Vitamin A and Vision Health
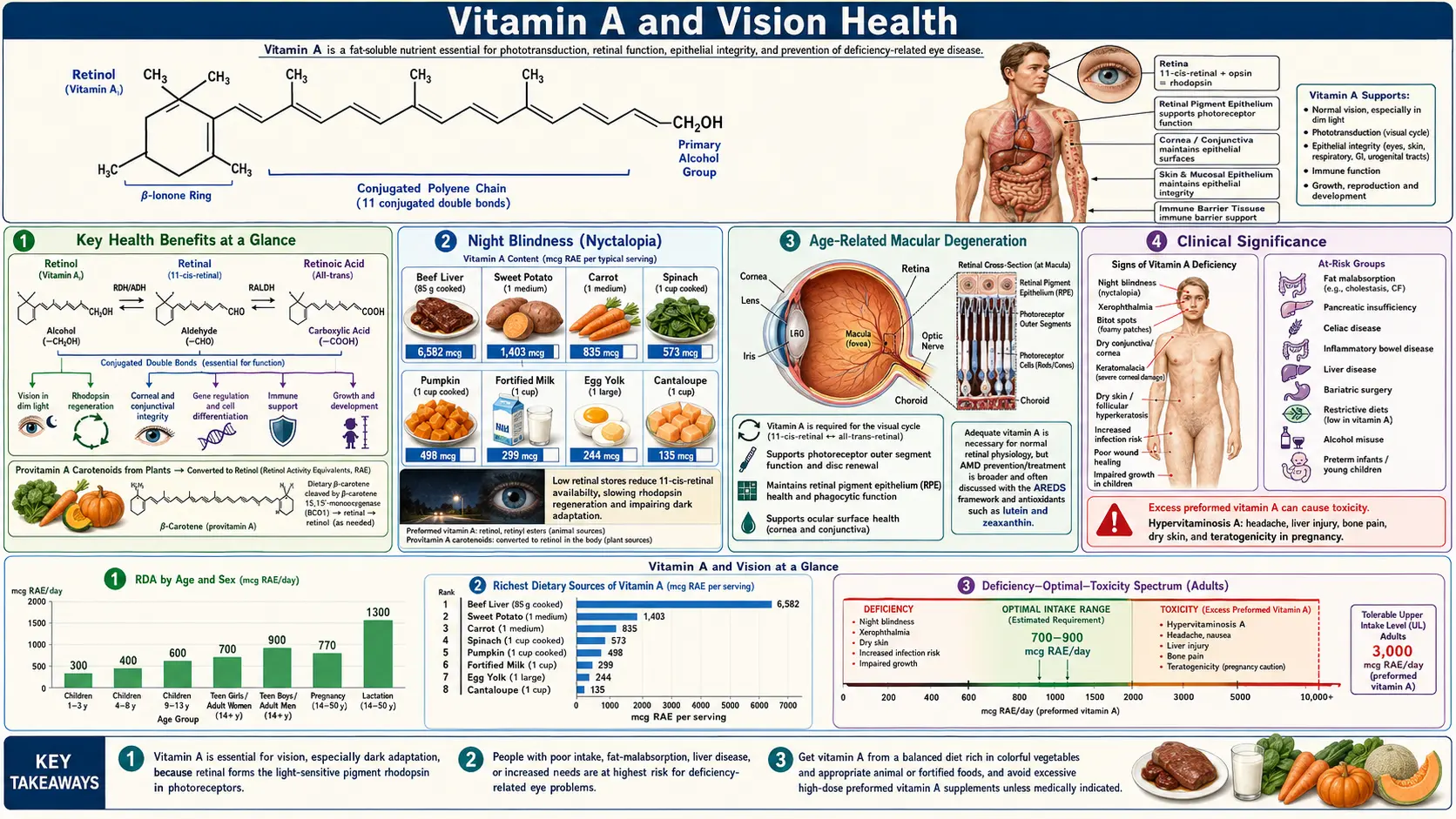
Vitamin A is arguably the single most important micronutrient for maintaining healthy eyesight. Its role in vision was among the earliest nutritional functions ever characterized, and deficiency of this fat-soluble vitamin remains the leading preventable cause of childhood blindness worldwide — affecting an estimated 190 million preschool-age children. From the quantum-level chemistry of 11-cis-retinal in rod photoreceptors, to the macular pigment built from lutein and zeaxanthin, to the retinal pigment epithelium that recycles vitamin A every day, healthy vision depends on a steady supply of this vitamin. Understanding how vitamin A participates in the visual cycle, supports retinal cell integrity, and protects against degenerative eye diseases is essential for both clinicians and patients.
