Vitamin B12 Deficiency: Diagnosis and Clinical Management
The diagnosis of vitamin B12 deficiency is a deceptively complex clinical challenge. Serum B12 levels, the most commonly ordered test, have significant limitations in sensitivity and specificity. Clinically significant B12 deficiency can exist despite "normal" serum levels, and conversely, low serum levels do not always indicate true tissue deficiency. A thorough understanding of the available diagnostic tools, their strengths and limitations, and the appropriate use of confirmatory biomarkers is essential for accurate diagnosis and timely treatment of this common and potentially serious condition.
Table of Contents
- Key Points at a Glance
- Serum B12 Limitations
- Methylmalonic Acid (MMA) as Gold Standard
- Homocysteine Elevation
- Holotranscobalamin (Active B12)
- Complete Blood Count Findings
- At-Risk Populations
- Schilling Test (Historical)
- Intrinsic Factor Antibodies
- Treatment Protocols
- Monitoring Response
- Research Papers
- Connections
- Featured Videos
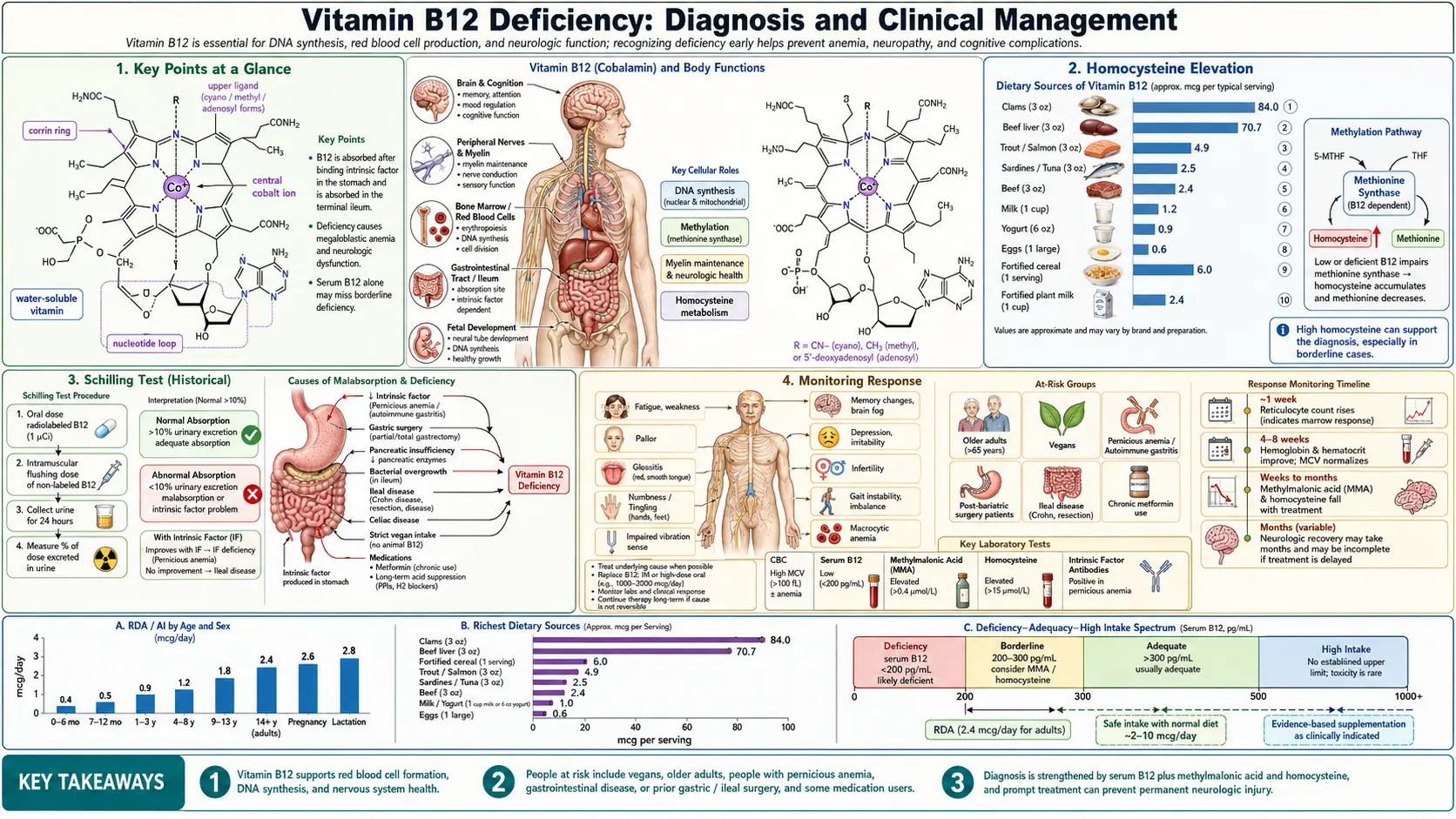
Key Points at a Glance
- Serum B12 is insensitive — up to one-third of truly deficient patients have "normal" results.
