Sulfites in Food and Wine: Allergic Reactions and Hidden Dangers
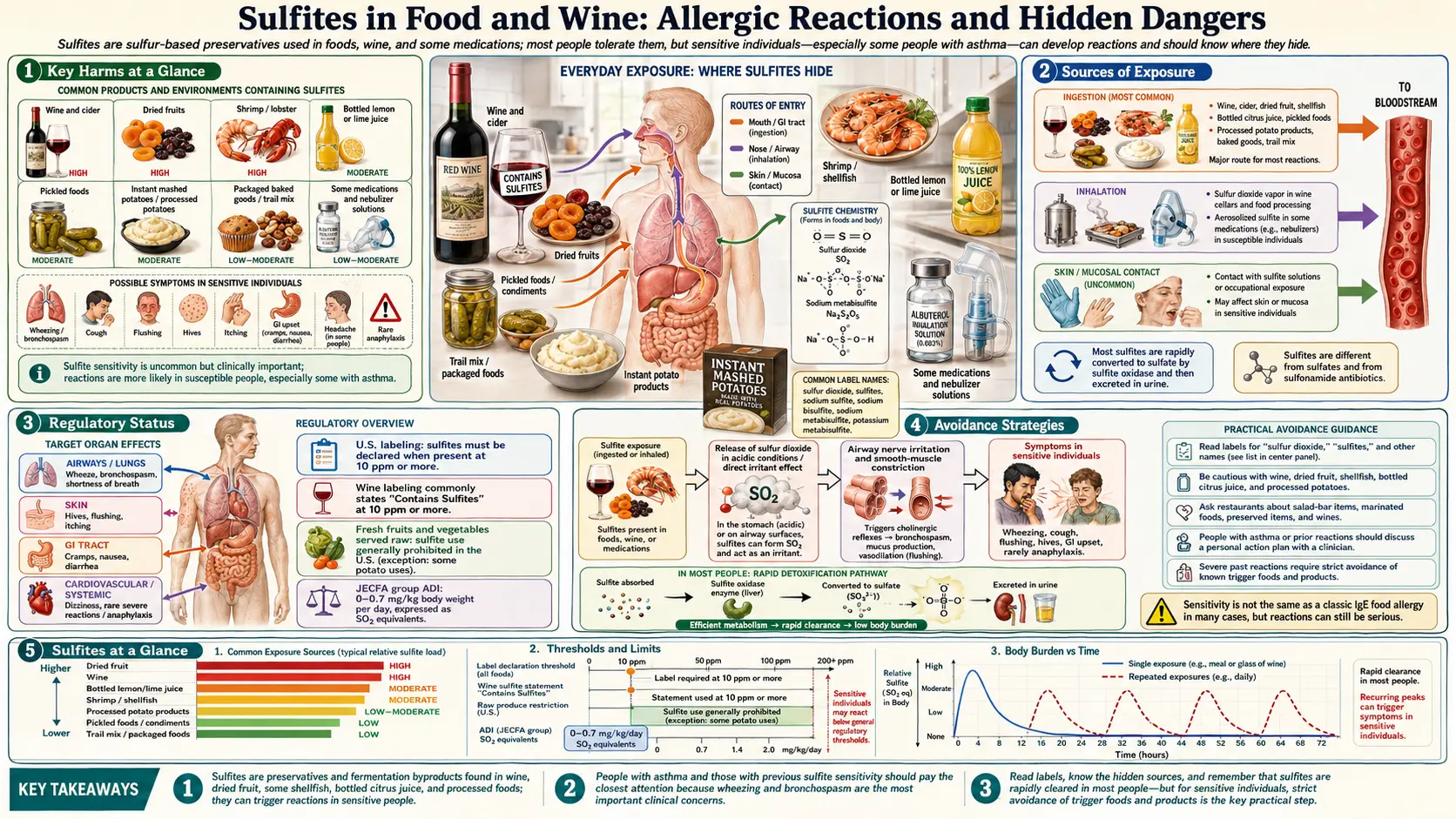
Sulfites are among the oldest food preservation compounds in human history, used in winemaking since antiquity, yet they remain one of the most medically significant food additives in the modern diet. For the majority of people they pose no risk, but for the estimated one percent of the general population — and five to thirteen percent of asthmatics — sulfite exposure can trigger reactions ranging from mild gastrointestinal distress to life-threatening anaphylaxis. Thirteen deaths linked to sulfited raw vegetables in the 1980s prompted the first major U.S. ban on a food additive used at restaurant salad bars, yet sulfites persist in hundreds of food products, wines, and medications with only partial labeling requirements.
