Sodium Nitrite and Sodium Nitrate: Carcinogenic Preservatives in Processed Meat
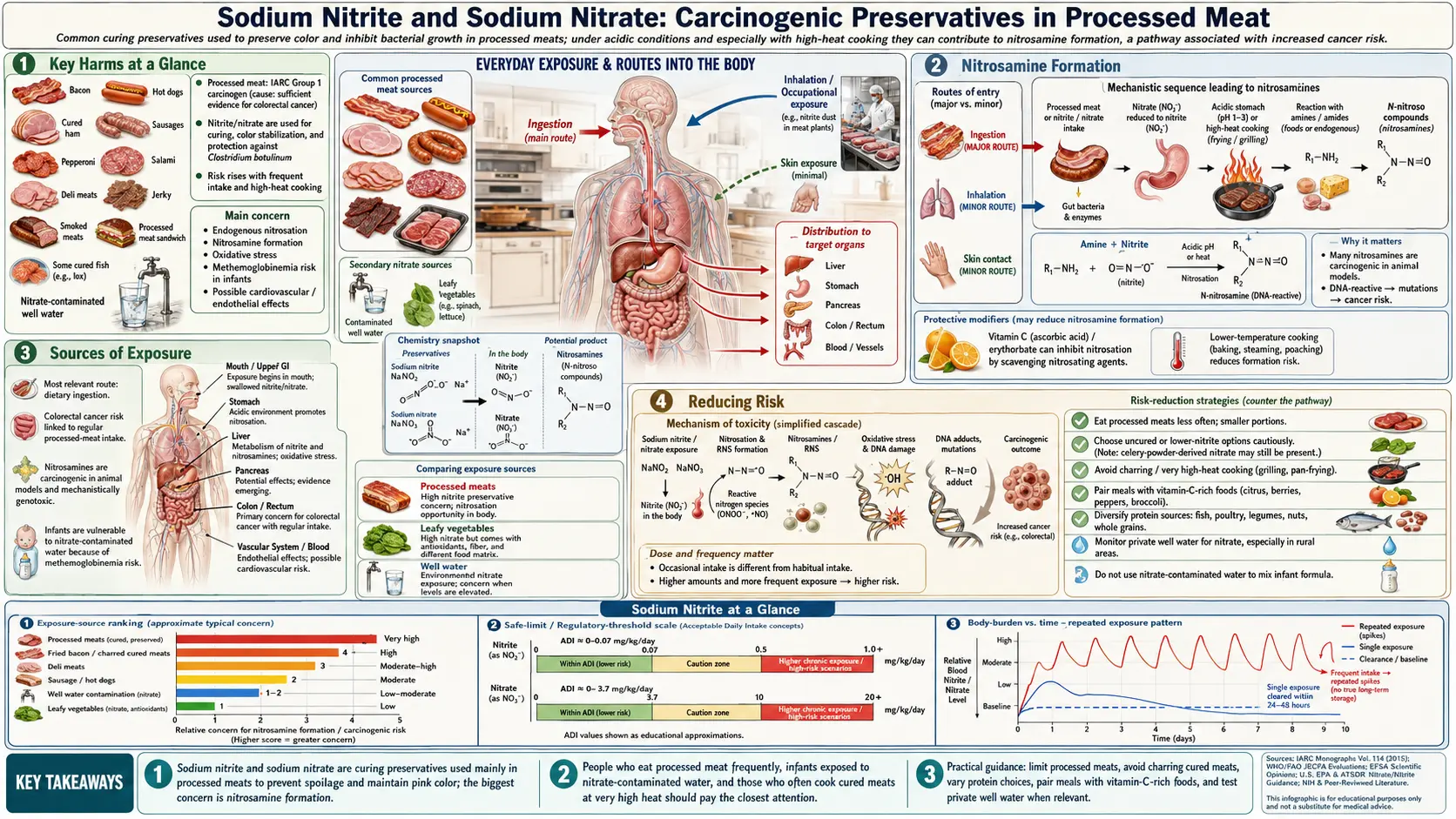
Sodium nitrite (NaNO2, E250) and sodium nitrate (NaNO3, E251) are inorganic salt preservatives that have been used in meat curing since the early twentieth century, having gradually replaced the traditional use of saltpeter (potassium nitrate) that dates back centuries further. These compounds serve as curing agents that simultaneously preserve meat from dangerous bacterial contamination, fix the pink or red color consumers associate with cured products, and contribute to the characteristic flavors of bacon, ham, hot dogs, and deli meats. Despite their functional importance, sodium nitrite and nitrate are now understood to be the primary reason the World Health Organization classified processed meat as a Group 1 carcinogen in 2015 — placing it in the same evidence category as tobacco smoke and asbestos.
