Vitamin C and Iron Absorption
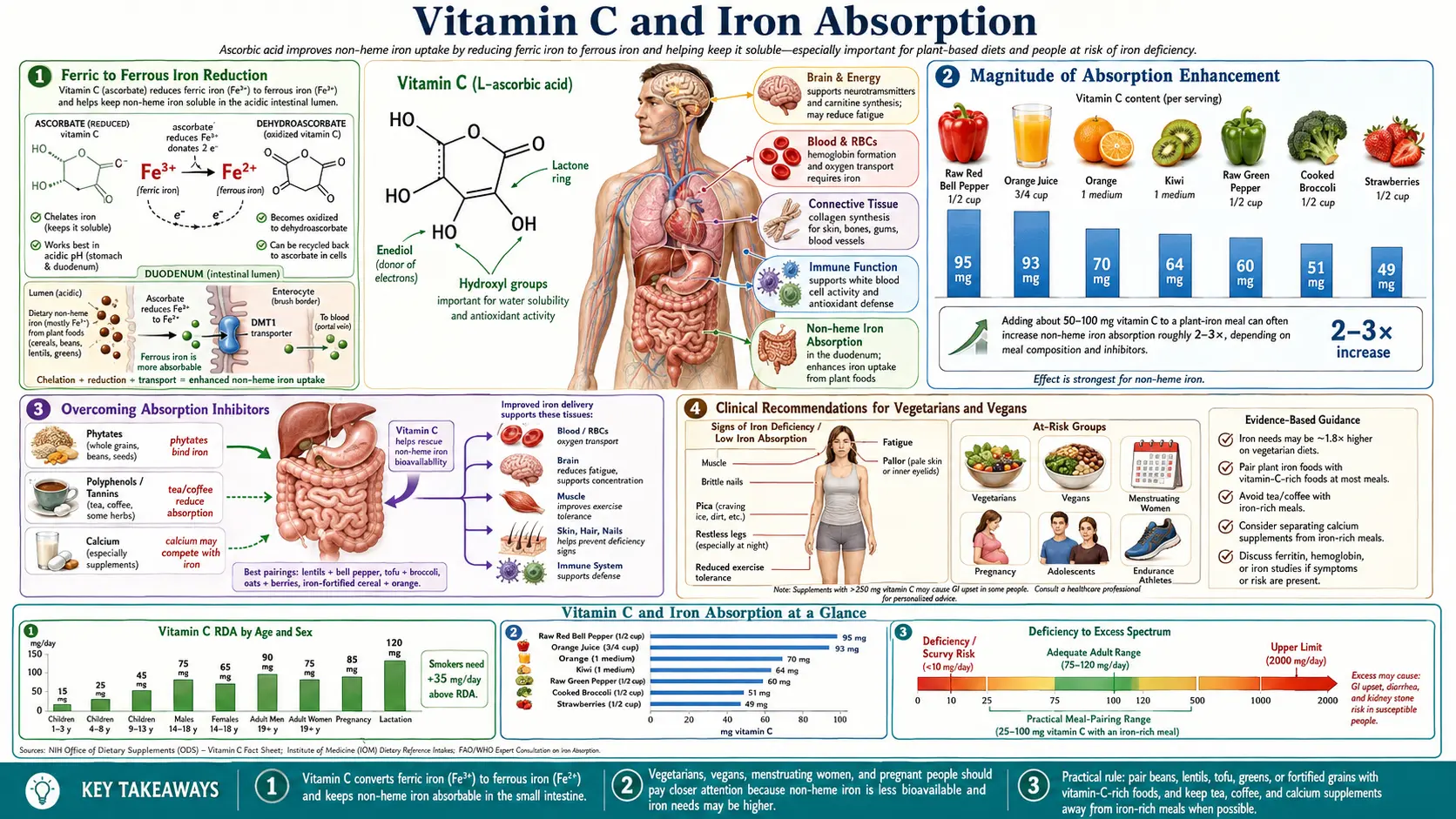
Iron deficiency is the most prevalent nutritional deficiency worldwide, affecting an estimated two billion people and causing significant morbidity through iron deficiency anemia (IDA). Vitamin C (ascorbic acid) is the most potent dietary enhancer of non-heme iron absorption, capable of increasing iron uptake from plant foods by three- to sixfold. Understanding this relationship is critical for preventing and treating iron deficiency, particularly in populations relying primarily on plant-based diets.
Table of Contents
- Ferric to Ferrous Iron Reduction
- Chelation Mechanism in the Gut
- DMT1 Transporter
- Magnitude of Absorption Enhancement
- Relevance for Plant-Based Diets
- Overcoming Absorption Inhibitors
- Iron Deficiency Anemia Prevention
- Meal Timing Strategies
- Clinical Recommendations for Vegetarians and Vegans
- Research Papers
- Connections
- Featured Videos
Ferric to Ferrous Iron Reduction
The chemistry of iron absorption begins with understanding the two oxidation states of dietary iron.