Table of Contents
- Key Harms at a Glance
- Overview
- How They Work
- Sources of Exposure
- Health Effects
- The 1986 FDA Ban
- Regulatory Status
- Wine and Sulfites
- Testing and Diagnosis
- Avoidance Strategies
- Research Papers and References
- Connections
- Featured Videos
Key Harms at a Glance
- Asthma attacks — trigger bronchospasm in 5–13% of asthmatics, sometimes fatally.
- Anaphylaxis — life-threatening reactions documented in sensitive individuals.
- Thirteen deaths in the 1980s — linked to sulfited raw salad-bar vegetables.
- Thiamine destruction — sulfite cleaves vitamin B1 in foods.
- Headache and flushing — common reactions from sulfited wine.
- Gastrointestinal distress — nausea, diarrhea, abdominal pain in sensitive consumers.
- Hidden in wine, dried fruits, shrimp, potatoes — labeling above 10 ppm required in the US.
1. Overview
Sulfites are a family of sulfur-containing compounds that release sulfur dioxide (SO2) in aqueous solution and in food systems. The term "sulfites" encompasses several distinct chemical entities, all regulated together because they share a common mechanism of action and produce equivalent biological effects at equivalent concentrations of free SO2:
- Sulfur dioxide (SO2) — the parent compound; a pungent, colorless gas used directly in winemaking and dried fruit processing
- Sodium sulfite (Na2SO3) — E221 — used in food processing and as a photographic developer
- Sodium bisulfite (NaHSO3) — E222 — more acidic than sodium sulfite; widely used in winemaking and food preservation
- Sodium metabisulfite (Na2S2O5) — E223 — the most commonly used solid sulfite in the food industry; releases SO2 upon contact with moisture
- Potassium metabisulfite (K2S2O5) — E224 — the preferred form in winemaking (Campden tablets) as it avoids adding sodium
- Potassium bisulfite (KHSO3) — E228 — used in wine and fermented beverages
All of these compounds act by releasing free SO2 in solution, and their preservative and sensitizing properties are expressed in terms of equivalent SO2 concentration, typically reported in parts per million (ppm) or milligrams per liter (mg/L).
The history of sulfite use in food preservation is ancient. Roman winemakers burned sulfur candles inside clay amphorae before filling them with wine, a practice documented by Pliny the Elder and Cato. The SO2 produced killed spoilage organisms, scavenged oxygen, and prevented the wine from turning to vinegar. This empirical technique persisted for two millennia before the chemistry was understood. By the nineteenth century, systematic addition of sulfite compounds to wine and food products became standard practice across Europe and subsequently worldwide.
Sulfites function simultaneously as antioxidants, antimicrobial agents, and enzymatic browning inhibitors — a combination of properties unmatched by most other single additives, which explains their continued widespread use despite known health concerns for sensitive individuals.
2. How They Work
Inhibition of Enzymatic Browning
Cut fruits and vegetables undergo rapid browning when the enzyme polyphenol oxidase (PPO) — released from damaged cells — catalyzes the oxidation of phenolic compounds to quinones, which then polymerize to form brown melanin pigments. This browning is purely enzymatic and does not reflect spoilage, but it renders fresh-cut produce commercially unacceptable within minutes to hours of cutting.
Sulfites prevent enzymatic browning by multiple mechanisms: direct inhibition of PPO through interaction with the copper-containing active site of the enzyme, reduction of the quinone intermediates back to colorless phenols before they can polymerize, and consumption of dissolved oxygen that would otherwise drive the oxidation reaction. Even very low concentrations of free SO2 (10–50 ppm) are sufficient to completely inhibit browning for many hours, which made sulfites the preservative of choice for salad bars, fresh-cut fruit displays, and restaurant produce preparation through the early 1980s.
Antimicrobial Activity
The antimicrobial effectiveness of sulfites depends critically on pH. In acidic environments (pH below 4), the predominant species is molecular SO2 (also called "free molecular SO2"), which readily crosses microbial cell membranes and inhibits key metabolic enzymes including alcohol dehydrogenase, pyruvate dehydrogenase, and various oxidoreductases. At higher pH values, SO2 dissociates to bisulfite and sulfite ions, which are less membrane-permeable and considerably less antimicrobial.