- MMA is the gold standard — highly specific for tissue-level B12 deficiency.
- Homocysteine distinguishes combined B12/folate deficiency patterns.
- Holotranscobalamin measures the metabolically active fraction and decreases early.
- Neurological symptoms can occur without anemia — up to 28% of neurological B12 deficiency has normal CBC.
- At-risk groups: vegans, elderly, pernicious anemia, metformin users, post-bariatric, Crohn's disease.
- High-dose oral B12 (1,000–2,000 mcg/day) matches IM efficacy for most patients, including pernicious anemia.
- Neurological recovery may require 3–12+ months; early treatment is critical.
Serum B12 Limitations
Despite being the first-line screening test for B12 deficiency, serum cobalamin measurement has well-documented shortcomings that clinicians must understand.
- Sensitivity issues: Serum B12 has a sensitivity of only approximately 65 to 95% for detecting clinically significant B12 deficiency. This means that up to one-third of patients with genuine tissue-level deficiency may have serum B12 levels within the "normal" reference range, particularly in the low-normal range of 200 to 400 pg/mL (148 to 295 pmol/L).
- Biologically inactive analogs: Standard serum B12 assays measure total cobalamin, including biologically inactive cobalamin analogues that cannot serve as enzyme cofactors. These analogues may constitute a significant proportion of measured B12, particularly in patients with small intestinal bacterial overgrowth or those consuming certain algal products marketed as B12 sources.
- Conditions causing falsely normal levels: Liver disease, myeloproliferative disorders, and chronic kidney disease can elevate serum B12 levels by increasing haptocorrin (transcobalamin I) concentrations, potentially masking functional B12 deficiency. Conversely, oral contraceptives, pregnancy, folate deficiency, and multiple myeloma can lower serum B12 without indicating true deficiency.
- Gray zone interpretation: Serum B12 levels between 200 and 400 pg/mL represent a diagnostic gray zone in which approximately 5 to 25% of patients will have metabolic evidence of tissue-level deficiency (elevated methylmalonic acid or homocysteine). These borderline cases require additional testing for accurate diagnosis.
- Assay variability: Different laboratory platforms may produce discrepant results for the same sample, with interassay variability of up to 20%. This variability complicates serial monitoring and comparison of results obtained at different laboratories.
Methylmalonic Acid (MMA) as Gold Standard
Serum or urine methylmalonic acid is widely considered the most reliable biomarker for confirming B12 deficiency at the tissue level.
- Biochemical rationale: Methylmalonyl-CoA mutase, which requires adenosylcobalamin as a cofactor, converts methylmalonyl-CoA to succinyl-CoA. When B12 is deficient, this enzyme's activity is impaired and methylmalonyl-CoA accumulates, leading to elevated methylmalonic acid (MMA) in the serum and urine. MMA elevation is highly specific to B12 deficiency and is not affected by folate status.
- Diagnostic performance: Serum MMA has a sensitivity of approximately 86 to 98% for B12 deficiency, significantly exceeding that of serum B12 alone. An MMA level above 0.4 micromol/L (some laboratories use a cutoff of 0.27 or 0.37 micromol/L) strongly supports the diagnosis of B12 deficiency, particularly when serum B12 is in the borderline range.