- Ferric Iron (Fe3+) – Non-heme iron in plant foods, fortified foods, and iron supplements exists predominantly in the ferric (Fe3+) oxidation state. Ferric iron is poorly soluble at the near-neutral pH of the duodenum and upper jejunum, where the majority of iron absorption occurs. At pH above 3, ferric iron rapidly forms insoluble ferric hydroxide precipitates that cannot be absorbed.
- Ferrous Iron (Fe2+) – The ferrous form of iron is far more soluble at intestinal pH and is the form actively transported across the apical membrane of enterocytes. Conversion of ferric to ferrous iron is therefore a critical rate-limiting step in non-heme iron absorption.
- Ascorbate as a Reducing Agent – Vitamin C is an effective reducing agent with a reduction potential that thermodynamically favors the conversion of Fe3+ to Fe2+. In the acidic environment of the stomach, ascorbic acid donates an electron to ferric iron, reducing it to the absorbable ferrous form. This reaction occurs rapidly and efficiently at gastric pH.
- Duodenal Cytochrome b (Dcytb) – The brush border of duodenal enterocytes expresses Dcytb, a ferric reductase that also reduces Fe3+ to Fe2+ at the cell surface. Intracellular ascorbate serves as the electron donor for Dcytb, linking vitamin C status directly to the enzymatic reduction of iron at the absorption site. Individuals with low vitamin C status have reduced Dcytb activity.
Chelation Mechanism in the Gut
- Soluble Iron-Ascorbate Complex – Beyond its role as a reducing agent, vitamin C forms a soluble chelate complex with iron in the acidic gastric environment. This iron-ascorbate complex remains soluble as it passes into the alkaline duodenum, where unchelated ferric iron would otherwise precipitate as insoluble ferric hydroxide. The chelate effectively shields iron from the pH-dependent precipitation that is the major barrier to non-heme iron absorption.
- Molar Ratio – Studies indicate that a molar ratio of ascorbate to iron of approximately 2:1 to 4:1 provides optimal enhancement of absorption. In practical terms, 25 to 75 mg of vitamin C consumed with a meal containing 5 to 10 mg of non-heme iron provides a meaningful boost to absorption.
- Protection Through the GI Tract – The iron-ascorbate chelate maintains iron in a soluble and bioavailable form throughout the transit from the stomach to the absorptive sites in the duodenum and proximal jejunum, a journey during which pH rises from 2 to approximately 6.5.
DMT1 Transporter
- Divalent Metal Transporter 1 (DMT1) – DMT1 (also known as SLC11A2 or NRAMP2) is the primary apical membrane transporter responsible for importing ferrous iron into duodenal enterocytes. DMT1 is a proton-coupled symporter that transports Fe2+ along with a hydrogen ion across the brush border membrane. It has high selectivity for divalent cations and does not transport ferric iron (Fe3+).
- Vitamin C and DMT1 Coupling – The absolute requirement of DMT1 for ferrous iron means that any factor increasing the proportion of Fe2+ at the brush border surface will enhance iron uptake. Vitamin C accomplishes this through both its direct chemical reducing action and its support of Dcytb enzymatic activity, making it the most important dietary factor enhancing DMT1-mediated iron transport.
- DMT1 Regulation – DMT1 expression is upregulated by iron deficiency through the iron-responsive element/iron regulatory protein (IRE/IRP) system. When iron stores are low, DMT1 mRNA is stabilized, increasing transporter density on the enterocyte surface. In this iron-deficient state, the enhancing effect of vitamin C becomes even more pronounced, as more transport capacity is available to handle the increased supply of ferrous iron.
- Intracellular Processing – Once inside the enterocyte, iron can be stored as ferritin or exported across the basolateral membrane by ferroportin. Hephaestin, a copper-dependent ferroxidase on the basolateral surface, oxidizes Fe2+ back to Fe3+ for loading onto plasma transferrin. Intracellular ascorbate may also play a role in maintaining the labile iron pool within enterocytes.
Magnitude of Absorption Enhancement
The enhancement of non-heme iron absorption by vitamin C has been quantified in numerous isotope absorption studies.
- Dose-Response Relationship – The enhancing effect of vitamin C on iron absorption follows a dose-response curve. Adding 25 mg of ascorbic acid to a meal approximately doubles iron absorption. Adding 50 mg triples it. Adding 100 mg can increase absorption four- to sixfold. The effect plateaus at approximately 100 to 200 mg per meal.
- Baseline Absorption Rates – Without enhancers, non-heme iron absorption from a mixed diet typically ranges from 2 to 10 percent. With adequate vitamin C, absorption can increase to 10 to 30 percent or more, depending on iron status and the presence of inhibitors.