Table of Contents
- Key Health Benefits at a Glance
- Rhodopsin and the Visual Cycle
- Rod and Cone Cell Function
- Night Blindness (Nyctalopia)
- Xerophthalmia and Corneal Damage
- Age-Related Macular Degeneration
- Retinal Health and Maintenance
- Beta-Carotene Conversion
- Clinical Significance
- Research Papers and References
- Connections
- Featured Videos
Key Health Benefits at a Glance
The following is a high-level summary of the evidence-backed vision benefits of adequate vitamin A status. Each is explored in more depth below, and every supporting study is linked in the Research Papers section.
- Powers the visual cycle – 11-cis-retinal, the chromophore of rhodopsin, is derived from vitamin A; without it, rod cells cannot generate the signal cascade that underlies dim-light vision.
- Rescues night blindness – Early-stage nyctalopia from vitamin A deficiency typically reverses within days to weeks of supplementation, a dramatic, measurable clinical response.
- Prevents xerophthalmia and keratomalacia – WHO-classified X1A–X3 stages of corneal and conjunctival disease are directly driven by deficiency; Sommer’s field studies established this as the leading cause of preventable childhood blindness.
- Cuts childhood blindness and mortality – Universal vitamin A supplementation in endemic regions reduces all-cause under-five mortality by ~24% while preventing Bitot’s spots, corneal ulcer, and keratomalacia.
- Supports macular pigment – Lutein and zeaxanthin, xanthophyll relatives of vitamin A, accumulate in the macula, filter blue light, and protect photoreceptors from photo-oxidative stress (AREDS2).
- Slows AMD progression – AREDS and AREDS2 formulations (antioxidants, zinc, lutein/zeaxanthin) cut progression to advanced age-related macular degeneration by ~25% in intermediate-AMD patients.
- Maintains photoreceptor outer segments – Rod and cone disc membranes turn over about 10% per day; vitamin A sustains pigment resynthesis and disc renewal.
- Supports retinal gene expression – Retinoic acid acts via RAR/RXR nuclear receptors to regulate genes for photoreceptor differentiation, RPE health, and retinal development.
- Converts efficiently from plant carotenoids – Beta-carotene yields retinol (12:1 RAE ratio on average), offering a safe non-teratogenic route to vitamin A sufficiency in pregnancy.
Rhodopsin and the Visual Cycle
The ability to see in dim light depends on a light-sensitive pigment called rhodopsin, which is found in the rod cells of the retina. Rhodopsin is composed of a protein called opsin bound to a chromophore derived from vitamin A known as 11-cis-retinal. When a photon of light strikes rhodopsin, 11-cis-retinal undergoes a conformational change to all-trans-retinal, triggering a cascade of biochemical signals that the brain interprets as vision.
After light absorption, all-trans-retinal must be recycled back to 11-cis-retinal through a series of enzymatic reactions collectively known as the visual cycle or retinoid cycle. This process takes place in the retinal pigment epithelium (RPE), a layer of cells directly behind the photoreceptors. Key enzymes in this pathway include retinol dehydrogenase, lecithin retinol acyltransferase (LRAT), and RPE65 isomerohydrolase. Without an adequate supply of vitamin A, regeneration of 11-cis-retinal is impaired, and visual sensitivity progressively declines.
Rod and Cone Cell Function
The human retina contains two classes of photoreceptor cells, each with distinct roles in vision:
- Rod cells – Approximately 120 million rod cells are distributed across the peripheral retina. They are exquisitely sensitive to light and mediate scotopic (low-light) vision. Rods contain rhodopsin and are responsible for detecting motion, shapes, and shades of grey in dim environments. They are the first photoreceptors affected by vitamin A deficiency.
- Cone cells – Roughly 6 million cone cells are concentrated in the macula, particularly the fovea. They require higher light intensity and are responsible for photopic (daylight) vision, color perception, and fine visual acuity. Cones use related visual pigments called photopsins (iodopsins), which also require retinal as a chromophore.
Both rod and cone photoreceptors depend on a continuous supply of vitamin A to maintain their outer segment disc membranes, where visual pigments are densely packed. The outer segments undergo constant renewal, with approximately 10% of the disc membrane being shed and replaced daily.
