Vitamin A and Immune System Function
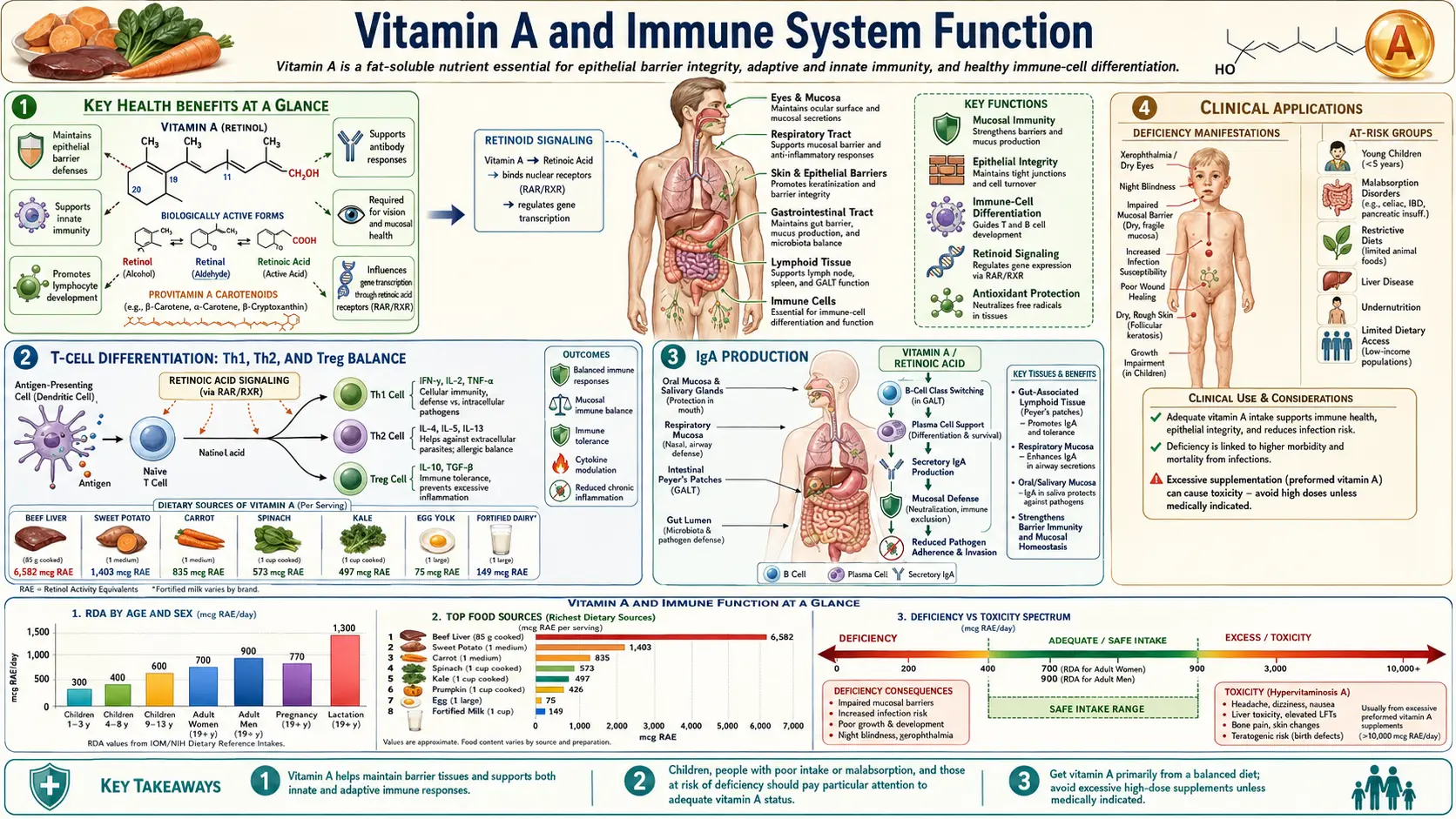
Vitamin A has long been recognized as the “anti-infective vitamin,” a term coined in the 1920s when researchers first observed that deficiency dramatically increased susceptibility to infectious diseases. Today, the immunological roles of vitamin A and its metabolically active derivatives — particularly all-trans retinoic acid (ATRA) — are understood in remarkable molecular detail. Vitamin A influences virtually every arm of the immune system, from the physical barrier defenses of mucosal epithelia, to the differentiation of T-helper cells, to the gut-homing imprint placed on lymphocytes by intestinal dendritic cells. At the public-health scale, two doses of high-dose vitamin A in measles-infected children reduce mortality by roughly 50–80%, and universal supplementation in deficient regions cuts all-cause childhood mortality by about 24% — ranking vitamin A among the most cost-effective nutritional interventions in global health.
