Menstrual Migraine and Hormonal Triggers
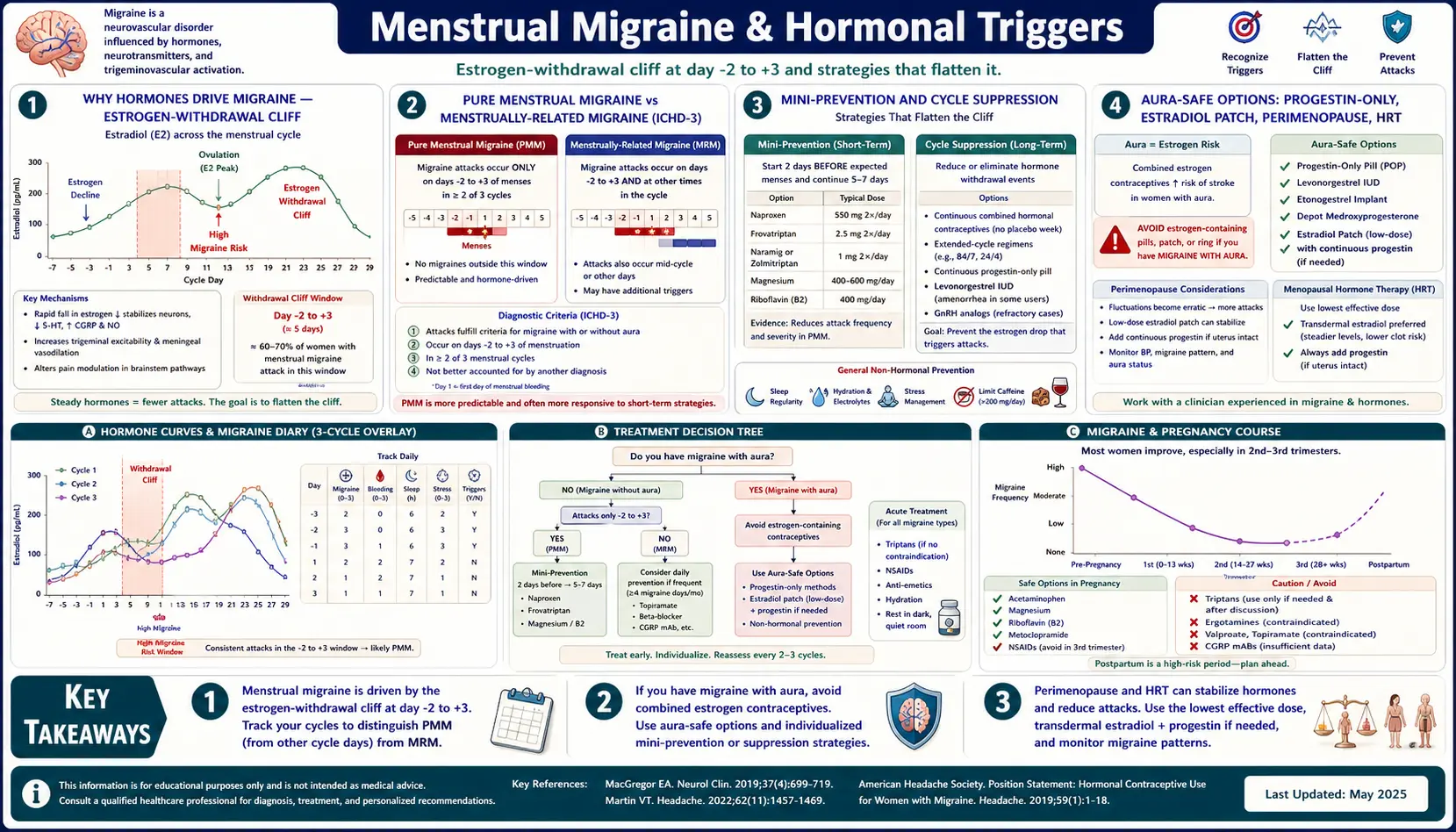
Table of Contents
- Why Hormones Drive Migraine
- The Estrogen-Withdrawal Hypothesis
- Pure Menstrual Migraine vs. Menstrually-Related Migraine
- The Headache Diary — Three Cycles to a Diagnosis
- Short-Term ("Mini") Prevention Around Your Period
- Continuous-Dose Contraceptives — Suppressing the Cycle
- The Aura Problem and Stroke Risk
- Progestin-Only Options for Aura Patients
- Transdermal Estradiol — MacGregor's Add-Back Strategy
- Perimenopause — When It Gets Worse Before It Gets Better
- HRT Decisions — Transdermal, Body-Identical, Cyclic vs. Continuous
- Surgical Menopause and Oophorectomy
- Pregnancy, Postpartum, and Breastfeeding
- Endometriosis, Adenomyosis, PCOS, Thyroid, and PMDD
- A Practical Plan You Can Bring to Your Doctor
- Key Research Papers
- Research Papers
- Connections
- Featured Videos
Why Hormones Drive Migraine
Before puberty, boys and girls get migraine at roughly the same rate. After puberty, women outnumber men about three to one. The gap opens the year estrogen begins cycling and narrows again after menopause. That single epidemiological fact tells you almost everything you need to know: migraine in women is, to a large extent, a hormonally-driven disease.