This pH dependence is why winemakers carefully monitor the pH of their wine: a wine at pH 3.2 requires far less total SO2 to achieve the same antimicrobial protection as a wine at pH 3.8. The microorganisms most sensitive to sulfites include wild yeasts, acetic acid bacteria (Acetobacter spp.), and lactic acid bacteria, making sulfites particularly valuable for preventing stuck fermentations and post-bottling spoilage.
Antioxidant Protection
Sulfites react directly with dissolved oxygen and with oxidized wine components, protecting color, flavor, and aroma compounds from oxidative degradation. Free SO2 binds reversibly to carbonyl compounds (particularly acetaldehyde produced during fermentation and oxidation), forming stable sulfonated adducts that neutralize the harsh, vinegary character that acetaldehyde imparts. This "bound SO2" is less antimicrobially active than free SO2 but still contributes to total sulfite content measurements.
Bleaching Action
Sulfites bleach naturally occurring pigments in foods by reducing conjugated double bonds in chromophoric molecules. This bleaching is exploited commercially in the production of maraschino cherries (natural cherry pigments are bleached out before artificial red dye is added), light-colored dried fruits, and certain seafood products where natural pigmentation is considered undesirable by the market. The same bleaching reaction can destroy carotenoids, anthocyanins, and other beneficial phytopigments in foods treated at high sulfite concentrations.
3. Sources of Exposure
Wine
Wine is the single most prominent dietary source of sulfite exposure for adults who consume alcohol. Total SO2 concentrations in wine typically range from 10 ppm in some red wines to 350 ppm in sweet white wines, though regulatory limits cap this in most jurisdictions. A standard 150 mL glass of wine containing 200 ppm SO2 delivers approximately 30 mg of SO2 — a potentially significant dose for sensitive individuals. All commercially produced wine contains some sulfites, even those labeled "organic" or "natural," because fermentation itself produces small amounts of SO2 as a metabolic byproduct of yeast.
Dried Fruits
Dried fruits contain the highest sulfite concentrations of any food category, often exceeding levels found in wine by a factor of ten. Sulfited dried apricots may contain 500–2000 ppm SO2, compared with the orange to brown color of unsulfured dried apricots. Sulfited apricots retain their bright orange color because the SO2 prevents carotenoid degradation and enzymatic browning. Other commonly sulfited dried fruits include golden raisins (sulfited; regular dark raisins typically are not), dried peaches, prunes, and dried figs. The high concentration combined with the small serving size means that even a small handful of dried apricots can deliver a sulfite dose comparable to several glasses of wine.
Seafood
Shrimp and other crustaceans are treated with sodium bisulfite or sodium metabisulfite immediately after harvest to prevent melanosis — the enzymatic blackening of the shell and flesh that begins within hours of catch and renders the product commercially unsaleable. Fresh, frozen, and processed shrimp products routinely contain 10–100 ppm residual sulfites. Lobster, crab, and crayfish products may be similarly treated. Fish and non-crustacean shellfish are generally not sulfited.
Potato Products
The potato processing industry relies heavily on sulfites to prevent browning of cut, peeled, and processed potatoes. French fries and hash browns from fast food restaurants, frozen potato products, dehydrated potato flakes and granules (instant mashed potatoes), and peeled raw potatoes sold to food service establishments all commonly contain sulfites. Concentrations in dehydrated potato products can reach 250–500 ppm. This source was central to the 1986 FDA salad bar ban, as raw sliced potatoes held at salad bars were among the highest-sulfite items served.
Beer and Fermented Beverages
Beer contains naturally occurring sulfites from yeast fermentation, typically 10–40 ppm in finished product. Some brewers add sulfites as preservatives or use sulfite-treated malt. Cider, mead, and other fermented beverages may be sulfited during production. Because U.S. labeling requirements for alcoholic beverages above 10 ppm apply regardless of beverage type, most beer and cider labels now carry "contains sulfites" declarations.
Condiments and Sauces
Commercially produced grape juice, wine vinegar, cider vinegar, and balsamic vinegar contain sulfites from the underlying grape or fruit fermentation. Many condiments including pickles, relishes, and prepared salads contain sulfites as preservatives. Lemon juice concentrates and other acidic condiments may be sulfited to prevent browning and extend shelf life.
Baked Goods and Dough
Sodium metabisulfite is used as a dough conditioner in commercial baking: it reduces disulfide bonds in gluten proteins, relaxing dough and making it easier to machine-process. Crackers, cookies, pie crusts, pizza dough, and some breads produced commercially may contain sulfites from this application. Residual levels in finished baked goods are typically low (below 10 ppm) because SO2 is volatile and dissipates during baking, but some residue remains.