Table of Contents
- Key Health Benefits at a Glance
- Mucosal Barrier Integrity
- Epithelial Cell Differentiation
- T-Cell Differentiation: Th1, Th2, and Treg Balance
- Retinoic Acid Signaling
- Gut-Associated Lymphoid Tissue (GALT)
- IgA Production
- Measles Mortality Reduction
- Deficiency and Infection Susceptibility
- Clinical Applications
- Research Papers and References
- Connections
- Featured Videos
Key Health Benefits at a Glance
The following is a high-level summary of the evidence-backed immune benefits of adequate vitamin A status. Each is explored in depth below, and the supporting landmark papers are listed in the Research Papers section.
- Maintains mucosal barriers – Retinoic acid stimulates goblet-cell differentiation and upregulates tight-junction proteins (claudin, occludin, ZO), keeping the respiratory, gut, and urogenital epithelia structurally and functionally intact.
- Cuts measles mortality by ~50–80% – Controlled trials in hospitalized children showed that two consecutive days of high-dose vitamin A dramatically reduced measles-related death, leading to the WHO’s universal treatment recommendation.
- Cuts all-cause under-five mortality by ~24% – Universal supplementation in at-risk populations is one of the most cost-effective public-health interventions on record (Stephensen 2001; WHO).
- Drives IgA class switching – Retinoic acid produced by intestinal dendritic cells triggers B-cell class switch to IgA and promotes plasma-cell survival in the lamina propria, producing the >3 g of secretory IgA made daily.
- Shapes the Th1/Th2/Treg/Th17 balance – Retinoic acid favors Th2 and Foxp3+ regulatory T-cell programs while suppressing pro-inflammatory Th17 differentiation, a pattern relevant to autoimmunity and food allergy.
- Imprints gut-homing on lymphocytes – Intestinal dendritic cells, conditioned by retinoic acid, imprint integrin α4β7 and CCR9 on activated T and B cells, directing them back to the mucosa where they are needed.
- Supports innate lymphoid cells – Type-3 innate lymphoid cells (ILC3s) require retinoic acid for development and IL-22 production, which strengthens epithelial defenses.
- Reduces pediatric diarrhea and pneumonia – Meta-analyses show deficient children have 2–3× the risk of lower respiratory infections and increased diarrheal severity; supplementation reduces both.
- Therapeutic leverage in leukemia – ATRA, the active metabolite, converted acute promyelocytic leukemia from one of the deadliest leukemias to one of the most curable — a translational triumph originating in nutritional biology.
Mucosal Barrier Integrity
The body’s first line of defense against pathogens consists of the epithelial surfaces that line the respiratory tract, gastrointestinal tract, urogenital tract, and skin. Vitamin A is essential for maintaining the structural and functional integrity of these mucosal barriers:
- Mucus production – Vitamin A stimulates goblet cell differentiation and mucin gene expression. Goblet cells produce the mucus layer that traps pathogens, allergens, and particulate matter, preventing them from reaching the underlying epithelium. In deficiency, goblet cells are lost and replaced by keratinized squamous epithelium through a process called squamous metaplasia.