- Specificity considerations: While MMA is specific to B12 deficiency (not elevated in isolated folate deficiency), it can be elevated by renal insufficiency (the most common confounding factor), dehydration, small intestinal bacterial overgrowth, and rare inherited disorders of methylmalonic acid metabolism. Renal function should be assessed when interpreting MMA levels.
- Urine MMA: Urine MMA (often measured as urinary methylmalonic acid-to-creatinine ratio) is an alternative to serum MMA that may be less affected by renal function. However, serum MMA remains more widely available and better validated in clinical practice.
- Clinical decision-making: An elevated MMA in the setting of low or borderline serum B12 confirms the diagnosis of B12 deficiency and warrants treatment. Conversely, a normal MMA in a patient with borderline serum B12 argues against clinically significant deficiency, though monitoring may be appropriate.
Homocysteine Elevation
- Biochemical basis: B12 is a cofactor for methionine synthase, which remethylates homocysteine to methionine. B12 deficiency impairs this reaction, leading to homocysteine accumulation. Plasma homocysteine levels above 13 to 15 micromol/L support the diagnosis of B12 deficiency when considered alongside other findings.
- Sensitivity but limited specificity: Homocysteine elevation is a sensitive marker of B12 deficiency (elevated in approximately 95% of deficient patients) but lacks specificity, as it is also elevated in folate deficiency, vitamin B6 deficiency, renal insufficiency, hypothyroidism, advancing age, and in individuals with MTHFR polymorphisms. It is most useful when combined with MMA measurement.
- Distinguishing B12 from folate deficiency: The combination of MMA and homocysteine is diagnostically powerful: B12 deficiency elevates both MMA and homocysteine, while folate deficiency elevates homocysteine but not MMA. This distinction is critical for directing appropriate therapy.
- Cardiovascular implications: Beyond its diagnostic utility, elevated homocysteine from B12 deficiency has independent pathophysiological significance as a cardiovascular risk factor. Treatment of B12 deficiency with consequent homocysteine reduction may confer cardiovascular benefit, though this remains debated based on large clinical trial data.
Holotranscobalamin (Active B12)
- Physiology: In the circulation, B12 is bound to two transport proteins: approximately 70 to 80% is bound to haptocorrin (transcobalamin I), which has no known delivery function, while 20 to 30% is bound to transcobalamin II, forming holotranscobalamin (holoTC). Only holoTC is taken up by cells via the transcobalamin receptor and represents the biologically available fraction of circulating B12.
- Diagnostic advantages: HoloTC measurement provides an assessment of the metabolically active fraction of B12 and may be a more sensitive early marker of B12 depletion. Studies suggest holoTC decreases before total serum B12 in the progression toward deficiency, making it a useful early indicator.
- Reference ranges: HoloTC levels below 35 to 40 pmol/L are generally considered indicative of B12 deficiency, while levels above 70 pmol/L effectively exclude deficiency. The intermediate range (35 to 70 pmol/L) requires additional confirmatory testing with MMA and/or homocysteine.
- Availability: HoloTC assays have become increasingly available in clinical laboratories, though they are not yet as universally accessible as serum B12 or MMA measurements. Some guidelines now recommend holoTC as a first-line test, either alone or in combination with serum B12.
Complete Blood Count Findings
The hematological manifestations of B12 deficiency, while classic, are neither universal nor specific, and their absence should not exclude the diagnosis.
- Megaloblastic anemia: The hallmark hematological finding is macrocytic (megaloblastic) anemia, characterized by elevated mean corpuscular volume (MCV) typically above 100 fL. The anemia results from impaired DNA synthesis in erythroid precursors, leading to nuclear-cytoplasmic dissynchrony and the production of abnormally large red blood cells (macroovalocytes).