Table of Contents
- Overview
- Key Harms at a Glance
- How They Work
- Nitrosamine Formation
- Cancer Risk (Health Effects)
- Other Health Effects
- Sources of Exposure (Exposure Routes)
- The "Uncured" Label Deception
- Regulatory Status (Safety)
- Reducing Risk (How to Avoid)
- Research Papers and References
- Connections
- Featured Videos
Key Harms at a Glance
- Group 1 carcinogen association — processed meat cured with nitrite is classified by IARC as Group 1 (carcinogenic to humans), largely driven by N-nitroso compound formation.
- Colorectal cancer — each 50 g/day of processed meat raises colorectal cancer risk by approximately 18%.
- Stomach and esophageal cancer — endogenous nitrosamine formation in acidic gastric conditions contributes to upper-GI cancer risk.
- Methemoglobinemia — high-dose nitrite exposure oxidizes hemoglobin, potentially fatal in infants ("blue baby syndrome").
- Cardiovascular disease — processed-meat consumption is associated with an 18–42% increased risk of ischemic heart disease and stroke.
- Migraine triggers — nitric-oxide-mediated vasodilation from cured meats is a well-recognized migraine provoker.
- Children disproportionately exposed — hot dogs and deli meats account for substantial per-kilogram intake in pediatric diets.
Overview
Sodium nitrite (chemical formula NaNO2) is an inorganic salt that appears as a white to slightly yellow crystalline powder highly soluble in water. In food regulation it carries the European Union additive code E250. Its close chemical relative, sodium nitrate (NaNO3, E251), is a more stable oxidized form that bacteria and stomach acid gradually convert into nitrite over time. Historically, meat curers used sodium nitrate (saltpeter) exclusively, but the twentieth century brought a shift toward direct application of sodium nitrite because it acts faster, is more controllable, and delivers a consistent result. Today most commercially cured products use sodium nitrite directly, sometimes in combination with sodium nitrate for long-aged products like dry-cured hams and salamis where a slow, sustained release of nitrite over weeks or months is desirable.
The use of nitrite curing expanded rapidly throughout the early 1900s as food manufacturers sought to prevent botulism — a potentially fatal paralytic illness caused by the toxin of Clostridium botulinum — in the expanding market for shelf-stable and semi-preserved meat products. By mid-century, sodium nitrite was nearly universal in the processed meat industry, and regulatory agencies in the United States and Europe established permitted maximum levels that remain largely in force today. What was not appreciated at the time was that the same chemistry enabling preservation also generates one of the most potent classes of carcinogens known to science.
How They Work
Sodium nitrite performs three distinct and commercially valuable functions in cured meat, each arising from its reactivity as a nitrogen oxide donor:
- Color fixation — nitrite dissociates in the acidic meat environment to yield nitrous acid, which reacts with the muscle protein myoglobin to form nitrosomyoglobin. This bright pink to red compound is thermally stable, converting upon cooking to the denatured nitrosohemochrome that gives cooked cured meats like bacon and hot dogs their characteristic and commercially expected pink color. Without nitrite, cooked cured meats would present the grey-brown of oxidized myoglobin, which consumers find unappetizing.
- Antimicrobial action — sodium nitrite inhibits the germination and outgrowth of Clostridium botulinum spores through a mechanism that involves chelation of iron in microbial metabolic enzymes and interference with sulfur-containing proteins. It also has meaningful activity against Listeria monocytogenes, Staphylococcus aureus, and other pathogens. This protective function is the primary justification regulators cite for permitting sodium nitrite despite its carcinogenic potential.
- Flavor development — nitrite participates in complex oxidative reactions during curing and cooking that produce the specific sulfur- and nitrogen-containing volatile compounds responsible for the distinctive "cured" flavor — the taste immediately recognizable as bacon or ham. Meat preserved by alternative methods lacks this flavor profile, which remains a commercial and consumer barrier to replacing nitrite.