About 60% of women with migraine notice attacks clustered around their period. For roughly 10–14%, every single attack is tied to the menstrual window. Menstrual attacks tend to be longer, more severe, less responsive to the usual acute medications, and more likely to recur within 24 hours. They are not in your head, not imagined, and not a character flaw — they are a predictable neurobiological response to a hormone that is falling out from under your brain once a month.
The good news: because menstrual migraine is predictable, it is one of the few migraine subtypes where you can schedule prevention. You know the enemy's arrival date. That changes the strategy.
The Estrogen-Withdrawal Hypothesis
The dominant explanation for why periods trigger migraine is the estrogen-withdrawal hypothesis, articulated most clearly by British headache specialist Anne MacGregor. The idea is straightforward. During the late luteal phase (the week before your period), estrogen levels are relatively high. Two to three days before bleeding begins, estrogen plummets. That steep drop — not the low level itself — is what destabilizes the trigeminovascular system and lowers the migraine threshold.
Evidence supporting this model:
- Attacks cluster in the narrow window from two days before bleeding (day -2) to three days after it starts (day +3) — exactly the interval of steepest estrogen decline.
- Giving supplemental estrogen during that window blunts or prevents attacks (see the estradiol patch section).
- The pill-free week of a standard 21/7 oral contraceptive produces the same estrogen cliff and triggers "withdrawal" migraine in many users.
- Pregnancy, when estrogen rises steadily and stays high, generally improves migraine dramatically by the second trimester.
- Surgical menopause with abrupt estrogen loss often worsens migraine before it eventually settles.
The estrogen-withdrawal model does not explain every menstrual attack — prostaglandin surges from the endometrium, serotonin fluctuations, and CGRP sensitivity all play roles — but it explains enough to drive real treatment decisions.
Pure Menstrual Migraine vs. Menstrually-Related Migraine
The International Classification of Headache Disorders, third edition (ICHD-3), splits menstrual migraine into two categories that sound similar but matter clinically.
Pure menstrual migraine without aura (PMM) — ICHD-3 code A1.1.1. Attacks occur only during the perimenstrual window (days -2 to +3) in at least two of every three cycles, and at no other time of the month. Roughly 10–14% of women with migraine meet this definition. PMM is the cleanest target: if your brain only fires during that five-day window, you only need prevention for five days a month.
Menstrually-related migraine without aura (MRM) — ICHD-3 code A1.1.2. Attacks occur reliably during the perimenstrual window and at other times throughout the cycle. This is far more common — about 35–50% of female migraine patients. Mini-prevention can still help, but you usually need a background preventive too.
The distinction matters because PMM often responds beautifully to short-term ("mini") prevention alone, while MRM usually needs a layered approach: daily preventive plus targeted perimenstrual coverage.
Both definitions specifically exclude aura. Menstrual attacks with aura exist but are less common and sit in a different risk category because of the stroke and contraceptive implications discussed below.
The Headache Diary — Three Cycles to a Diagnosis
Before your doctor can prescribe mini-prevention, you both need to know whether your migraines are actually menstrually linked. Memory is unreliable. A three-month headache diary is.
Track, every day:
- Cycle day (day 1 = first day of full bleeding, not spotting).
- Headache: yes / no, and if yes the severity (0–10) and duration.
- Aura: yes / no, and type (visual, sensory, speech).
- Medications taken and whether they worked.
