Vitamin C and Immune Defense
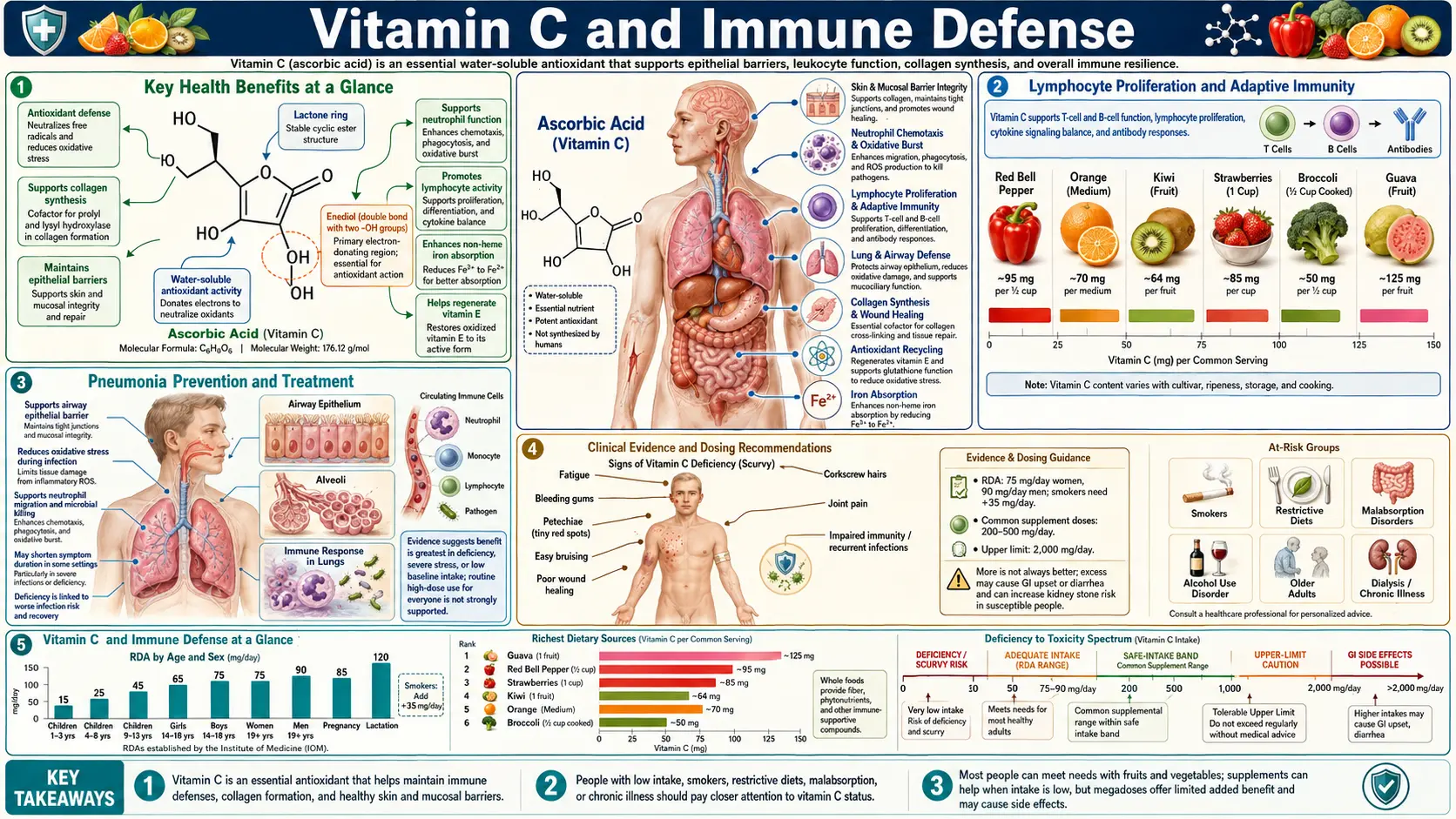
Vitamin C (ascorbic acid) is one of the most extensively studied nutrients in the context of immune function, and it occupies an almost unique position: immune cells concentrate ascorbate at levels 10 to 100 times higher than plasma, reflecting how much they depend on it. Both innate defenses (neutrophils, macrophages, NK cells, antimicrobial peptides, type I interferons) and adaptive responses (T and B lymphocytes, antibody production, epigenetic TET-enzyme regulation) require adequate vitamin C status to perform optimally. This article reviews the cellular mechanisms, the high-quality Cochrane evidence on the common cold, the pneumonia-prevention trials, the mixed picture from IV-vitamin-C sepsis research, and the evidence-based dosing for healthy and at-risk populations.