Sodium nitrate functions as a slow-release reservoir: bacteria and reducing agents in the meat gradually convert nitrate to nitrite over the extended curing periods used for dry-cured products. This provides sustained antimicrobial and color-fixing activity across aging periods that can span months to years for traditional hams and charcuterie.
Nitrosamine Formation
The central toxicological concern with sodium nitrite is its ability to react with secondary amines — nitrogen-containing compounds naturally present in protein-rich foods — to generate N-nitroso compounds, more commonly called nitrosamines. This reaction is not hypothetical; it is well-characterized, reproducible chemistry that occurs under conditions routinely encountered in both food preparation and human digestion.
The Chemistry
In acidic conditions, sodium nitrite generates nitrous acid (HNO2), which further yields the nitrosonium ion (NO+). This electrophilic species attacks the nitrogen atom of a secondary amine (R2NH) to produce an N-nitrosamine (R2N-N=O). Two of the most toxicologically significant nitrosamines formed from processed meat are:
- N-nitrosodimethylamine (NDMA) — formed from dimethylamine, a common degradation product of trimethylamine oxide found abundantly in fish and some meats. NDMA is classified by IARC as a Group 2A (probable human) carcinogen. It is also the nitrosamine that attracted widespread attention in 2019 when it was discovered as a contamination in the drugs ranitidine and some sartan blood pressure medications.
- N-nitrosodiethylamine (NDEA) — formed from diethylamine; also a Group 2A carcinogen with strong hepatocarcinogenic activity in animal models.
High-Heat Cooking
The rate of nitrosamine formation increases dramatically with temperature. When bacon, sausages, or hot dogs are fried, grilled, or barbecued, the high-heat environment accelerates the reaction between residual nitrite and meat amines. Frying bacon at typical pan temperatures can generate nitrosamine concentrations exceeding 100 micrograms per kilogram of cooked meat. The longer and hotter the cooking, the higher the yield.
Gastric Formation
Even cold, uncooked processed meat (as eaten in sandwiches or as deli slices) undergoes nitrosamine formation in the stomach. The highly acidic gastric environment (pH 1.5–3.5) is chemically ideal for nitrosamine synthesis, and the stomach's high protein content provides abundant amine substrates. There is therefore no preparation method that eliminates the risk of nitrosamine exposure from sodium nitrite-containing products.
Ascorbic Acid as a Partial Inhibitor
Vitamin C (ascorbic acid) competes with amines for the nitrosonium ion, reducing it to nitric oxide (NO) before it can react with secondary amines. This reaction is the reason why the FDA and meat manufacturers began adding ascorbic acid or its isomer erythorbic acid to cured meats, particularly bacon, starting in the 1970s. Studies confirmed that this practice significantly reduces nitrosamine levels in cooked products. However, ascorbic acid does not eliminate nitrosamine formation — it reduces it partially, and the reduction depends on the ratio of ascorbic acid to nitrite, cooking temperature, and cooking time. Nitrosamines are still found in cured meats that contain added ascorbate. Additionally, ascorbic acid is rapidly consumed and loses protective capacity in the harsh gastric environment.
Cancer Risk
In October 2015, the International Agency for Research on Cancer (IARC) — the cancer research arm of the WHO — published a landmark evaluation concluding that processed meat is a Group 1 carcinogen (sufficient evidence of causing cancer in humans). The evaluation, conducted by 22 experts from 10 countries and published in The Lancet Oncology (Bouvard et al., 2015), reviewed more than 800 epidemiological studies. The key cancer associations were:
Colorectal Cancer
The evidence for a causal link between processed meat and colorectal cancer was deemed sufficient for the Group 1 classification. The dose-response analysis found that consuming 50 grams of processed meat per day — roughly one hot dog or two strips of bacon — is associated with an 18% relative increase in colorectal cancer risk. Colorectal cancer is the third most common cancer worldwide and the second leading cause of cancer death, making even modest relative risk increases consequential at a population level.
Stomach Cancer
Multiple studies across diverse populations have linked high consumption of cured, smoked, and nitrite-containing meats to elevated gastric cancer risk. The stomach is a site of particular concern because the acidic environment promotes in situ nitrosamine formation, and the gastric mucosa is directly exposed to these carcinogens at high concentrations. Countries with traditionally high consumption of cured and salt-preserved meats — Japan, Korea, parts of Eastern Europe — have historically had elevated rates of gastric cancer compared to populations with lower processed meat intake.