- Other triggers — sleep loss, skipped meals, red wine, weather fronts, stress.
Free options include the Migraine Buddy app, the N1-Headache app, or a plain paper calendar. After three complete cycles, overlay your headache days on your cycle days. If the cluster is obviously in the day -2 to +3 window, you have your answer. Bring the diary to your appointment; it will save an hour of discussion.
Short-Term ("Mini") Prevention Around Your Period
Mini-prevention means starting a medication two days before your expected period and continuing for five to seven days through the high-risk window. You take nothing the rest of the month. For women with regular cycles and clear PMM, this approach prevents roughly 50–60% of perimenstrual attacks with minimal cumulative drug exposure.
The evidence-based regimens:
- Frovatriptan 2.5 mg twice daily for 6 days, starting day -2. This is the best-studied option — Silberstein's 2004 trial established the protocol and it remains first-line. Frovatriptan's 26-hour half-life (the longest of any triptan) is what makes it suitable for scheduled dosing.
- Naratriptan 1 mg twice daily for 5 days, starting day -2. Similar concept, shorter half-life, slightly less robust evidence but often cheaper and available as generic.
- Naproxen 550 mg twice daily for 6–7 days, starting day -2. Non-hormonal, inexpensive, and additionally useful if you also have menstrual cramps or heavy bleeding. Pair with a proton pump inhibitor if you have any reflux or gastritis history.
- Zolmitriptan 2.5 mg two or three times daily for 5 days is an alternative if frovatriptan is unavailable or not covered.
Mini-prevention works only if your cycles are reasonably predictable (within two or three days). If your cycles are irregular, you will miss the window half the time. In that case, cycle suppression (see below) is usually a better approach.
A practical tactic: keep a rescue acute medication (a second triptan, a gepant like ubrogepant or rimegepant, or a ditan like lasmiditan) on hand for breakthrough attacks that punch through mini-prevention. See the triptans and gepants article for acute-treatment choices.
Continuous-Dose Contraceptives — Suppressing the Cycle
If the estrogen-withdrawal hypothesis is correct, the cleanest solution is to stop the withdrawal from happening. Continuous-dose combined oral contraceptives (COCs) do exactly that: you skip the placebo/pill-free week and take active pills every day, eliminating the monthly estrogen drop.
Options that work:
- Continuous monophasic COC — any standard low-dose (20–30 mcg ethinyl estradiol) monophasic pill, taken 365 days a year, skipping the placebo week. Brands like Seasonique, Amethyst, and Lybrel are pre-packaged for this pattern, but any monophasic generic can be used continuously at a fraction of the cost.
- Extended-cycle COC — 84 active days then a 7-day break, or 84 active plus 7 days of low-dose estrogen. Fewer withdrawal windows per year (four instead of thirteen) but does not eliminate them.
- Vaginal ring (etonogestrel/ethinyl estradiol) used continuously, changing the ring every 3–4 weeks without a ring-free week.
Calhoun's 2012 review of hormonal contraception in migraine made the key point: it is the stable estrogen level that matters, not the absolute dose. Continuous dosing prevents the cliff; cyclic dosing recreates it every month.
Critical caveat: combined (estrogen-containing) contraceptives are only appropriate in women with migraine without aura. If you have aura of any kind, read the next section before considering any estrogen-containing product.
The Aura Problem and Stroke Risk
Migraine with aura roughly doubles the baseline ischemic stroke risk in young women. Combined oral contraceptives, by raising estrogen-mediated clotting factors, add their own modest stroke risk. When you stack them, the combined risk rises enough that the World Health Organization, the American College of Obstetricians and Gynecologists, and the CDC all classify combined hormonal contraceptives as contraindicated (category 4) in women with migraine with aura. Smoking on top of this is the tripwire that turns a small absolute risk into a meaningful one.
What this means in practice:
- If you have any aura — visual zigzags, scotomas, sensory tingling, speech disturbance — combined pills, combined patches, and combined vaginal rings are off the table for migraine prevention.
- A single lifetime aura episode counts. Mention it to your prescriber.
- Progestin-only methods and transdermal-estradiol add-back (different from a contraceptive patch) are the remaining hormonal options.
See the aura article for how to identify whether what you experience meets the diagnostic definition.
Progestin-Only Options for Aura Patients
Progestin-only contraception carries no measurable added stroke risk and is safe in migraine with aura. Several forms exist, and any of them — when they successfully suppress ovulation and therefore suppress the cyclic estrogen drop — can reduce menstrual migraine frequency.