Night Blindness (Nyctalopia)
Night blindness, or nyctalopia, is typically the earliest clinical manifestation of vitamin A deficiency. Patients report difficulty seeing in dim light, problems with dark adaptation after exposure to bright light, and impaired ability to drive at night. The underlying mechanism is a reduction in rhodopsin concentration in rod cells due to insufficient 11-cis-retinal availability.
Key clinical features of nyctalopia include:
- Delayed dark adaptation – The time required for the eyes to adjust from bright to dim conditions is significantly prolonged, often exceeding 30 minutes.
- Elevated scotopic threshold – Electroretinography (ERG) reveals reduced rod-mediated responses, with elevated thresholds for detecting dim flashes of light.
- Reversibility – In early stages, night blindness responds rapidly to vitamin A supplementation, with measurable improvement in dark adaptation within days to weeks.
- Historical significance – Ancient Egyptian medical papyri described treating night blindness with liver consumption, an approach that unknowingly provided concentrated vitamin A.
Xerophthalmia and Corneal Damage
Xerophthalmia is the clinical spectrum of ocular disease caused by vitamin A deficiency, ranging from mild conjunctival drying to complete corneal destruction. The World Health Organization (WHO) classifies xerophthalmia into progressive stages:
- Conjunctival xerosis (X1A) – The conjunctiva loses its normal moist, glistening appearance and becomes dry, thickened, and wrinkled. Goblet cells, which produce the mucin layer of the tear film, are lost due to squamous metaplasia of the epithelium.
- Bitot’s spots (X1B) – Foamy, triangular, white or silvery plaques appear on the bulbar conjunctiva, typically in the temporal interpalpebral zone. These deposits consist of keratinized epithelial debris and commensal bacteria (often Corynebacterium xerosis). Bitot’s spots are a hallmark clinical sign of vitamin A deficiency.
- Corneal xerosis (X2) – The corneal epithelium becomes dry, hazy, and irregular. Punctate keratopathy may develop, and the corneal surface loses its protective tear film coverage.
- Corneal ulceration and keratomalacia (X3) – In severe deficiency, the cornea undergoes liquefactive necrosis (keratomalacia). This devastating condition can progress rapidly from a small corneal ulcer to full-thickness stromal melting and perforation, often resulting in permanent blindness.
Age-Related Macular Degeneration
Age-related macular degeneration (AMD) is the leading cause of irreversible vision loss in adults over 50 in developed countries. While AMD is a multifactorial disease involving genetic, vascular, and inflammatory components, nutritional status plays a significant role in disease progression.
Vitamin A and its carotenoid precursors have been studied extensively in relation to AMD:
- Lutein and zeaxanthin – These xanthophyll carotenoids (structurally related to vitamin A precursors) accumulate in the macula, where they form macular pigment. They filter damaging blue light and act as antioxidants, protecting photoreceptors and RPE cells from photo-oxidative stress.
- AREDS and AREDS2 studies – The Age-Related Eye Disease Studies demonstrated that supplementation with antioxidant vitamins (including beta-carotene in AREDS, later replaced by lutein and zeaxanthin in AREDS2) reduced the risk of progression to advanced AMD by approximately 25%.
- RPE cell health – The retinal pigment epithelium depends on vitamin A metabolism for phagocytosis of shed photoreceptor outer segments, visual cycle support, and maintenance of Bruch’s membrane. Dysfunction of these processes contributes to drusen formation and RPE atrophy.
Retinal Health and Maintenance
Beyond its direct role in phototransduction, vitamin A supports overall retinal health through several mechanisms:
- Photoreceptor outer segment renewal – Rod and cone outer segments undergo continuous turnover. Vitamin A is required for the synthesis of new disc membranes and the proper folding and insertion of visual pigment molecules.
- Interphotoreceptor retinoid-binding protein (IRBP) – This carrier protein shuttles retinoids between photoreceptors and the RPE through the subretinal space. Adequate vitamin A status ensures efficient IRBP-mediated transport.
- Gene expression regulation – Retinoic acid, derived from vitamin A, acts as a transcription factor ligand via retinoic acid receptors (RARs) and retinoid X receptors (RXRs). These receptors regulate genes involved in retinal development, photoreceptor differentiation, and cellular survival.