- Tight junctions – Retinoic acid upregulates the expression of tight junction proteins including claudins, occludin, and zonula occludens proteins. These molecular complexes seal the spaces between adjacent epithelial cells, preventing paracellular passage of pathogens and toxins.
- Antimicrobial peptides – Vitamin A supports the production of defensins and other antimicrobial peptides by epithelial cells. These small cationic proteins directly kill bacteria, fungi, and enveloped viruses by disrupting their membrane integrity.
- Epithelial turnover – The rapid turnover of mucosal epithelial cells (every 3–5 days in the intestine) requires continuous vitamin A signaling to maintain proper differentiation and replacement of damaged or senescent cells.
Epithelial Cell Differentiation
Retinoic acid acts as a master regulator of epithelial cell fate decisions. Through binding to nuclear retinoic acid receptors (RARs) and retinoid X receptors (RXRs), it controls the expression of hundreds of genes involved in cell proliferation, differentiation, and apoptosis.
In vitamin A deficiency, several pathological changes occur in epithelial tissues:
- Squamous metaplasia – Normal columnar or cuboidal epithelium is replaced by stratified squamous epithelium. This affects the respiratory tract (increasing susceptibility to pneumonia), the urinary tract (predisposing to urinary tract infections), and the conjunctiva (leading to xerophthalmia).
- Loss of ciliated cells – In the respiratory epithelium, ciliated cells that normally propel mucus and trapped particles upward via the mucociliary escalator are lost. This impairs clearance of inhaled pathogens and increases the risk of lower respiratory infections.
- Keratinization – Epithelial surfaces become dry, thickened, and keratinized, losing their ability to secrete protective substances and creating an environment favorable for bacterial colonization.
T-Cell Differentiation: Th1, Th2, and Treg Balance
One of the most significant immunological roles of vitamin A lies in directing the differentiation of naive CD4+ T helper cells into specific effector subtypes. Retinoic acid profoundly influences the balance among T helper 1 (Th1), T helper 2 (Th2), T helper 17 (Th17), and regulatory T cell (Treg) populations:
- Th1/Th2 balance – Retinoic acid generally promotes Th2 differentiation while modulating excessive Th1 responses. Th2 cells produce cytokines (IL-4, IL-5, IL-13) that drive humoral immunity, including antibody production and defense against extracellular parasites. In vitamin A deficiency, there is often an imbalanced Th1 response, leading to excessive inflammation without effective pathogen clearance.
- Regulatory T cells (Tregs) – Retinoic acid is a potent inducer of Foxp3+ regulatory T cells, particularly in the gut mucosa. Tregs suppress excessive immune activation and maintain tolerance to dietary antigens and commensal microorganisms. This function is critical for preventing autoimmune reactions and food allergies.
- Th17 suppression – Retinoic acid inhibits the differentiation of Th17 cells, which produce the pro-inflammatory cytokine IL-17. While Th17 cells are important for defense against extracellular bacteria and fungi, excessive Th17 activity is implicated in autoimmune diseases including inflammatory bowel disease, psoriasis, and rheumatoid arthritis.
Retinoic Acid Signaling
Retinoic acid (RA) is the primary mediator of vitamin A’s immunological effects. It is produced locally in tissues by a two-step enzymatic process: retinol is first oxidized to retinaldehyde by alcohol dehydrogenases, then to retinoic acid by retinaldehyde dehydrogenases (RALDH enzymes).
- All-trans retinoic acid (ATRA) – The most biologically active form, ATRA binds to RARs (alpha, beta, and gamma subtypes) which heterodimerize with RXRs to form transcription factor complexes. These bind to retinoic acid response elements (RAREs) in the promoter regions of target genes.