- Desogestrel 75 mcg daily (Cerazette, Slynd) — a "newer-generation" progestin-only pill that reliably suppresses ovulation in most users. Often the first choice for aura patients who want a daily oral option.
- Levonorgestrel IUD (Mirena, Liletta, Kyleena) — five to eight years of protection. Does not suppress ovulation in everyone but drastically reduces menstrual bleeding and prostaglandin load, which helps many women.
- Depot medroxyprogesterone (Depo-Provera) — injection every 12 weeks. Reliably suppresses ovulation. Watch for bone-density effects with long-term use and weight gain in susceptible individuals.
- Etonogestrel implant (Nexplanon) — three years, small rod in the upper arm. Suppresses ovulation in most cycles.
Progestin-only methods do not have the same clean dose-response relationship with migraine that combined pills do, so expect a trial-and-error period. Some women get dramatic relief; a smaller fraction get worse (usually because of irregular spotting that amounts to continuous mini-withdrawal events). Give any method three to four months before judging it.
Transdermal Estradiol — MacGregor's Add-Back Strategy
For women with regular cycles and clear PMM who either cannot take combined contraceptives (because of aura) or who simply want to preserve their cycle, MacGregor developed an elegant alternative: leave the natural cycle alone, but plug the estrogen cliff with a short course of transdermal estradiol around the period.
The classic regimen:
- Apply a 100 mcg estradiol patch two days before expected period onset (day -2).
- Leave it in place for seven days, replacing at the standard patch interval (twice-weekly patches require a change mid-course).
- Remove on day +5. Resume normal cycle.
Lower doses (50 or 75 mcg) have been trialed and appear less effective; 100 mcg is the minimum that reliably blunts the estrogen drop. Gel formulations (1.5 mg estradiol gel daily during the window) are an alternative when patches irritate the skin or detach. This approach is not contraceptive — you still need a separate non-estrogen birth-control method if pregnancy is a concern — and it is not universally covered by U.S. insurance. But it is the most physiologically targeted hormonal intervention we have for PMM.
Perimenopause — When It Gets Worse Before It Gets Better
Many women assume menopause means migraine relief. Eventually, yes. But the four to ten years of perimenopause that precede the final period are often the worst migraine years of a woman's life, and nobody warns patients about it.
The physiology: during perimenopause, ovarian follicles become erratic. Estrogen does not gradually decline — it swings wildly, sometimes higher than normal, sometimes crashing. Every crash is an estrogen-withdrawal event, and crashes can happen multiple times per cycle rather than once. Progesterone production also falls unpredictably, which amplifies the swings. Cycles shorten, lengthen, skip entirely, then return with a vengeance.
Patterns patients commonly report:
- Migraines that used to be monthly become weekly or near-daily.
- Previously reliable mini-prevention stops working because cycles are no longer predictable.
- Hot flashes, night sweats, and sleep disruption independently lower the migraine threshold.
- A sudden increase in migraine frequency in a woman in her forties is often the first clue perimenopause has started, months or years before any cycle irregularity.
Average age at menopause in the U.S. is 51. If your migraines are getting worse in your early-to-mid forties, perimenopause is almost certainly part of the picture. The workup is clinical — FSH levels are unreliable during perimenopause because they fluctuate as much as estrogen does — but a pattern of worsening migraine plus hot flashes, sleep disruption, or cycle change is enough to act on.
HRT Decisions — Transdermal, Body-Identical, Cyclic vs. Continuous
Hormone replacement therapy (HRT, now often called MHT — menopausal hormone therapy) can stabilize perimenopausal and postmenopausal migraine, but the form of HRT matters enormously for headache outcomes.
- Transdermal estradiol is strongly preferred over oral estrogen. Oral estrogen passes through the liver, produces erratic blood levels, and in many women triggers more migraine, not less. Transdermal patches or gels deliver steady estradiol directly into the bloodstream and avoid the first-pass effect. If a doctor offers you an oral estrogen pill for menopause and you have migraine, ask specifically for the patch or gel.
- Body-identical (micronized) progesterone — sold as Prometrium in the U.S. and Utrogestan elsewhere — is generally better tolerated than synthetic progestins (medroxyprogesterone, norethindrone) for migraine patients. It also aids sleep, a secondary migraine benefit.