Maraschino Cherries and Preserved Fruits
Maraschino cherries undergo an intensive sulfite-bleaching process: fresh cherries are submerged in a high-concentration sulfite solution (sulfur dioxide or sodium bisulfite) that completely bleaches the natural cherry pigments, creating a nearly white substrate that is then re-colored with artificial red dye (typically FD&C Red No. 40), sweetened, and flavored. The finished product may contain residual sulfites along with the extensive other additives used in the process.
Medications
Sulfites are used as antioxidants and preservatives in numerous pharmaceutical preparations, including injectable medications (epinephrine, dexamethasone, dopamine, some local anesthetics), nebulized bronchodilators, and some oral preparations. The medical significance is considerable: a sulfite-sensitive asthmatic receiving emergency treatment with a sulfite-containing epinephrine injection may experience an exacerbated bronchospasm from the sulfite preservative in the medication intended to treat them. Sulfite-free formulations exist for most critical medications and should be specified for known sulfite-sensitive patients.
Molasses and Corn Syrup
Sulfur dioxide is used in the processing of both cane sugar molasses and some corn syrups as a processing aid. Residual sulfites in molasses can be substantial (up to several hundred ppm), and molasses is a common ingredient in dark breads, baked beans, gingerbread, and other foods where its flavor is valued. Consumers who are sulfite-sensitive and consume molasses-containing products may not associate their reactions with sulfite exposure because molasses is not intuitively recognized as a sulfite source.
4. Health Effects
Sulfite Sensitivity and Intolerance
Sulfite sensitivity (also called sulfite intolerance or sulfite hypersensitivity) is estimated to affect approximately one percent of the general population, but prevalence is dramatically higher among asthmatic populations: studies have reported rates of five to thirteen percent of asthmatic individuals experiencing adverse reactions to sulfite exposure at doses commonly encountered in food and wine. The condition is not a true food allergy in the IgE-mediated sense for most affected individuals, though IgE-mediated mechanisms do exist in a subset.
Respiratory Symptoms
The most clinically significant and potentially life-threatening manifestation of sulfite sensitivity is bronchoconstriction. Symptoms include:
- Bronchospasm — sudden airway narrowing that can be severe and rapid in onset
- Wheezing and stridor — audible high-pitched breathing due to narrowed airways
- Chest tightness and dyspnea — the sensation of inability to breathe adequately
- Cough — often persistent and unresponsive to usual antitussive measures
- Rhinitis and nasal congestion — common in milder reactions
Respiratory reactions typically begin within minutes to two hours of sulfite ingestion and may require bronchodilator treatment or, in severe cases, emergency medical intervention. Pre-existing asthma is the strongest risk factor for severe respiratory sulfite reactions.
Cutaneous and Systemic Reactions
Dermatological manifestations of sulfite sensitivity include urticaria (hives), angioedema (deep tissue swelling, potentially affecting the throat and airway), flushing, and pruritus. These reactions may occur independently of or in combination with respiratory symptoms. Gastrointestinal symptoms — nausea, abdominal cramping, diarrhea — are also reported and may be the primary manifestation in non-asthmatic sensitive individuals.
Anaphylaxis
Anaphylaxis — the severe, multi-system allergic reaction involving hemodynamic compromise — has been documented in response to sulfite ingestion, though it is uncommon. Deaths attributed to sulfite-induced anaphylaxis have been reported in the medical literature and in FDA adverse event reports. The mechanism in confirmed anaphylactic cases is likely IgE-mediated, involving immune sensitization to sulfite-protein conjugates rather than a pure pharmacological irritant effect.
Headaches and Migraines
A well-recognized but poorly understood association exists between wine consumption and migraine headaches. While many compounds in wine (histamine, tyramine, tannins, alcohol itself) have been implicated, sulfites are frequently cited by patients who report reactions specifically to white or sweet wines — which typically contain higher added sulfite levels than red wines. Prospective studies on sulfite-specific headache induction are limited, but the clinical observation is sufficiently common to be addressed in most sulfite sensitivity literature.
Mechanism: Sulfite Oxidase Deficiency
A central biochemical mechanism underlying sulfite sensitivity involves sulfite oxidase, a molybdenum-containing mitochondrial enzyme that converts sulfite to the non-toxic sulfate, enabling urinary excretion. Individuals with reduced sulfite oxidase activity — whether due to genetic polymorphisms, molybdenum deficiency, or other factors — cannot adequately detoxify dietary sulfite loads and are consequently more vulnerable to accumulation of free sulfite in tissues.