- Comparison with Other Enhancers – Vitamin C is the most potent known dietary enhancer of non-heme iron absorption. Meat, fish, and poultry (the "MFP factor") also enhance non-heme iron absorption but are less effective milligram-for-milligram than ascorbic acid. Organic acids such as citric acid and malic acid have modest enhancing effects but are far weaker than ascorbate.
- Long-Term Impact – While single-meal isotope studies show dramatic enhancements, the long-term impact on iron status is moderated by homeostatic regulation of iron absorption. Nevertheless, controlled supplementation trials confirm that regular vitamin C intake with meals meaningfully improves iron status indicators (serum ferritin, hemoglobin) over weeks to months.
Relevance for Plant-Based Diets
- Non-Heme Iron Predominance – Vegetarian and vegan diets provide iron exclusively in the non-heme form, which has inherently lower bioavailability (2 to 20 percent) compared to heme iron from animal sources (15 to 35 percent). This makes the enhancing effect of vitamin C particularly critical for individuals following plant-based eating patterns.
- Iron-Rich Plant Foods – Legumes (lentils, chickpeas, beans), tofu and tempeh, fortified cereals and breads, dark leafy greens (spinach, kale, Swiss chard), quinoa, nuts, and seeds are important plant-based iron sources. However, many of these foods also contain absorption inhibitors, making simultaneous vitamin C consumption essential.
- Practical Pairing Examples – Lentil soup with tomatoes and bell peppers; spinach salad with strawberries and lemon dressing; bean chili with tomato sauce; iron-fortified cereal with orange juice; tofu stir-fry with broccoli and red peppers. These combinations leverage the enhancing effect of vitamin C-rich foods consumed alongside iron-rich plant foods.
- Prevalence of Deficiency – While vegetarians and vegans typically consume as much or more total iron than omnivores, their iron stores (as measured by serum ferritin) tend to be lower, and iron deficiency anemia is more prevalent in these populations. Strategic vitamin C consumption with meals is one of the most effective and practical interventions.
Overcoming Absorption Inhibitors
Several dietary compounds inhibit non-heme iron absorption. Vitamin C can partially or fully overcome these inhibitory effects.
- Phytates (Inositol Hexaphosphate) – Phytic acid, found in whole grains, legumes, nuts, and seeds, is the most potent dietary inhibitor of iron absorption. Phytate binds ferric iron in insoluble complexes that resist digestion. Vitamin C can overcome phytate inhibition by reducing iron to the ferrous form before phytate binding occurs and by competing for iron binding through chelation. Studies show that 50 mg of ascorbic acid can counteract the inhibitory effect of moderate phytate loads.
- Tannins and Polyphenols – Tannins in tea, coffee, red wine, and certain fruits bind non-heme iron, reducing absorption by 50 to 90 percent. A single cup of tea consumed with a meal can reduce iron absorption by 60 percent. Vitamin C partially counteracts this effect, though very high tannin loads may require proportionally larger amounts of ascorbate. The practical recommendation is to consume tea and coffee between meals rather than with meals, and to include vitamin C-rich foods when polyphenol-rich beverages are consumed with iron-containing meals.
- Calcium – Calcium inhibits both heme and non-heme iron absorption, likely through an intracellular mechanism in the enterocyte. Doses of 300 to 600 mg of calcium (typical of a glass of milk or calcium supplement) can reduce iron absorption by 30 to 50 percent. Vitamin C can mitigate some of this inhibition, but the most practical strategy is to separate calcium and iron intake temporally.
- Soy Protein – Soy protein contains a specific inhibitory factor (possibly related to conglycinin) that reduces iron absorption independently of phytate. This is relevant for vegetarians and vegans who rely on soy products as protein sources. Consuming vitamin C with soy-based meals helps counter this effect.
- Oxalates – Oxalic acid in spinach, rhubarb, beet greens, and Swiss chard binds iron (and calcium) in insoluble complexes. While spinach is often cited as iron-rich, the bioavailability of its iron is very low due to high oxalate content. Vitamin C has a limited ability to overcome oxalate inhibition. Choosing low-oxalate greens like kale, collards, or bok choy provides more bioavailable iron.
Iron Deficiency Anemia Prevention
- Global Burden – Iron deficiency anemia affects approximately 1.2 billion people worldwide, with the highest prevalence in preschool children, women of reproductive age, and pregnant women in low- and middle-income countries. IDA reduces work capacity, impairs cognitive development in children, increases maternal mortality, and compromises immune function.
- Vitamin C in Public Health Interventions – Several public health strategies have successfully used vitamin C to enhance iron nutrition at the population level. Adding ascorbic acid to iron-fortified foods (flour, rice, condiments) improves the bioavailability of fortification iron. School meal programs that include vitamin C-rich fruits and vegetables alongside iron-rich foods have improved iron status in children.