- Continuous combined HRT (daily estrogen + daily progesterone) is usually preferable to cyclic HRT for migraine. Cyclic regimens deliberately recreate a monthly withdrawal bleed, which can re-trigger menstrual migraine in women who had finally escaped it.
- Dose matters. Start low (25–37.5 mcg patch) and titrate up only as needed. Higher doses reduce flushing and sleep disruption but can reignite migraine.
- Aura caveat remains relevant. Transdermal estradiol in physiologic (menopausal) doses does not carry the stroke risk of a combined oral contraceptive — the dose is roughly a quarter to a sixth — and is generally considered acceptable in women with migraine with aura, though guidance varies. Discuss with a prescriber who knows the difference.
Surgical Menopause and Oophorectomy
Removal of both ovaries — bilateral oophorectomy — is a hormonal catastrophe for most migraine patients. Overnight, estrogen crashes from premenopausal levels to zero, with no taper. Migraine typically worsens dramatically in the weeks and months after surgery, and may not resettle for a year or more.
If you have migraine and are considering oophorectomy (for endometriosis, ovarian masses, cancer risk, or another indication), bring the topic up explicitly with your surgeon and headache specialist before the operation. Starting transdermal estradiol on the day of surgery and continuing it at least until the natural age of menopause (~51) blunts the crash and is usually appropriate even in women who would otherwise be ambivalent about HRT. Hysterectomy without oophorectomy does not cause this problem because the ovaries continue to function.
Pregnancy, Postpartum, and Breastfeeding
Pregnancy is, for most migraine patients, a reprieve. The data:
- First trimester: mixed. Roughly a third get better, a third unchanged, a third worse. Nausea and sleep disruption from pregnancy itself can confuse the picture.
- Second trimester: 50–80% of women experience dramatic improvement or complete remission. Estrogen is high, steady, and rising. This is the honeymoon.
- Third trimester: improvement usually continues, though late-pregnancy sleep disruption, reflux, and back pain can trigger attacks unrelated to hormones.
- Postpartum: estrogen plummets within 24–72 hours of delivery. Roughly 30–40% of women have a migraine within the first week postpartum, and the first four to six weeks are a high-risk window. Sleep deprivation on top of the estrogen crash is a double hit.
- Breastfeeding generally extends the reprieve because lactation suppresses ovulation and keeps estrogen stable at a lower but flat level. Migraines often return when breastfeeding ends or menses resumes.
Acute medications during pregnancy: acetaminophen is first-line. Metoclopramide (antiemetic with migraine-aborting properties) is generally considered safe. NSAIDs should be avoided after 20 weeks (kidney and ductus-arteriosus concerns) and especially in the third trimester. Triptans — particularly sumatriptan, which has the largest registry data — are not formally approved but are widely used when benefits outweigh risks; large observational cohorts have not shown clear harm. Ergotamine and DHE are contraindicated. Gepants and CGRP monoclonal antibodies lack pregnancy data and are generally stopped before conception.
Breastfeeding-safe acute options: acetaminophen, ibuprofen (preferred NSAID while nursing because of its short half-life and low milk transfer), sumatriptan (minimal transfer, considered compatible), eletriptan (even lower transfer). Avoid aspirin while nursing because of Reye-syndrome concerns. The LactMed database (free, NIH) is the authoritative source when a specific drug comes up.
Endometriosis, Adenomyosis, PCOS, Thyroid, and PMDD
Menstrual migraine rarely travels alone. The conditions that cluster with it share hormonal machinery, and treating one often helps the others.
- Endometriosis — women with endometriosis have roughly twice the rate of migraine. The same prostaglandin surge that drives endometriosis pain contributes to menstrual headache. Cycle-suppressing hormonal therapy can address both. See the endometriosis overview and the hormonal therapy options deep-dive.
- Adenomyosis — endometrial tissue embedded in the uterine muscle. Causes heavy, painful periods with a high prostaglandin load. Often goes undiagnosed for years. MRI or transvaginal ultrasound makes the diagnosis. Treatment overlaps with endometriosis and almost always helps menstrual migraine.
- Polycystic ovary syndrome (PCOS) — the irregular cycles of PCOS make mini-prevention nearly impossible because you cannot predict the window. Restoring cycle regularity with a combined pill (in the absence of aura) or cycle suppression often unlocks other migraine strategies.