Table of Contents
- Key Health Benefits at a Glance
- Neutrophil Function
- Macrophage Activation
- Natural Killer Cell Enhancement
- Lymphocyte Proliferation and Adaptive Immunity
- Cathelicidin, Antimicrobial Peptides, and Interferons
- Common Cold: Meta-Analysis Results
- Pneumonia Prevention and Treatment
- Sepsis and Critical Care: IV Vitamin C Protocols
- High-Dose Vitamin C Research
- The Linus Pauling Legacy
- Clinical Evidence and Dosing Recommendations
- Research Papers and References
- Connections
- Featured Videos
Key Health Benefits at a Glance
Before the detailed mechanisms, the following is a high-level summary of the evidence-backed immune benefits of adequate vitamin C status. Each bullet is developed below, and supporting studies are linked in the Research Papers section.
- Concentrates in immune cells – Neutrophils, lymphocytes, and monocytes accumulate vitamin C at 10–100 times plasma levels, highlighting its central role in immune defense.
- Boosts neutrophil function – Ascorbate enhances chemotaxis, phagocytosis, oxidative burst, and apoptotic clearance, reducing collateral tissue damage at infection sites.
- Supports NK cell cytotoxicity – Supplementation has been shown to enhance NK cell killing of virus-infected and transformed cells in clinical studies.
- Supports lymphocyte biology and epigenetics – Vitamin C is a cofactor for the TET DNA-demethylation enzymes, shaping T-cell differentiation and memory formation.
- Shortens common colds – The Cochrane meta-analysis (Hemilä & Chalker, 2013) documented 8% shorter colds in adults and 14% shorter in children with regular supplementation; cold incidence is cut roughly in half in people under severe physical stress.
- Prevents pneumonia in stressed populations – Three controlled trials in military and institutional populations showed 80%+ reductions in pneumonia incidence with 200 mg–2 g/day supplementation.
- Works with vitamin D on antimicrobial peptides – Ascorbate synergizes with vitamin D to upregulate cathelicidin (LL-37), a peptide that directly kills bacteria, fungi, and enveloped viruses.
- Enhances type I interferon production – Vitamin C supports IFN-alpha and IFN-beta output, amplifying the antiviral state in neighboring cells.
- Restores critically low levels in sepsis – Septic patients exhibit scorbutic plasma levels; IV repletion is an active research area even as trial results remain mixed.
- Supports mucosal barrier immunity – Ascorbate supports beta-defensin production by respiratory and gastrointestinal epithelium, reinforcing the first line of defense.
Neutrophil Function
Neutrophils are the first responders of the innate immune system, and vitamin C plays an essential role in multiple aspects of their function.
- Chemotaxis – Vitamin C enhances the directed migration of neutrophils toward sites of infection. Studies demonstrate that ascorbate-replete neutrophils exhibit significantly improved chemotactic movement compared to vitamin C-deficient cells. The nutrient supports cytoskeletal reorganization required for cell motility through its effects on microtubule assembly and actin polymerization.
- Phagocytosis – The engulfment and internalization of pathogens by neutrophils is augmented in the presence of adequate vitamin C. Ascorbate enhances recognition of opsonized bacteria and facilitates the membrane remodeling necessary for phagosome formation. Vitamin C-deficient individuals show measurably reduced phagocytic capacity.
- Oxidative Burst – During the respiratory burst, neutrophils generate reactive oxygen species (ROS) to kill ingested microorganisms. Vitamin C supports the NADPH oxidase enzyme complex responsible for superoxide production. Simultaneously, ascorbate protects the neutrophil itself from oxidative self-damage through its antioxidant properties, thereby extending neutrophil lifespan and functional capacity.