- Hypersegmented neutrophils: The presence of neutrophils with six or more nuclear lobes (or more than 5% of neutrophils with five lobes) on the peripheral blood smear is a sensitive and relatively specific marker of megaloblastic anemia. Hypersegmented neutrophils may appear before macrocytosis becomes evident and persist after other features resolve with treatment.
- Pancytopenia: Severe B12 deficiency can produce pancytopenia (anemia, leukopenia, and thrombocytopenia) due to ineffective hematopoiesis affecting all cell lineages. In severe cases, the bone marrow appearance can mimic myelodysplastic syndrome or acute leukemia, leading to diagnostic confusion.
- Neurological symptoms without anemia: Critically, neurological manifestations of B12 deficiency can occur in the complete absence of anemia or macrocytosis. Up to 28% of patients with neurological B12 deficiency have normal hematological indices. Concurrent iron deficiency or thalassemia trait can mask macrocytosis, producing a normal or even low MCV despite significant B12 deficiency.
- Pseudothrombotic microangiopathy: Severe B12 deficiency can produce a laboratory picture resembling thrombotic thrombocytopenic purpura (TTP), with schistocytes on the peripheral smear, elevated lactate dehydrogenase, elevated indirect bilirubin, and thrombocytopenia. Recognition of this B12-related pseudoTTP prevents unnecessary plasmapheresis.
At-Risk Populations
Certain populations have a substantially elevated risk of B12 deficiency and merit proactive screening.
- Vegans and strict vegetarians: B12 is found naturally only in animal-derived foods (meat, fish, dairy, eggs). Vegans who do not supplement or consume B12-fortified foods will inevitably develop deficiency, typically within 2 to 5 years of depleting hepatic stores. Lacto-ovo vegetarians are also at increased risk if dairy and egg consumption is limited.
- Elderly populations: The prevalence of B12 deficiency increases substantially with age, affecting an estimated 10 to 15% of adults over age 60. The primary mechanism is food-cobalamin malabsorption due to atrophic gastritis (present in 20 to 30% of elderly individuals), which reduces gastric acid and pepsin secretion needed to release B12 from food proteins.
- Pernicious anemia: This autoimmune condition, characterized by antibodies against gastric parietal cells and/or intrinsic factor, destroys the mechanism for B12 absorption in the terminal ileum. Pernicious anemia is the most common cause of severe B12 deficiency in developed countries, with a prevalence of approximately 1 to 2% in individuals over age 60.
- Metformin users: Metformin, the most widely prescribed diabetes medication, reduces B12 absorption in 10 to 30% of users through a mechanism involving calcium-dependent ileal membrane changes. The risk increases with higher metformin doses and longer duration of therapy. Annual B12 monitoring is recommended for patients on long-term metformin.
- Gastric bypass patients: Roux-en-Y gastric bypass and other bariatric procedures that exclude the gastric fundus or terminal ileum substantially impair B12 absorption. Lifelong B12 supplementation, often via parenteral route, is required for all post-gastric bypass patients. Deficiency can develop within 1 to 3 years post-surgery without supplementation.
- Other at-risk groups: Patients with inflammatory bowel disease (particularly Crohn's disease affecting the terminal ileum), celiac disease, chronic proton pump inhibitor or H2 receptor antagonist use, nitrous oxide exposure, and those with pancreatic insufficiency are also at increased risk for B12 deficiency.
Schilling Test (Historical)
- Principle: The Schilling test, developed by Robert Schilling in 1953, was the gold standard for evaluating the cause of B12 malabsorption for nearly five decades. It involved oral administration of radiolabeled cyanocobalamin followed by measurement of 24-hour urinary excretion of the labeled vitamin. An intramuscular injection of unlabeled B12 was given to saturate binding proteins and ensure renal excretion of absorbed labeled B12.
- Staged testing: The test was performed in stages: Stage I (labeled B12 alone) assessed overall absorption; Stage II (labeled B12 plus intrinsic factor) determined whether malabsorption was due to intrinsic factor deficiency (pernicious anemia); and Stage III (after antibiotic treatment) evaluated bacterial overgrowth as a cause of malabsorption.