In the airway, inhaled or aspirated sulfite in acidic gastric reflux can form sulfurous acid and SO2 gas directly at bronchial epithelial surfaces, triggering bronchoconstriction through direct irritant effects, stimulation of afferent nerve endings (via transient receptor potential channels), and possibly through prostaglandin-mediated pathways independent of IgE. This non-IgE mechanism explains why sulfite reactions in asthmatics do not always fit the classic pattern of IgE-mediated food allergy.
IgE-Mediated vs. Non-IgE Mechanisms
Two distinct pathophysiological pathways account for sulfite reactions:
- IgE-mediated (true allergic) reactions — sulfite or sulfite-modified proteins bind to IgE antibodies on mast cells and basophils, triggering histamine and mediator release. These reactions can cause true anaphylaxis and typically respond to epinephrine and antihistamines.
- Non-IgE pharmacological reactions — direct bronchospasm from inhaled SO2 gas formed in the acidic stomach, prostaglandin-mediated airway inflammation, or neurogenic mechanisms. These reactions are particularly common in aspirin-sensitive asthmatics and may involve the same COX-dependent pathway responsible for aspirin-induced bronchospasm.
5. The 1986 FDA Ban
The Salad Bar Epidemic
Through the early 1980s, restaurant salad bars across the United States routinely used sulfite sprays to maintain the appearance of fresh-cut fruits and vegetables. High-sulfite solutions were applied directly to lettuce, tomatoes, avocado, mushrooms, potatoes, and other produce items to prevent browning and extend the time produce could remain visually appealing on display. Concentrations applied to raw produce were often far higher than in other sulfite-treated foods because there was no regulatory framework specifically governing this use.
Between 1983 and 1985, the FDA received reports of 13 deaths and over 500 adverse reactions attributed to sulfites on raw fruits and vegetables in restaurant and grocery settings. The victims were predominantly asthmatic individuals who consumed sulfite-treated salad bar items without any warning of additive presence. These cases represented the first clear epidemiological signal of severe dietary sulfite toxicity in the modern food supply.
FDA Action and the 1986 Rule
In response to the mounting adverse event reports, the FDA took formal regulatory action in 1986 — a process that was unusually swift by regulatory standards, reflecting the severity of the documented harm. The agency:
- Revoked the GRAS (Generally Recognized As Safe) status of sulfites on fresh fruits and vegetables intended to be served raw to consumers
- Banned the use of sulfites on fresh produce sold in grocery stores and served at restaurant salad bars — the first major federal prohibition of a food additive that had previously been freely used
- Established mandatory labeling requirements for all packaged foods containing more than 10 ppm sulfite compounds, requiring disclosure on the ingredient label
- Extended labeling requirements to restaurant menus and wine lists through subsequent rulemaking
Significance and Legacy
The 1986 sulfite ban was a landmark moment in U.S. food safety regulation, demonstrating that the FDA would act to prohibit a traditional food additive when sufficient evidence of consumer harm accumulated. It established the principle that GRAS status is not permanent and can be revoked based on post-market safety data. The 10 ppm labeling threshold — rather than a complete prohibition — reflected a policy judgment that low-level sulfite exposure in packaged foods posed a manageable risk when properly labeled, while high-level exposure on unlabeled fresh produce in institutional settings was unacceptable.
The ban did not affect sulfites in wine, which were considered a separate category governed by different regulatory authorities (the Bureau of Alcohol, Tobacco, and Firearms, now TTB, rather than the FDA). The wine exemption has been a source of ongoing controversy, as wine delivers some of the highest per-serving sulfite doses of any commonly consumed food or beverage.
6. Regulatory Status
United States — FDA Requirements
The FDA requires that sulfite compounds be declared on the ingredient label of any packaged food containing 10 ppm or more of total sulfites. This threshold was set at a level intended to be protective for sulfite-sensitive individuals while acknowledging that trace sulfite contamination below 10 ppm is difficult to avoid in the food supply and is unlikely to cause reactions at such low doses. Acceptable declarations include the specific compound used (e.g., "sodium bisulfite," "sulfur dioxide") or the collective term "sulfites."
Foods containing below 10 ppm sulfites are not required to declare them, creating a potential concern for highly sensitive individuals who may react to even very low doses. There is no requirement to declare sulfites used as processing aids (as opposed to functional preservatives) if residual levels in the finished food fall below 10 ppm.