Esophageal Cancer
Several prospective cohort studies have found positive associations between processed meat consumption and esophageal cancer, particularly esophageal adenocarcinoma. The mechanistic pathway is plausible: nitrosamines are swallowed and contact the esophageal mucosa before reaching the stomach, and the esophagus is exposed to carcinogens in the same bolus of food containing the cured meat.
Pancreatic Cancer
A meta-analysis published in the British Journal of Cancer found a 19% increased risk of pancreatic cancer per 50 grams of processed meat consumed daily. Pancreatic cancer has one of the worst prognoses of any cancer, with a five-year survival rate below 12%. The mechanism may involve nitrosamines reaching pancreatic tissue via the bloodstream after gastrointestinal absorption.
Other Health Effects
Methemoglobinemia
Sodium nitrite oxidizes ferrous iron (Fe2+) in hemoglobin to ferric iron (Fe3+), creating methemoglobin — a form of hemoglobin unable to carry oxygen. High nitrite exposure can cause methemoglobinemia, clinically presenting as cyanosis, shortness of breath, and in severe cases death by asphyxiation. Infants under six months are most vulnerable because fetal hemoglobin is more susceptible to oxidation and infant gut bacteria convert nitrate to nitrite more readily than adult flora. The classic presentation, historically called "blue baby syndrome," was primarily associated with nitrate-contaminated well water rather than cured meat, but the same biochemistry applies. The FDA's permitted limits in food are set below levels that would cause acute methemoglobinemia in adults; however, infants should not consume cured meats or high-nitrate well water.
Cardiovascular Effects
Nitric oxide (NO), the downstream metabolite of both dietary nitrite and nitrate, is a potent vasodilator. In controlled clinical settings, inorganic nitrate from vegetables is being investigated as a therapeutic vasodilator for hypertension. However, the delivery matrix matters: the oxidative and inflammatory milieu of processed meat consumption is associated with adverse rather than beneficial cardiovascular outcomes. Multiple large prospective studies have linked high processed meat consumption to increased risk of cardiovascular disease, ischemic stroke, and heart failure, with risk estimates ranging from 18% to 42% increased risk for high versus low consumers.
Migraine Triggering
Clinicians have long observed that cured meats are a common dietary trigger for migraine headaches in susceptible individuals. The mechanism is thought to involve nitric oxide- mediated vasodilation of intracranial blood vessels, a process that can initiate or amplify the vascular changes underlying migraine. Some patients with migraines experience complete or substantial relief when processed meat is eliminated from the diet.
Potential Link to Neurodegeneration
Researcher Suzanne de la Monte at Brown University has proposed a mechanistic link between dietary nitrosamines and Alzheimer's disease. Her research, published between 2006 and 2014, suggests that nitrosamines can impair insulin signaling in the brain by damaging insulin receptor substrates and mitochondrial function, leading to a condition she described as "type 3 diabetes" of the brain. In animal studies, nitrosamine administration reproduced features of Alzheimer's pathology including brain atrophy, tau phosphorylation, and amyloid precursor protein accumulation. While this hypothesis remains under investigation and has not been definitively established in human populations, the correlation between rising nitrosamine dietary exposure and increasing Alzheimer's prevalence in developed countries is consistent with the proposed mechanism.
Sources of Exposure
- Bacon — one of the highest-risk processed meat products because high-heat frying combines residual nitrite with abundant meat amines at temperatures that maximize nitrosamine yield.
- Hot dogs and frankfurters — nearly universal use of sodium nitrite; a staple food for children in many countries.
- Ham and deli meats — sliced ham, turkey breast, roast beef, salami, bologna, and other deli counter products almost invariably contain sodium nitrite.
- Sausages — breakfast sausage, Italian sausage, bratwurst, kielbasa, pepperoni, chorizo, and many other varieties.
- Beef jerky and meat snacks — most commercially produced jerky uses sodium nitrite for preservation and color.
- Smoked fish — commercially smoked salmon, whitefish, and other smoked seafood products frequently contain sodium nitrite.
- Canned meats — canned ham, corned beef, Vienna sausages, and luncheon meats commonly contain sodium nitrite.