- Antioxidant protection – Retinal tissues are highly susceptible to oxidative damage due to constant light exposure and high metabolic activity. Carotenoid forms of vitamin A contribute to the retina’s antioxidant defense system.
Beta-Carotene Conversion
Beta-carotene is the most important provitamin A carotenoid in the human diet. It is converted to retinal by the enzyme beta-carotene 15,15′-dioxygenase (BCMO1/BCO1), primarily in the intestinal mucosa. Several factors influence the efficiency of this conversion:
- Conversion ratio – The current estimate is that 12 micrograms of dietary beta-carotene yields approximately 1 microgram of retinol activity equivalent (RAE). This ratio varies considerably among individuals.
- Genetic polymorphisms – Variants in the BCMO1 gene can reduce conversion efficiency by 30–70%. Individuals carrying these polymorphisms may be poor converters and more dependent on preformed vitamin A sources.
- Dietary fat – Beta-carotene is fat-soluble and requires co-ingestion of dietary fat (at least 3–5 grams per meal) for adequate absorption from the gut.
- Food matrix effects – Beta-carotene from raw vegetables is less bioavailable than from cooked or processed foods, as cooking disrupts plant cell walls and releases carotenoids from their protein-lipid complexes.
Clinical Significance
Vitamin A deficiency remains a major global health problem, affecting an estimated 190 million preschool-age children and 19 million pregnant women worldwide, predominantly in South and Southeast Asia and sub-Saharan Africa. Clinical considerations include:
- WHO supplementation programs – High-dose vitamin A supplementation (200,000 IU for children over 12 months) has been shown to reduce all-cause childhood mortality by approximately 24% in deficient populations, largely through reduction of measles-related and diarrhea-related deaths.
- Pregnancy and teratogenicity – While deficiency poses risks to maternal and fetal health, excessive preformed vitamin A intake (above 10,000 IU/day) during pregnancy is teratogenic, potentially causing craniofacial, cardiac, and neural tube defects. Beta-carotene supplementation does not carry this risk.
- Assessment of status – Serum retinol levels below 0.70 micromol/L indicate deficiency. The modified relative dose response (MRDR) test and retinol isotope dilution assays provide more accurate assessments of hepatic vitamin A stores.
- Therapeutic applications – Beyond deficiency treatment, vitamin A derivatives (retinoids) are used therapeutically in dermatology (isotretinoin for acne, tretinoin for photoaging) and oncology (all-trans retinoic acid for acute promyelocytic leukemia).
Maintaining adequate vitamin A status through a balanced diet containing both preformed vitamin A (liver, dairy, eggs) and provitamin A carotenoids (orange and dark green vegetables, fruits) is fundamental to preserving lifelong vision health. Individuals at risk of deficiency should be identified early, and supplementation programs remain a cornerstone of public health efforts to prevent avoidable blindness.
Research Papers and References
The following are landmark and frequently cited research papers underpinning the claims on this page. Links resolve to the publisher DOI or PubMed record.
Foundational Reviews and Historical Overviews
- Sommer A. Vitamin A deficiency and clinical disease: an historical overview. J Nutr. 2008;138(10):1835-1839.
- Stephensen CB. Vitamin A, infection, and immune function. Annu Rev Nutr. 2001;21:167-192.
Visual Cycle and Rhodopsin Chemistry
- PubMed — RPE65 and the retinoid visual cycle
- PubMed — Rhodopsin, 11-cis-retinal and phototransduction
Xerophthalmia, Childhood Blindness, and Supplementation
- PubMed — Xerophthalmia WHO classification and field studies
- PubMed — Vitamin A supplementation and child mortality meta-analyses
- PubMed — Vitamin A and measles mortality RCTs
Age-Related Macular Degeneration (AREDS / AREDS2)
Beta-Carotene Conversion and Genetics
External Authoritative Resources
- NIH Office of Dietary Supplements — Vitamin A Fact Sheet for Health Professionals
- Linus Pauling Institute Micronutrient Information Center — Vitamin A
- Harvard T.H. Chan School — Nutrition Source: Vitamin A
- WHO — Micronutrient deficiencies / xerophthalmia
- PubMed — Vitamin A and vision research
Connections
- Vitamin A
- Vitamin A and Immune Function
- Macular Degeneration
- Cataracts
- Glaucoma
- Vitamin C
- Vitamin E
- Vitamin D3
- Zinc
- Selenium
- Organ Meats
- Eggs
- Oxidative Stress
- Acne