. 2020. (Overlap with endometriosis.)
- Vannuccini S, Petraglia F. Recent advances in understanding and managing adenomyosis. F1000Res / Climacteric. 2019.
- ESHRE Endometriosis Guideline Group. ESHRE guideline: endometriosis. Hum Reprod Open. 2022.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on adenomyosis, its overlap with endometriosis, and its treatment:
- Adenomyosis and endometriosis overlap
- Adenomyosis MRI diagnosis and junctional zone
- MUSA ultrasound criteria for adenomyosis
- Levonorgestrel IUD for adenomyosis
- Adenomyomectomy and fertility outcomes
- Uterine artery embolization for adenomyosis
- Adenomyosis, IVF, and ultra-long GnRH protocols
- Adenomyosis and obstetric outcomes in pregnancy
Connections
- Endometriosis
- Uterine Fibroids
- Laparoscopy: Excision vs Ablation
- Diagnosis Delay and Imaging
- Hormonal Therapy Options
- Pelvic Floor Therapy and Central Sensitization
- Fertility and Endometriosis
- Diet and Lifestyle
- Endo Belly and Gut Connection
- Bladder and Bowel Endometriosis
- Infertility
- Perimenopause
- Polycystic Ovary Syndrome
- Ovarian Cysts
- Anemia
- Magnesium
- Vitamin D3
- Ultra Processed Foods