- Vegetables (with protective context) — nitrate is found naturally in beets, celery, spinach, lettuce, and other vegetables, sometimes at concentrations far exceeding those in cured meat. However, these vegetables simultaneously contain vitamin C, polyphenols, and other antioxidants that inhibit nitrosamine formation, which is why vegetable consumption is associated with cancer reduction, not cancer promotion.
The "Uncured" Label Deception
In response to growing consumer awareness of sodium nitrite's cancer risk, the processed meat industry developed a new product category marketed with labels including "uncured," "no nitrites added," "no nitrates added," and "naturally cured." These labels are systematically misleading.
The overwhelming majority of products bearing these labels use celery powder, celery juice, celery extract, or beet powder as curing agents. Celery is extraordinarily rich in naturally occurring nitrate — one of the highest-nitrate vegetables in the food supply. When celery powder is added to meat together with a bacterial starter culture, the bacteria convert celery nitrate to nitrite during the curing process. The finished product contains the same compound — nitrite — as conventionally cured meat.
- Equivalent or higher nitrite levels — multiple analytical studies have measured nitrite concentrations in "uncured" products and found levels comparable to, and in some cases higher than, conventionally cured equivalents. A 2011 study in the Journal of Food Protection confirmed that consumer-level nitrite exposure from "natural" products is not meaningfully lower than from conventional products.
- Identical chemistry — the nitrosamine-forming reaction is determined by the presence of nitrite, secondary amines, and heat or acid, not by the source of the nitrite. Celery-derived nitrite and synthetic sodium nitrite are chemically identical; a nitrosamine formed from one is indistinguishable from a nitrosamine formed from the other.
- USDA labeling rules — the USDA permits the "no nitrites added" label as long as no synthetic sodium nitrite is directly added, even if nitrite from celery powder performs the identical curing function. An asterisk and small-print qualification noting "except those naturally occurring in celery powder" is required but routinely overlooked by consumers.
- Consumer confusion — surveys have consistently found that consumers interpret "uncured" and "no nitrites added" labels as signaling a healthier or lower-cancer-risk product. This belief is false, and the labels are broadly considered by consumer advocacy groups and independent researchers to constitute deceptive marketing.
Regulatory Status
United States
The FDA permits sodium nitrite in cured meat at a maximum of 200 parts per million (ppm) in finished products, and sodium nitrate at a maximum of 500 ppm. These limits were established decades ago and have not been substantially revised despite the WHO Group 1 carcinogen classification. The Center for Science in the Public Interest (CSPI) has filed multiple petitions over the years requesting the FDA to ban or more strictly limit sodium nitrite in processed meat, citing carcinogenicity data. The agency has declined to act on each occasion, deferring to the antimicrobial necessity argument.
European Union
The EU permits sodium nitrite at 150 mg/kg (ppm) in most cured meat products, a limit lower than the US maximum. The European Food Safety Authority (EFSA) has conducted multiple re-evaluations of nitrite safety and has been progressively reducing permitted levels in response to accumulating evidence. Several EU member states, including France, have engaged in parliamentary debates about phasing out nitrite in cured meats altogether.
California Proposition 65 and SB 343
California's Proposition 65 requires businesses to provide warnings when products expose consumers to listed carcinogens. N-nitrosodimethylamine (NDMA) and other nitrosamines are listed Proposition 65 carcinogens. California Senate Bill 343, introduced in response to the WHO classification, proposed mandatory cancer warning labels on processed meat products sold in California. The meat industry lobbied vigorously against the measure, and while the bill did not become law in its original form, it reflected growing legislative recognition of processed meat's cancer risk.
Reducing Risk
- Reduce or eliminate processed meat consumption — the single most effective strategy. The WHO evidence shows a clear dose-response relationship: lower consumption means lower risk.
- Cook at lower temperatures — if consuming processed meat, avoid high-heat frying or open-flame grilling. Microwaving or baking at moderate oven temperatures produces significantly fewer nitrosamines than pan-frying or grilling.
- Add vitamin C — consuming vitamin C-rich foods (citrus fruits, bell peppers, strawberries) alongside processed meat can partially inhibit in vivo nitrosamine formation by competing for the nitrosonium ion. Look for products that already contain ascorbic acid or erythorbic acid.
- Do not be misled by "uncured" labels — products labeled uncured or "no nitrites added" typically contain celery-derived nitrites and carry equivalent cancer risk.