Alcoholic Beverages
Wine and beer in the United States are regulated by the Alcohol and Tobacco Tax and Trade Bureau (TTB) rather than the FDA. TTB requires that any wine or beer containing 10 ppm or more sulfite compounds bear the statement "contains sulfites" on the label. This requirement — which took effect in 1988 — applies to all wines sold in the United States, including imports. The U.S. was among the first countries to mandate sulfite labeling on wine labels, a requirement that has since been adopted by the EU and many other jurisdictions.
European Union — E-Number Labeling
The EU regulates sulfites as food additives under Regulation (EC) No 1333/2008, assigning them E-numbers E220 through E228. EU food labeling law requires declaration of any sulfite compound that contributes 10 mg/kg (10 ppm) or more SO2 equivalent in the finished product. The declaration must use either the E-number or the descriptive name (e.g., "sulphur dioxide," "sodium metabisulphite") followed by the allergen indicator "sulphites" or "sulfites" in bold type, because sulfites are classified as a major food allergen under EU law.
Codex Alimentarius — ADI and Permitted Levels
The Joint FAO/WHO Expert Committee on Food Additives (JECFA) has evaluated sulfites multiple times and established an Acceptable Daily Intake (ADI) of 0–0.7 mg/kg body weight per day expressed as SO2. For a 70 kg adult, this corresponds to a maximum daily intake of 49 mg SO2. Analysis of actual dietary exposures suggests that heavy consumers of wine, dried fruits, and processed foods can exceed this ADI on a regular basis. The Codex general standard for food additives specifies permitted maximum levels for sulfites in numerous food categories ranging from 10 ppm in some products to 2000 ppm in certain dried fruits.
Wine Exemption Controversy
Critics of the current regulatory framework point out an inherent inconsistency: sulfites are banned from fresh vegetables because of documented fatalities, yet wine — which can contain ten times the sulfite concentration of a treated salad bar item — is exempt from similar restrictions and carries only a label warning. The exemption reflects both the historical and cultural centrality of wine in many societies and the technical reality that SO2 is difficult to replace in winemaking without significant quality compromise. Organic winemaking regulations represent one area where stricter sulfite limits are imposed voluntarily by producers.
7. Wine and Sulfites
Natural vs. Added Sulfites
A fundamental point often misunderstood by consumers is that all wine contains some sulfites, regardless of whether any have been added. Yeast — the microorganism responsible for alcoholic fermentation — produces SO2 as a metabolic byproduct during fermentation, typically generating 5–20 ppm in the finished wine. This naturally occurring sulfite cannot be removed without affecting wine quality. Therefore, the claim that a wine is "sulfite-free" is technically inaccurate; the most accurate term is "no added sulfites" or "no sulfites added."
Why Sulfites Are Added
Winemakers add SO2 at multiple stages of production for several reasons: as a sanitizing agent for equipment and barrels, to inhibit wild yeast and bacteria at harvest, to protect must and juice from oxidation between crushing and fermentation, to halt fermentation at a desired residual sugar level (in sweet wine production), and to protect finished wine during bottling and storage. Without added sulfites, wines have dramatically shorter shelf lives and are far more vulnerable to spoilage and oxidation — two glasses in the same bottle can taste entirely different if opened days apart without SO2 protection.
Sulfite Levels by Wine Type
Sulfite concentrations vary considerably by wine type, reflecting different production requirements:
- Dry red wines — typically 50–150 ppm total SO2; the lower pH and higher tannin content of many reds means less SO2 is needed for equivalent antimicrobial protection
- Dry white wines — typically 100–200 ppm; more vulnerable to oxidation than reds due to lower tannin content, requiring higher SO2 levels
- Sweet wines and dessert wines — often 200–350 ppm; the high residual sugar creates risk of refermentation in the bottle, requiring elevated SO2 to suppress yeast activity
- Sparkling wines — variable but typically 50–200 ppm; CO2 provides some protective effect, partially substituting for SO2
Organic Wine Regulations
In the United States, the USDA National Organic Program distinguishes between two categories: wines labeled "organic wine" cannot contain any added sulfites (though naturally occurring sulfites up to 100 ppm are acceptable), while wines labeled "made with organic grapes" may contain added sulfites up to 100 ppm total. The stricter "organic wine" category is rarely used commercially in the U.S. because the prohibition on added sulfites severely limits shelf life and shelf stability.
In the European Union, organic wine regulations are more permissive: added sulfites are permitted in organic wines but at lower maximum levels than in conventional wine — 100 ppm for organic red wines and 150 ppm for organic whites and rosés, compared with 150 and 200 ppm respectively for conventional wines under normal EU limits.