- Discontinuation: The Schilling test has been largely unavailable since the early 2000s due to discontinuation of the radiolabeled B12 reagent. Its loss has created a diagnostic gap in evaluating the etiology of B12 malabsorption, though serological testing for intrinsic factor antibodies and anti-parietal cell antibodies provides a partial substitute.
Intrinsic Factor Antibodies
- Diagnostic role: Intrinsic factor antibodies (IFAb) are highly specific (close to 100%) for pernicious anemia, making a positive result essentially diagnostic. However, sensitivity is limited, with IFAb detected in only approximately 50 to 70% of patients with confirmed pernicious anemia. A negative IFAb result therefore does not exclude the diagnosis.
- Types of antibodies: Two types of intrinsic factor antibodies exist: Type I (blocking) antibodies prevent the binding of B12 to intrinsic factor, while Type II (precipitating or binding) antibodies bind the B12-intrinsic factor complex and prevent its attachment to the ileal receptor. Type I antibodies are more commonly measured and clinically relevant.
- Anti-parietal cell antibodies: Antibodies against gastric parietal cells (specifically targeting the H+/K+ ATPase proton pump) are more sensitive (approximately 80 to 90%) but less specific than IFAb for pernicious anemia, as they are also found in other autoimmune conditions and in up to 10% of the general population. They are most useful in combination with IFAb testing.
- Gastrin levels: Fasting serum gastrin is elevated in pernicious anemia due to loss of acid-mediated negative feedback on gastrin-secreting G cells. Elevated gastrin supports the diagnosis of atrophic gastritis and can complement antibody testing.
Treatment Protocols
Treatment of B12 deficiency varies based on the underlying cause, severity of deficiency, and presence of neurological involvement.
Intramuscular Injections
- Standard protocol: For documented B12 deficiency with neurological involvement or malabsorption, the standard treatment is intramuscular (IM) cyanocobalamin or hydroxocobalamin 1,000 mcg. A typical regimen is daily injections for 7 days, then weekly injections for 4 weeks, followed by monthly injections indefinitely (or until the underlying cause is corrected).
- Hydroxocobalamin vs. cyanocobalamin: Hydroxocobalamin has a longer retention time in the body (requiring less frequent injections in some protocols) and is preferred in many European countries. Cyanocobalamin is more widely used in North America. Both are effective when administered on appropriate schedules.
- Neurological emergency protocol: For patients with acute or rapidly progressive neurological symptoms (myelopathy, severe neuropathy), more aggressive initial dosing with daily IM injections for 10 to 14 days is recommended. Some experts advocate for every-other-day injections for 3 weeks in severe neurological cases.
Sublingual Administration
- Mechanism: Sublingual B12 is absorbed directly through the oral mucosa, bypassing the gastrointestinal tract. This route may be advantageous for patients with gastric or ileal malabsorption, though the amount absorbed sublingually varies among individuals.
- Efficacy evidence: Limited studies comparing sublingual to intramuscular B12 have shown equivalent efficacy in correcting serum B12 and MMA levels. A commonly used sublingual dose is 1,000 to 5,000 mcg daily. However, the evidence base is smaller than for IM or high-dose oral routes.
High-Dose Oral Therapy
- Rationale: Approximately 1 to 2% of oral B12 is absorbed by passive diffusion along the entire intestinal mucosa, independent of intrinsic factor and ileal receptor function. At a dose of 1,000 to 2,000 mcg orally, passive absorption delivers 10 to 20 mcg, sufficient to meet daily requirements (2.4 mcg).
- Clinical evidence: Multiple randomized controlled trials and systematic reviews have demonstrated that high-dose oral B12 (1,000 to 2,000 mcg daily) is as effective as intramuscular injections for correcting B12 deficiency and normalizing MMA levels, even in patients with pernicious anemia and food-cobalamin malabsorption.
- Advantages: Oral therapy is less invasive, more convenient, less costly, and does not require healthcare provider visits for administration. It empowers patient self-management and improves adherence in many populations.
- Limitations: Oral therapy may not be appropriate for patients with severe neurological involvement (where rapid repletion is critical), those with significant ileal disease or resection, or patients with poor medication adherence. Some experts recommend initiating treatment with IM injections and transitioning to oral therapy after neurological stabilization.