- Choose truly nitrite-free alternatives — a small number of artisan and specialty meat producers make genuinely nitrite-free products. These will typically have a grey-brown color after cooking and a shorter shelf life. Ask producers directly about their curing methods.
- Protect children — children have higher food intake relative to body weight and developing biological systems more vulnerable to carcinogens. Hot dogs and deli meats are among the most consumed processed meat products by children. Limiting or eliminating them from children's diets is especially important.
- Replace with fresh alternatives — fresh-cooked chicken, turkey, or roast beef provide protein without nitrite; legumes and eggs are nitrosamine-free plant and animal protein alternatives.
Research Papers and References
The following research papers and authoritative sources underpin the claims on this page. Links resolve to DOI or PubMed records.
- Bouvard V, Loomis D, Guyton KZ, et al. Carcinogenicity of consumption of red and processed meat. Lancet Oncology. 2015;16(16):1599-1600.
- International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 114: Red Meat and Processed Meat. Lyon: IARC; 2018. IARC Monographs Vol. 114
- Micha R, Wallace SK, Mozaffarian D. Red and processed meat consumption and risk of incident coronary heart disease, stroke, and diabetes mellitus: a systematic review and meta-analysis. Circulation. 2010;121(21):2271-2283.
- Cross AJ, Leitzmann MF, Gail MH, et al. A prospective study of red and processed meat intake in relation to cancer risk. PLoS Medicine. 2007;4(12):e325.
- Larsson SC, Wolk A. Meat consumption and risk of colorectal cancer: a meta-analysis of prospective studies. International Journal of Cancer. 2006;119(11):2657-2664.
- Jakszyn P, Gonzalez CA. Nitrosamine and related food intake and gastric and oesophageal cancer risk: a systematic review of the epidemiological evidence. World Journal of Gastroenterology. 2006;12(27):4296-4303.
- Larsson SC, Hakansson N, Wolk A. Dietary sodium nitrite and nitrate and risk of pancreatic cancer: a prospective study. British Journal of Cancer. 2006;94(6):906-910.
- de la Monte SM, Tong M, Ngon J, Setshedi M, Wands JR. Nitrosamine exposure exacerbates high fat diet-mediated type 2 diabetes mellitus, non-alcoholic steatohepatitis, and neurodegeneration with features of Alzheimer's disease. Journal of Alzheimer's Disease. 2009;17(4):827-840.
- Hord NG, Tang Y, Bryan NS. Food sources of nitrates and nitrites: the physiologic context for potential health benefits. American Journal of Clinical Nutrition. 2009;90(1):1-10.
- Sebranek JG, Bacus JN. Cured meat products without direct addition of nitrate or nitrite: what are the issues? Meat Science. 2007;77(1):136-147.
- Sindelar JJ, Milkowski AL. Human safety controversies surrounding nitrate and nitrite in the diet. Nitric Oxide. 2012;26(4):259-266.
- Tricker AR. N-nitroso compounds and man: sources of exposure, endogenous formation and occurrence in body fluids. European Journal of Cancer Prevention. 1997;6(3):226-268.
- Milkowski A, Garg HK, Coughlin JR, Bryan NS. Nutritional epidemiology in the context of nitric oxide biology: a risk-benefit evaluation for dietary nitrite and nitrate. Nitric Oxide. 2010;22(2):110-119.
- Rohrmann S, Overvad K, Bueno-de-Mesquita HB, et al. Meat consumption and mortality — results from the European Prospective Investigation into Cancer and Nutrition. BMC Medicine. 2013;11:63.
- Santarelli RL, Pierre F, Corpet DE. Processed meat and colorectal cancer: a review of epidemiologic and experimental evidence. Nutrition and Cancer. 2008;60(2):131-144.
- Gilchrist M, Shore AC, Benjamin N. Inorganic nitrate and nitrite and control of blood pressure. Cardiovascular Research. 2011;89(3):492-498.
Connections
- Migraine
- Preservatives
- Sodium Nitrite (Preservatives)
- Hypertension
- Sodium Benzoate
- Food Additives
- Sulfites
- Cancer
- Cardiovascular Disease
- Stroke
- Vitamin C
- Iron
- Detox Protocols
- Carrageenan
- Beef
- rBGH
- Ractopamine