- Thyroid disease — hypothyroidism, particularly autoimmune (Hashimoto's), is overrepresented in chronic migraine cohorts. TSH, free T4, and thyroid antibodies are a reasonable screen in anyone with a new escalation of migraine frequency. See the Hashimoto's overview and the pregnancy and Hashimoto's deep-dive, which is also relevant to anyone navigating postpartum migraine.
- Premenstrual dysphoric disorder (PMDD) — severe mood, anxiety, and irritability in the luteal phase. The neurobiology overlaps with menstrual migraine (altered serotonin and GABA sensitivity to progesterone metabolites). SSRIs used luteal-phase-only (days 14–28) treat PMDD and may reduce menstrual migraine severity as a bonus.
A Practical Plan You Can Bring to Your Doctor
Print this. Bring it in. Ask which steps fit your situation.
- Three-cycle headache diary. Confirm the pattern and whether you have aura.
- Basic labs: CBC, ferritin (heavy periods can make you iron-deficient, which lowers the migraine threshold), TSH + free T4, vitamin D, and consider thyroid antibodies.
- Magnesium glycinate 400–600 mg daily, riboflavin 400 mg daily — both have randomized-trial evidence in general migraine prevention and a low side-effect profile. See the supplements article.
- If cycles are regular and you have PMM: trial mini-prevention with frovatriptan 2.5 mg BID x 6 days starting day -2, or naproxen 550 mg BID as a non-hormonal alternative.
- If cycles are irregular or mini-prevention fails, and you have no aura: discuss continuous-dose combined contraception.
- If you have aura: progestin-only options (desogestrel, levonorgestrel IUD) or MacGregor's transdermal estradiol add-back around menses.
- If you are perimenopausal: consider transdermal HRT (body-identical progesterone, continuous rather than cyclic), and keep a CGRP preventive on the table for background control.
- If attacks remain frequent despite the above: add a daily background preventive — a CGRP monoclonal antibody, atogepant, topiramate, or a beta-blocker. See the CGRP and preventives article.
- Monitor medication overuse. Triptans more than 10 days a month, or combination analgesics more than 15 days a month, convert episodic migraine into chronic migraine. See the medication-overuse article.
Menstrual migraine is not a character of your cycle you are stuck with. It is a specific, well-characterized neurobiological event with specific, well-characterized interventions. Most women who work through this list in order find a combination that cuts their attack frequency at least in half, often more.
Key Research Papers
- MacGregor EA, et al. Incidence of migraine relative to menstrual cycle phases of rising and falling estrogen. Neurology. 2006.
- Silberstein SD, et al. Frovatriptan for the short-term prevention of menstrual migraine. Neurology. 2004.
- Calhoun AH. Hormonal contraceptives and migraine with aura: is there still a risk? Headache. 2012.
- Sacco S, et al. Hormonal contraceptives and risk of ischemic stroke in women with migraine: a consensus statement. Curr Pain Headache Rep. 2017.
Research Papers
For further reading, the following PubMed topic searches return current peer-reviewed work on menstrual migraine and the hormonal interventions discussed above:
- Menstrual migraine and estrogen withdrawal
- Frovatriptan short-term prevention
- Naproxen for menstrual migraine
- Continuous-dose oral contraceptives and migraine
- Migraine with aura, stroke risk, and oral contraceptives
- Transdermal estradiol for menstrual migraine
- Perimenopause and migraine
- Hormone replacement therapy and migraine
- Pregnancy, trimester effects, and migraine
- Postpartum and breastfeeding-safe migraine treatment
- Endometriosis and migraine comorbidity
- PMDD, serotonin, and menstrual migraine
Connections
- Endometriosis
- Endometriosis — Hormonal Therapy Options
- Chronic Migraine and Medication Overuse
- Magnesium, Riboflavin, and Supplements
- CGRP Inhibitors and Preventives
- Triptans and Gepants — Acute Treatment
- Aura and Visual Disturbances
- Hashimoto's Thyroiditis
- Pregnancy and Hashimoto's
- Migraine Overview
- Perimenopause
- Polycystic Ovary Syndrome
- Diet Triggers and Elimination Protocol
- Vestibular Migraine
- Menopause and HRT
- Migraine in Pregnancy and Breastfeeding
- Stroke
- Perimenopause Symptom Tracker and Hormonal Testing