- Neutrophil Extracellular Traps (NETs) – Emerging research suggests that vitamin C may modulate the formation of NETs, web-like structures of DNA and antimicrobial proteins that neutrophils release to trap and kill extracellular pathogens.
- Apoptosis and Clearance – Vitamin C promotes timely apoptosis of spent neutrophils and enhances their clearance by macrophages (efferocytosis), a process critical for resolving inflammation and preventing tissue damage at infection sites.
Macrophage Activation
Macrophages serve as both phagocytic defenders and orchestrators of immune responses. Vitamin C influences macrophage function through several mechanisms.
- Enhanced Phagocytic Activity – Ascorbate-loaded macrophages demonstrate greater capacity to engulf and destroy bacteria, fungi, and cellular debris. Vitamin C supports the generation of phagolysosomal killing mechanisms.
- Cytokine Production – Vitamin C modulates macrophage cytokine output, promoting the release of pro-inflammatory mediators during active infection while supporting the transition to anti-inflammatory signaling during the resolution phase. This dual role helps prevent excessive inflammation while maintaining pathogen clearance.
- M1/M2 Polarization – Research indicates that vitamin C may influence macrophage polarization, supporting the pro-inflammatory M1 phenotype during acute infection and facilitating the shift to the tissue-repair M2 phenotype during recovery.
Natural Killer Cell Enhancement
Natural killer (NK) cells provide rapid cytotoxic responses against virus-infected cells and tumor cells. Vitamin C supports NK cell function in several ways.
- Cytotoxic Activity – Supplementation with vitamin C has been shown to enhance NK cell cytolytic activity by 20 to 50% in some clinical studies. Ascorbate supports the production and release of perforin and granzymes, the primary effector molecules of NK cell killing.
- NK Cell Proliferation – Vitamin C promotes the expansion of NK cell populations during immune challenge, ensuring adequate numbers of these critical effector cells are available during viral infections.
- Recognition of Targets – Adequate vitamin C status supports the expression of activating receptors on the NK cell surface, improving the ability to detect and respond to abnormal cells.
Lymphocyte Proliferation and Adaptive Immunity
The adaptive immune system, comprising T cells and B cells, also depends on vitamin C for optimal function.
- T Cell Proliferation – Vitamin C is required for robust clonal expansion of T lymphocytes following antigen recognition. In vitro studies show that ascorbate enhances mitogen-stimulated T cell proliferation by supporting the metabolic demands of rapidly dividing cells.
- T Cell Differentiation – Vitamin C influences epigenetic regulation through its role as a cofactor for ten-eleven translocation (TET) enzymes, which catalyze DNA demethylation. This activity is important for proper T cell maturation and differentiation into effector and memory subsets.
- B Cell Function and Antibody Production – Adequate ascorbate status supports B cell maturation and immunoglobulin synthesis. Vitamin C-deficient animals produce lower antibody titers in response to immunization.
Cathelicidin, Antimicrobial Peptides, and Interferons
- Cathelicidin (LL-37) – Vitamin C works synergistically with vitamin D to upregulate the expression of cathelicidin, a potent antimicrobial peptide produced by neutrophils, macrophages, and epithelial cells. LL-37 directly kills bacteria, fungi, and enveloped viruses by disrupting their membranes, and also serves as a chemoattractant for immune cells.
- Defensins – Ascorbate supports the production of beta-defensins by epithelial barrier cells, contributing to mucosal immunity in the respiratory and gastrointestinal tracts.
- Interferon Production – Vitamin C enhances the synthesis of type I interferons (IFN-alpha and IFN-beta), which are critical for antiviral defense. Interferons induce an antiviral state in neighboring cells by upregulating hundreds of interferon-stimulated genes (ISGs) that inhibit viral replication at multiple stages. Cell culture studies demonstrate increased interferon output from ascorbate-treated leukocytes exposed to viral stimuli.
Common Cold: Meta-Analysis Results
The relationship between vitamin C and the common cold has been studied in numerous randomized controlled trials. The Cochrane Collaboration meta-analysis by Hemilä and Chalker (2013) provides the most comprehensive summary.