Monitoring Response
- Reticulocyte response: The earliest hematological response to B12 replacement is a reticulocyte crisis, typically occurring 5 to 7 days after initiating treatment. The reticulocyte count peaks at 7 to 10 days and returns to normal as anemia corrects. Absence of a reticulocyte response should prompt reconsideration of the diagnosis or investigation of concurrent iron or folate deficiency.
- Hemoglobin recovery: Hemoglobin levels begin to rise within 1 to 2 weeks and typically normalize within 6 to 8 weeks. MCV may take longer to normalize (up to 8 to 12 weeks) as the macrocytic red cell population is gradually replaced by normal-sized cells.
- MMA and homocysteine normalization: Serum MMA and homocysteine levels begin to decline within days of starting treatment and typically normalize within 1 to 2 weeks. Persistent elevation after adequate treatment may indicate concurrent renal insufficiency, inadequate dosing, or incorrect diagnosis.
- Neurological recovery timeline: Neurological improvement is slower than hematological recovery and follows a characteristic pattern. Sensory symptoms (paresthesias) often begin to improve within weeks. Proprioceptive and motor recovery may take 3 to 12 months. Cognitive improvement is variable and may continue for up to 2 years after starting treatment.
- Follow-up testing schedule: After initiating treatment, serum B12 should be rechecked at 1 to 2 months to confirm adequate repletion. MMA may be repeated at 2 to 3 months to confirm metabolic correction. For patients on long-term replacement, annual serum B12 and complete blood count monitoring is recommended. More frequent monitoring (every 3 to 6 months) is warranted when compliance is uncertain or when neurological symptoms are present.
- Potassium monitoring: In patients with severe megaloblastic anemia, initiation of B12 therapy can cause rapid cellular uptake of potassium as red cell production accelerates, potentially producing dangerous hypokalemia. Serum potassium should be monitored closely during the first 48 to 72 hours of treatment in severely anemic patients, particularly in the elderly or those with cardiac disease.
Summary
Accurate diagnosis of B12 deficiency requires awareness of the limitations of serum B12 measurement and appropriate use of confirmatory biomarkers including methylmalonic acid, homocysteine, and holotranscobalamin. At-risk populations, including vegans, the elderly, patients with pernicious anemia, metformin users, and post-gastric bypass patients, should receive proactive screening. Treatment options include intramuscular injections (standard of care for neurological involvement), high-dose oral therapy (evidence-based alternative for most patients), and sublingual administration. Monitoring should include both hematological and metabolic markers, and neurological recovery should be assessed over months to years. Early diagnosis and prompt treatment remain the most important factors in preventing permanent neurological damage.
Research Papers
- PubMed — Methylmalonic acid in B12 deficiency diagnosis (topic search)
- PubMed — Holotranscobalamin in B12 deficiency (topic search)
- PubMed — Oral vs. intramuscular B12 in pernicious anemia (topic search)
- PubMed — Metformin-induced B12 deficiency (topic search)
- PubMed — Subacute combined degeneration of the spinal cord (topic search)
- PubMed — Intrinsic factor antibodies in pernicious anemia (topic search)
- PubMed — Nitrous oxide and B12 deficiency (topic search)
- PubMed — Vegan diet and B12 status (topic search)
- PubMed — Food-cobalamin malabsorption in the elderly (topic search)
- NIH Office of Dietary Supplements — Vitamin B12 Fact Sheet for Health Professionals
- Linus Pauling Institute — Vitamin B12
- Harvard T.H. Chan School of Public Health — Vitamin B12
Connections
- Vitamin B12
- B12 and the Nervous System
- Anemia
- Complete Blood Count
- Homocysteine
- Vitamin B12 Test
- Iron
- Vitamin B9
- Folate and Mental Health
- Vitamin B6 and Homocysteine
- Vitamin B6
- Depression
- Crohn's Disease
- Celiac Disease
- Thrombocytopenia
- Methionine
- Folate and Pregnancy