The "Red Wine Headache" and Sulfites
Red wine headaches are commonly attributed to sulfites, but the evidence for this specific attribution is weak. Red wines typically contain lower total SO2 than white wines, yet red wine headaches are reported more frequently than white wine headaches in the general population. Compounds more plausibly responsible include histamine and other biogenic amines, tannins, tyramine, and possibly a recently identified compound quercetin glucuronide, which some individuals metabolize to a form that inhibits aldehyde dehydrogenase, preventing normal acetaldehyde clearance. True sulfite-triggered reactions in white or sweet wine consumers are likely explained by the higher SO2 content of those wine types rather than by red wine specifically.
8. Testing and Diagnosis
Oral Sulfite Challenge
The definitive diagnostic test for sulfite sensitivity is a supervised oral challenge conducted in a clinical setting with emergency equipment available. The patient ingests capsules containing measured amounts of sodium metabisulfite (typically beginning at 1–5 mg SO2 equivalent and increasing to 25–200 mg) under observation. Pulmonary function (FEV1) is measured before and after each dose. A positive challenge is defined as a clinically significant fall in FEV1 (typically 20% or greater) or the development of clinical symptoms requiring treatment. Double-blind placebo-controlled challenge protocols are used in research settings.
Challenge testing carries inherent risk of severe bronchospasm and should only be performed where bronchodilator therapy and oxygen are immediately available. Pre-challenge spirometry must confirm adequate baseline lung function to proceed safely.
Elimination and Reintroduction Diet
A practical approach in non-emergency clinical settings is a structured sulfite elimination diet followed by systematic reintroduction. The patient eliminates all known high-sulfite foods (wine, dried fruits, shrimp, commercial condiments, processed potatoes) for two to four weeks while documenting symptom frequency and severity. If symptoms improve on elimination, systematic reintroduction of individual food categories can identify specific triggers and threshold doses.
Sulfite Test Strips
Commercial test strips are available that can detect sulfite concentrations in foods and wines through a colorimetric reaction. These strips typically have detection ranges of 10–500 ppm and can be used by consumers to screen foods before consumption. Accuracy is sufficient for gross screening but not for precise quantitative measurement; laboratory methods (ion chromatography, Monier-Williams distillation) are required for definitive quantification. Test strips are available in home brewing supply stores and from online retailers.
Skin Testing and IgE Measurement
Skin prick testing with sulfite solutions and serum-specific IgE measurement to sulfite-protein conjugates have been used in research settings to identify the IgE-mediated subset of sulfite-sensitive patients. These tests are not standardized and not commercially available as routine clinical tests in most centers. A positive result would support an IgE-mediated mechanism and guide management toward strict avoidance and epinephrine prescription, similar to classic food allergy management.
9. Avoidance Strategies
Label Reading
Reading ingredient labels is the most important avoidance strategy for sulfite-sensitive individuals. In the United States, any food containing 10 ppm or more sulfite compounds must list them by name in the ingredient declaration. Look for: sulfur dioxide, sodium sulfite, sodium bisulfite, sodium metabisulfite, potassium bisulfite, potassium metabisulfite. Be aware that some labeling uses the term "sulfites" generically. Wines in the U.S. must carry the statement "contains sulfites" if above the 10 ppm threshold.
Choosing Organic and Unsulfured Products
For individuals who consume wine, choosing wines labeled "no sulfites added" or wines from producers committed to minimal-intervention winemaking reduces but does not eliminate sulfite exposure (naturally occurring sulfites from fermentation will remain). For dried fruits, seek out products specifically labeled "unsulfured" or "sulfite-free" — unsulfured dried apricots will be brown rather than orange, a reliable visual indicator. Unsulfured versions of most common dried fruits are available in health food stores and online.
Prioritize Fresh Foods
Whole, fresh, unprocessed foods have no added sulfites. A diet centered on fresh vegetables, fresh fruit, unprocessed meats, plain dairy, and home-cooked meals using whole ingredients dramatically reduces sulfite exposure compared to a diet heavy in processed, packaged, and restaurant foods. Fresh potatoes — unlike commercial frozen or dehydrated potato products — are not sulfited. Fresh shrimp from a trusted fishmonger may have been treated at the vessel, but concentrations in fresh (not processed) shrimp are generally lower than in frozen or previously treated products.