- Incidence – Regular supplementation with 200 mg or more of vitamin C daily did not reduce the incidence of colds in the general population. However, in individuals under heavy physical stress (marathon runners, soldiers in subarctic environments, skiers), regular vitamin C supplementation reduced cold incidence by approximately 50%.
- Duration – Consistent supplementation reduced cold duration by 8% in adults and 14% in children. This translates to roughly half a day shorter illness for adults and a full day shorter for children.
- Severity – Several trials reported reduced symptom severity scores in the supplemented groups, with less time confined to bed or absent from work or school.
- Therapeutic Use – Taking vitamin C after cold symptoms begin has shown inconsistent results, though some trials using doses of 6 to 8 grams on the first day of symptoms have demonstrated benefit.
Pneumonia Prevention and Treatment
- Prevention – Three controlled trials in military recruits and other populations have shown that vitamin C supplementation (200 mg to 2 g daily) reduced pneumonia incidence by 80% or more in the supplemented groups. These findings are particularly relevant for populations under physical stress or living in close quarters.
- Treatment – Observational data and limited trial evidence suggest that vitamin C supplementation in hospitalized pneumonia patients may reduce duration of illness and improve outcomes, particularly when baseline vitamin C levels are low.
- Elderly Populations – Older adults in institutional settings, who frequently have suboptimal vitamin C status, appear to derive the greatest benefit from supplementation in pneumonia prevention.
Sepsis and Critical Care: IV Vitamin C Protocols
The use of intravenous vitamin C in critically ill patients has generated significant clinical interest.
- Rationale – Critically ill patients, especially those with sepsis, exhibit dramatically depleted plasma vitamin C levels, often reaching scorbutic concentrations below 11 micromol/L. This depletion correlates with organ dysfunction and mortality. Intravenous administration bypasses the saturable intestinal absorption that limits oral dosing.
- Marik Protocol – Dr. Paul Marik’s combination protocol using IV vitamin C (1.5 g every 6 hours), hydrocortisone, and thiamine generated initial excitement with a retrospective study showing dramatic mortality reduction. Subsequent randomized controlled trials (VITAMINS, VICTAS) produced mixed results.
- LOVIT Trial – The large multicenter LOVIT trial did not demonstrate benefit for high-dose IV vitamin C (50 mg/kg every 6 hours) in sepsis, and suggested possible harm. This study has led to more cautious clinical recommendations.
- Ongoing Research – Despite mixed trial results, research continues into optimal dosing strategies, patient selection criteria, and the timing of IV vitamin C administration relative to the onset of sepsis.
High-Dose Vitamin C Research
- Pharmacokinetics – Oral vitamin C absorption is saturable, with bioavailability dropping from nearly 100% at low doses to below 50% at doses exceeding 1 gram. Intravenous administration can achieve plasma concentrations 30 to 70 times higher than the maximum achievable orally, as documented by Padayatty and colleagues (Ann Intern Med 2004).
- Pro-Oxidant Activity – At the very high plasma concentrations achievable only through IV administration, vitamin C can generate hydrogen peroxide in the extracellular space. This pro-oxidant effect is selectively toxic to certain cancer cells while normal cells are protected by catalase activity. This mechanism underlies research into high-dose IV vitamin C as an adjunctive cancer therapy.
- Safety Considerations – High-dose IV vitamin C is generally well tolerated but is contraindicated in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency due to the risk of hemolytic anemia. It may also interfere with certain glucometer readings and should be used cautiously in patients with renal insufficiency due to the risk of oxalate nephropathy.
The Linus Pauling Legacy
Nobel laureate Linus Pauling brought vitamin C into mainstream consciousness with his 1970 book Vitamin C and the Common Cold. Pauling advocated for gram-dose supplementation, far exceeding the RDA, based on evolutionary arguments (most mammals synthesize their own vitamin C) and emerging clinical data. While some of Pauling’s more expansive claims were not fully validated by subsequent research, his advocacy catalyzed decades of productive scientific investigation. Modern immunology has largely confirmed that vitamin C plays a far more significant role in immune defense than was appreciated before Pauling’s work, even if the optimal supplemental doses remain debated.