- Dendritic cell conditioning – Intestinal dendritic cells express high levels of RALDH enzymes, enabling them to produce retinoic acid locally. This conditions the dendritic cells to imprint gut-homing markers (integrin alpha4beta7 and CCR9) on the T and B cells they activate, ensuring immune cells migrate to the intestinal mucosa where they are needed.
- Macrophage polarization – Retinoic acid promotes the differentiation of monocytes into tissue-resident macrophages with anti-inflammatory (M2-like) properties, supporting tissue repair and resolution of inflammation.
Gut-Associated Lymphoid Tissue (GALT)
The gastrointestinal tract houses the largest mass of lymphoid tissue in the body, collectively known as the gut-associated lymphoid tissue. GALT includes Peyer’s patches, isolated lymphoid follicles, mesenteric lymph nodes, and the diffuse immune cells scattered throughout the lamina propria. Vitamin A is critical for GALT function at multiple levels:
- Peyer’s patch development – Retinoic acid signaling is required during embryonic development for the proper formation of Peyer’s patches, the organized lymphoid structures that sample antigens from the intestinal lumen via specialized M cells.
- Lymphocyte homing – As noted above, retinoic acid produced by intestinal dendritic cells induces expression of gut-homing receptors on activated lymphocytes. Without this imprinting, immune cells fail to migrate to the gut mucosa effectively.
- Oral tolerance – The GALT must distinguish between harmful pathogens and harmless dietary proteins and commensal bacteria. Retinoic acid promotes oral tolerance by inducing Tregs and anti-inflammatory cytokines (IL-10, TGF-beta) in the gut environment.
- Innate lymphoid cells – Type 3 innate lymphoid cells (ILC3s) in the gut require retinoic acid for their development and function. ILC3s produce IL-22, which strengthens epithelial barrier function and stimulates antimicrobial peptide production.
IgA Production
Secretory immunoglobulin A (sIgA) is the dominant antibody isotype at mucosal surfaces and represents the single most abundantly produced immunoglobulin in the human body, with daily production exceeding 3 grams. Vitamin A is essential for optimal IgA responses:
- Class switch recombination – Retinoic acid promotes immunoglobulin class switching from IgM to IgA in B cells. This process occurs primarily in Peyer’s patches and mesenteric lymph nodes, where B cells are exposed to retinoic acid produced by local dendritic cells.
- Plasma cell differentiation – After class switching, B cells must differentiate into IgA-secreting plasma cells. Retinoic acid enhances this differentiation and promotes the survival of IgA plasma cells in the lamina propria.
- Immune exclusion – Secretory IgA molecules coat pathogens and toxins in the intestinal lumen, preventing their attachment to and penetration of the epithelium. This process, called immune exclusion, is the primary mechanism by which mucosal immunity prevents enteric infections.
- Deficiency consequences – Vitamin A-deficient individuals show markedly reduced fecal and salivary IgA levels, contributing to their heightened susceptibility to gastrointestinal and respiratory infections.
Measles Mortality Reduction
The relationship between vitamin A and measles outcomes represents one of the most compelling examples of nutritional immunology translated into public health practice:
- Mortality reduction – Multiple randomized controlled trials have demonstrated that high-dose vitamin A supplementation (200,000 IU for children over 12 months) during acute measles illness reduces measles-related mortality by approximately 50–80% in hospitalized children.
- WHO recommendation – The World Health Organization recommends administering two doses of vitamin A (given on consecutive days) to all children diagnosed with measles, regardless of their country of residence. This recommendation applies even in developed nations.
- Mechanism – Measles virus itself depletes vitamin A stores by increasing urinary losses and reducing intestinal absorption. The resulting deficiency impairs immune clearance of the virus and compromises epithelial barrier recovery, leading to secondary bacterial pneumonia and diarrhea – the proximate causes of measles death.
Deficiency and Infection Susceptibility
Vitamin A deficiency creates a vicious cycle with infectious disease: deficiency increases infection risk, and infections further deplete vitamin A stores through reduced absorption, increased metabolic demand, and urinary losses. Specific infectious disease associations include:
- Respiratory infections – Deficient children have a 2–3 fold increased risk of lower respiratory tract infections, including pneumonia, which is the leading cause of childhood death globally.