Restaurant Communication
Individuals with known sulfite sensitivity should communicate their sensitivity to restaurant staff, specifically asking about the use of sulfite solutions on produce, sulfited pre-cut vegetables, and sulfite-treated shrimp. Since the 1986 ban, raw produce at restaurant salad bars should not be treated with sulfites, but practices vary and the ban applies only to raw produce intended to be served without further preparation — other sulfite uses in restaurant kitchens are not prohibited.
Medical Alert and Epinephrine
Individuals with a history of severe sulfite reactions — particularly those involving anaphylaxis, severe bronchospasm, or angioedema — should carry a prescription epinephrine auto-injector and wear medical identification indicating sulfite sensitivity. Healthcare providers should document sulfite sensitivity prominently in medical records and ensure that sulfite-containing medications are avoided where alternatives exist, particularly in injectable and inhaled medications.
10. Research Papers and References
Key research papers and authoritative sources. Links resolve to DOI, PubMed, or the issuing agency.
- Simon RA. Sulfite sensitivity. Annals of Allergy. 1986;56(4):281–288.
- Vally H, Misso NL. Adverse reactions to the sulphite additives. Gastroenterology and Hepatology from Bed to Bench. 2012;5(1):16–23.
- Lester MR. Sulfite sensitivity: significance in human health. Journal of the American College of Nutrition. 1995;14(3):229–232.
- Vally H, Carr A, El-Saleh J, Thompson PJ. Wine-induced asthma: a placebo-controlled assessment of its pathogenesis. Journal of Allergy and Clinical Immunology. 1999;103(1):41–46.
- Gunnison AF, Jacobsen DW. Sulfite hypersensitivity: a critical review. Critical Reviews in Toxicology. 1987;17(3):185–214.
- Taylor SL, Bush RK, Selner JC, et al. Sensitivity to sulfited foods among sulfite-sensitive subjects with asthma. Journal of Allergy and Clinical Immunology. 1988;81(6):1159–1167.
- U.S. Food and Drug Administration. Sulfites: Action on Use of Sulfites as Preservatives on Fresh Fruits and Vegetables Intended to be Served or Sold Unpackaged and Unlabeled; Final Rule. 21 CFR Part 182, 51 Federal Register 25012. FDA; 1986.
- U.S. Food and Drug Administration. Sulfites in Food: 21 CFR 101.100 and 182.3616 — Sodium bisulfite. FDA; 2023.
- Asero R, Bottazzi G. Hypersensitivity to aspirin and sulfite: is there a link? Allergy and Asthma Proceedings. 2007;28(6):652–655.
- Codex Alimentarius Commission. General Standard for Food Additives (GSFA): Sulfur Dioxide and Sulfites. Codex Stan 192-1995 (Rev. 2023). FAO/WHO; 2023.
- European Food Safety Authority. Scientific Opinion on the re-evaluation of sulfur dioxide (E 220), sodium sulfite (E 221), sodium bisulfite (E 222), sodium metabisulfite (E 223), potassium metabisulfite (E 224), calcium sulfite (E 226), calcium bisulfite (E 227) and potassium bisulfite (E 228) as food additives. EFSA Journal. 2016;14(4):4438.
- Alcohol and Tobacco Tax and Trade Bureau. Sulfite Labeling Requirements for Wine. TTB Ruling 88-2. Washington: TTB; 1988.
- Bahna SL. Food additive hypersensitivities: a cause or a coincidence of childhood allergies? Annals of Allergy, Asthma and Immunology. 2004;93(Suppl 3):S30–35.
- Jacobsen DW, Simon RA, Singh M. Sulfite oxidase deficiency and cobalamin protection in sulfite-sensitive asthmatics. Journal of Allergy and Clinical Immunology. 1984;73(1):135.
- Waterhouse AL. Wine phenolics. Annals of the New York Academy of Sciences. 2002;957:21–36.
- Ramos E, Cabral JM, Fonseca L, et al. Adverse reactions to sulfites in food and drugs. Allergy. 1994;49(7):600–601.
- Walker R. Toxicology of sorbic acid and sorbates. Food and Chemical Toxicology. 1990;28(5):357–372. [Reference context: sulfite comparative risk assessment]
- PubMed — sulfite sensitivity asthma search
Connections
- Food Additives
- Preservatives
- Asthma
- Vitamin B1
- Migraine
- Sodium Benzoate
- Sodium Nitrite
- Potassium Sorbate
- Sulfur
- Potassium
- Molybdenum
- Allergies
- Aspirin
- Elimination Diet
- Headache
- BHT
- Food Intolerance
- Triggers and Flare Management