- Supplementation Trials – Randomized controlled trials have demonstrated that co-supplementation with vitamin C and iron is more effective at improving hemoglobin and ferritin levels than iron supplementation alone, particularly in populations with marginal vitamin C status.
- Reducing Iron Supplement Side Effects – Some evidence suggests that vitamin C may reduce the gastrointestinal side effects (nausea, constipation, abdominal pain) of oral iron supplements by improving absorption efficiency, potentially allowing lower effective doses. This is clinically relevant because gastrointestinal side effects are the primary reason for non-adherence to iron supplementation.
Meal Timing Strategies
- Simultaneous Consumption – The enhancing effect of vitamin C on iron absorption requires simultaneous presence of both nutrients in the duodenum. Taking vitamin C hours before or after an iron-containing meal provides little or no enhancement. For maximum benefit, vitamin C-rich foods or supplements should be consumed during the meal.
- Optimal Meal Composition – The ideal iron-enhancing meal minimizes inhibitors and maximizes enhancers. A meal built around iron-rich foods (legumes, fortified grains, dark greens) with generous vitamin C-rich additions (citrus, peppers, tomatoes, berries) and limited tea, coffee, and high-calcium items will maximize iron absorption.
- Cooking Considerations – Vitamin C is heat-sensitive and partially destroyed during cooking. To preserve the iron-enhancing effect, add vitamin C-rich foods at the end of cooking, consume raw fruits and vegetables alongside cooked iron-rich dishes, or use a glass of citrus juice as a mealtime beverage.
- Iron Supplement Timing – When taking iron supplements, consuming them with a glass of orange juice (providing approximately 70 mg of vitamin C) or a vitamin C tablet significantly improves absorption compared to taking the supplement with water alone. Iron supplements are best absorbed on an empty stomach, but if gastrointestinal side effects occur, taking them with a small amount of food plus vitamin C is a reasonable compromise.
Clinical Recommendations for Vegetarians and Vegans
- Daily Iron Targets – The Institute of Medicine recommends that vegetarians consume 1.8 times the RDA for iron (14.4 mg for men and postmenopausal women; 32.4 mg for premenopausal women) to compensate for lower bioavailability of non-heme iron. Meeting these elevated targets requires deliberate dietary planning.
- Vitamin C Strategy – Include at least 25 to 75 mg of vitamin C with each iron-containing meal. This is easily achievable through dietary sources: one medium orange (70 mg), one cup of strawberries (85 mg), half a cup of red bell pepper (95 mg), or one cup of broccoli (80 mg).
- Food Preparation Techniques – Soaking and sprouting legumes and grains reduces phytate content by 30 to 70 percent, improving iron bioavailability. Fermentation (as in sourdough bread or tempeh) also degrades phytates. Combining these preparation methods with vitamin C consumption at meals provides the highest non-heme iron bioavailability.
- Monitoring Iron Status – Vegetarians and vegans should have their iron status assessed regularly through serum ferritin testing. A ferritin level below 30 micrograms per liter suggests depleted stores and warrants dietary modification or supplementation. Women of reproductive age, athletes, and frequent blood donors on plant-based diets are at the highest risk.
- Cast Iron Cookware – Cooking acidic, vitamin C-rich foods (such as tomato sauce) in cast iron cookware can increase the iron content of the food by several milligrams per serving. The acidic environment and the reducing action of vitamin C leach small amounts of iron from the cooking surface in a bioavailable form.
- Avoid Competing Nutrients at Iron Meals – Separate calcium supplements and high-calcium foods from primary iron meals. Consume tea and coffee between meals rather than during meals. Time iron-focused meals for when inhibitor intake is minimized and vitamin C intake is maximized.
Research Papers
Curated PubMed topic searches on Vitamin C and iron absorption. Each link opens a live PubMed search that aggregates the current literature.
- Vitamin C and iron absorption
- Ascorbic acid and non-heme iron bioavailability
- Phytate inhibition of iron absorption — Vitamin C
- Tannins and iron absorption
- DMT1 and duodenal iron transport
- Duodenal cytochrome b (Dcytb) and ascorbate
- Iron deficiency anemia in vegetarian diets
- Iron fortification with ascorbic acid
- Ferritin response to Vitamin C + iron supplementation
- Hallberg — iron absorption and ascorbic acid
- Calcium-iron absorption interaction
- Pregnancy, iron deficiency, and Vitamin C
Connections
- Vitamin C
- Iron
- Vitamin C and Immune Defense
- Vitamin C and Collagen
- Anemia
- Calcium
- Zinc
- Lentils
- Beef
- Spinach
- Hemoglobin A1C
- Fatigue
- Vitamin B12