- Diarrheal diseases – Impaired mucosal immunity and reduced IgA production increase susceptibility to rotavirus, Escherichia coli, and other enteric pathogens. Diarrheal episodes are more frequent, more severe, and longer-lasting in deficient children.
- Malaria – Observational studies suggest that vitamin A deficiency is associated with increased malaria parasite density and clinical malaria episodes, although supplementation trial results have been mixed.
- HIV/AIDS – Vitamin A deficiency is common in HIV-infected individuals and is associated with accelerated disease progression, increased viral load, and higher mortality. However, supplementation strategies in HIV require careful consideration of potential interactions with antiretroviral therapy.
Clinical Applications
The immunological functions of vitamin A have numerous clinical applications beyond treatment of frank deficiency:
- Supplementation programs – Universal vitamin A supplementation programs targeting children aged 6–59 months in deficient populations have reduced all-cause mortality by approximately 24%, representing one of the most cost-effective public health interventions available.
- Autoimmune disease – The ability of retinoic acid to promote Tregs and suppress Th17 responses has generated interest in therapeutic applications for autoimmune conditions. Clinical trials are exploring retinoic acid supplementation in inflammatory bowel disease and type 1 diabetes.
- Cancer immunology – All-trans retinoic acid (ATRA) is already used therapeutically in acute promyelocytic leukemia and is being investigated for its ability to modulate immune responses in solid tumors by reducing myeloid-derived suppressor cell populations.
Ensuring adequate vitamin A intake through dietary sources or supplementation programs remains a cornerstone strategy for supporting immune competence, particularly in vulnerable populations including young children, pregnant and lactating women, and individuals in resource-limited settings where infectious diseases continue to exact a devastating toll.
Research Papers and References
The following are landmark and frequently cited research papers underpinning the claims on this page. Links resolve to the publisher DOI or PubMed record.
Foundational Reviews and Historical Overviews
- Sommer A. Vitamin A deficiency and clinical disease: an historical overview. J Nutr. 2008;138(10):1835-1839.
- Stephensen CB. Vitamin A, infection, and immune function. Annu Rev Nutr. 2001;21:167-192.
Mucosal Immunity, IgA, and Retinoic Acid Signaling
- PubMed — Retinoic acid, IgA class-switch and dendritic-cell imprinting of gut-homing lymphocytes
- PubMed — Retinoic acid and Foxp3+ Treg induction in intestinal mucosa
- PubMed — Retinoic acid suppression of Th17 differentiation
- PubMed — ILC3 development, IL-22, and retinoic acid
Measles, Infection, and Supplementation Trials
- PubMed — Vitamin A and measles mortality RCTs
- PubMed — Vitamin A supplementation and all-cause child mortality meta-analyses
- PubMed — Vitamin A deficiency and pediatric pneumonia / diarrhea
Retinoids in Therapeutics (APL, Autoimmunity)
- PubMed — All-trans retinoic acid (ATRA) in acute promyelocytic leukemia
- PubMed — Retinoic acid in inflammatory bowel disease
External Authoritative Resources
- NIH Office of Dietary Supplements — Vitamin A Fact Sheet for Health Professionals
- Linus Pauling Institute Micronutrient Information Center — Vitamin A
- Harvard T.H. Chan School — Nutrition Source: Vitamin A
- WHO — Vitamin A deficiency and public health
- PubMed — Vitamin A and immune function research
Connections
- Vitamin A
- Vitamin A and Vision
- Vitamin C
- Vitamin D3
- Vitamin E
- Zinc
- Selenium
- Iron
- Immune Boosting
- Organ Meats
- Macular Degeneration
- Inflammatory Bowel Disease
- Gut-Brain Axis
- Pneumonia
- Leukemia
- Malaria