Clinical Evidence and Dosing Recommendations
- RDA vs. Optimal Intake – The current RDA for vitamin C (75 mg for women, 90 mg for men) is designed to prevent scurvy with a margin of safety. However, immune cell saturation requires higher intakes. Research by Levine and colleagues (PNAS 1996) suggests that 200 mg daily is sufficient to saturate plasma and circulating immune cells in healthy individuals.
- During Infection – Metabolic demand for vitamin C increases dramatically during active infection. Some experts recommend 1 to 2 grams daily during acute illness, divided into multiple doses to maximize absorption.
- At-Risk Populations – Smokers require an additional 35 mg daily due to increased oxidative stress. The elderly, individuals under physical stress, and those with chronic illness may also benefit from higher intakes.
- Upper Limit – The tolerable upper intake level is set at 2 grams daily for adults, primarily based on the risk of gastrointestinal discomfort and diarrhea at higher doses. Serious adverse effects from oral vitamin C are rare.
- Food Sources – The richest dietary sources include bell peppers, citrus fruits, kiwi, strawberries, broccoli, Brussels sprouts, and tomatoes. Five servings of fruits and vegetables daily typically provide 200 mg or more of vitamin C.
This content is provided for informational purposes only and does not constitute medical advice. Consult a qualified healthcare provider before beginning high-dose vitamin C supplementation, particularly with a history of kidney stones, G6PD deficiency, or chronic kidney disease.
Research Papers and References
The following are landmark and frequently cited research papers underpinning the claims on this page. Links resolve to the publisher DOI or PubMed record.
Pharmacokinetics
- Levine M, Conry-Cantilena C, Wang Y, et al. Vitamin C pharmacokinetics in healthy volunteers: evidence for a recommended dietary allowance. PNAS. 1996;93(8):3704-3709.
- Padayatty SJ, Sun H, Wang Y, et al. Vitamin C pharmacokinetics: implications for oral and intravenous use. Annals of Internal Medicine. 2004;140(7):533-537.
Immune Function — Mechanism and Review
- Carr AC, Maggini S. Vitamin C and immune function. Nutrients. 2017;9(11):1211.
- PubMed — Vitamin C and neutrophil function (topic search)
- PubMed — Vitamin C and NK cell cytotoxicity (topic search)
- PubMed — Vitamin C, TET enzymes, and T-cell epigenetics (topic search)
Common Cold
- Hemilä H, Chalker E. Vitamin C for preventing and treating the common cold. Cochrane Database of Systematic Reviews. 2013;(1):CD000980.
- PubMed — Vitamin C and common cold duration meta-analyses (topic search)
Pneumonia and Respiratory Infection
- PubMed — Vitamin C and pneumonia prevention trials (topic search)
- PubMed — Vitamin C and respiratory infection in the elderly (topic search)
Sepsis and Critical Care
- PubMed — HAT protocol (hydrocortisone, ascorbic acid, thiamine) and sepsis (topic search)
- PubMed — LOVIT trial and high-dose IV vitamin C in sepsis (topic search)
- PubMed — VITAMINS trial and vitamin C in septic shock (topic search)
Antimicrobial Peptides and Interferons
- PubMed — Vitamin C, cathelicidin, and antimicrobial peptides (topic search)
- PubMed — Ascorbate and type I interferon antiviral defense (topic search)
External Authoritative Resources
- NIH Office of Dietary Supplements — Vitamin C Fact Sheet for Health Professionals
- Linus Pauling Institute — Micronutrient Information Center: Vitamin C
- Harvard T.H. Chan School of Public Health — The Nutrition Source: Vitamin C
- PubMed — Vitamin C and immune function systematic reviews (topic search)
Connections
- Vitamin C
- Vitamin C and Collagen
- Vitamin C and Iron Absorption
- Zinc
- Vitamin D3
- Vitamin A
- Immune Boosting
- Elderberry
- Echinacea
- Selenium
- Cold and Flu Treatments
- Pneumonia
- Sepsis
- Cancer
- Vitamin E and Immune Function