BHT (Butylated Hydroxytoluene): Synthetic Antioxidant in Your Food
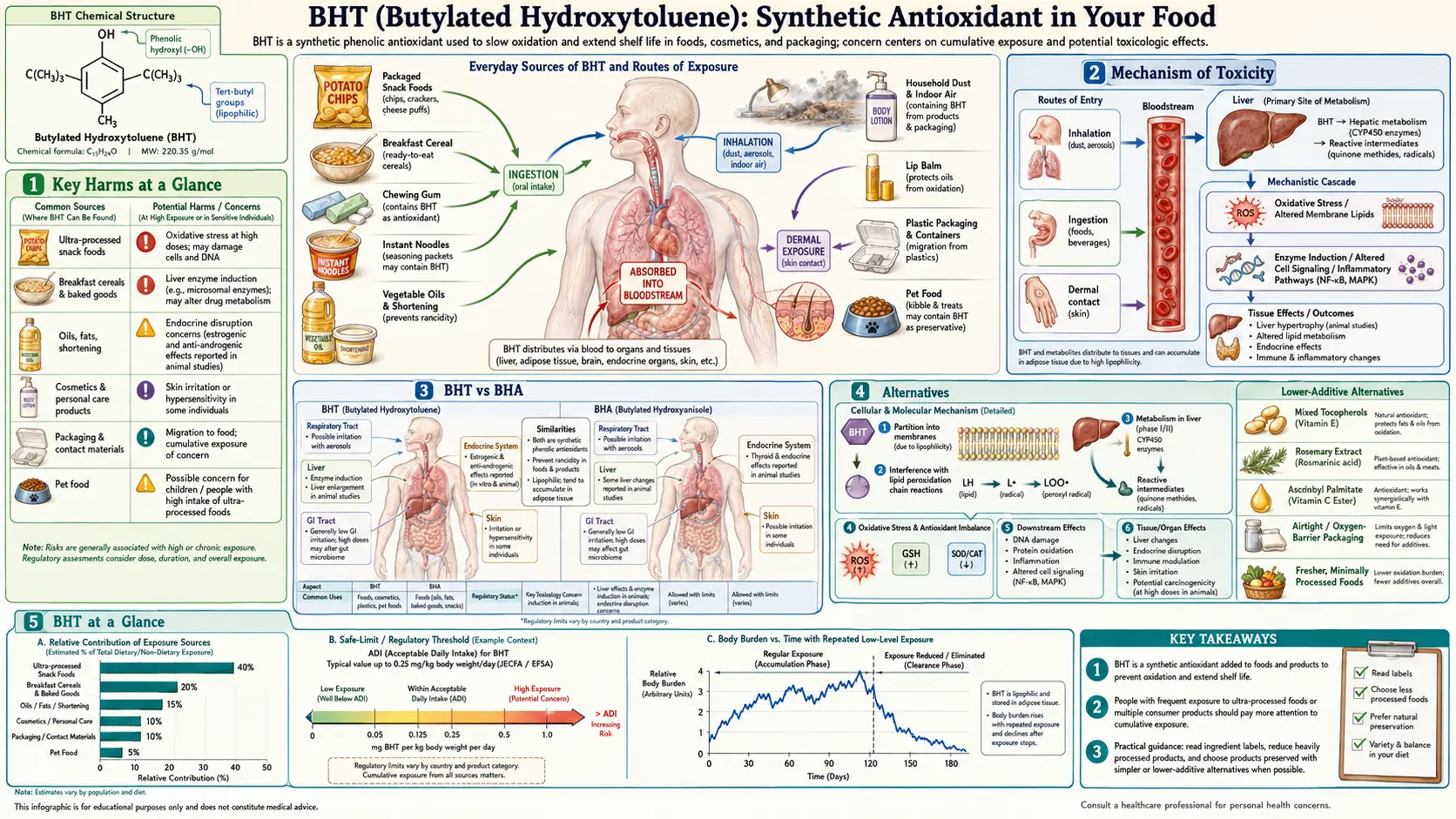
Table of Contents
- Key Harms at a Glance
- Overview
- Sources
- Mechanism of Toxicity
- Health Effects
- BHT vs BHA
- Regulatory Status
- The Packaging Problem
- Alternatives
- Research Papers and References
- Connections
- Featured Videos
Key Harms at a Glance
- Tumor promoter — enhances chemically initiated tumors in bladder, thyroid, and liver in rodent bioassays.
- Lung hemorrhage in mice — species-specific pulmonary toxicity at high doses.
- Liver and kidney enlargement — consistent in rodent feeding studies.
- Thyroid disruption — reduces circulating T3/T4 in animal studies.
- Endocrine-disrupting potential — weak estrogenic and anti-androgenic activity in vitro.
- Bioaccumulative — detected in human adipose tissue, blood plasma, and breast milk.
- Packaging migration — BHT enters food from wax cartons and plastic films without appearing on labels.
Overview
BHT, or butylated hydroxytoluene, is a synthetic phenolic antioxidant with the chemical name 2,6-di-tert-butyl-4-methylphenol. It is listed on food labels as E321 in the European Union and is used worldwide to prevent oxidative rancidity in fats, oils, and fat-containing foods. BHT is derived from petroleum-based precursors — specifically from p-cresol and isobutylene — making it a fully synthetic compound with no natural analog in the human diet.
BHT works by donating a hydrogen atom to free radicals generated during lipid oxidation, thereby interrupting the chain reaction that causes fats to become rancid and off-flavored. This extends shelf life significantly, which is why it became ubiquitous in the processed food industry following its introduction in the 1940s and 1950s.
BHT is frequently paired with BHA (butylated hydroxyanisole), another synthetic antioxidant. The two compounds act synergistically, providing greater antioxidant protection together than either does alone. Both are oil-soluble and migrate readily into fatty food matrices as well as into food from the packaging materials in which it is incorporated.
While BHT is considered safe by regulatory agencies at current permitted levels, a substantial body of animal research has raised questions about its effects on the liver, lungs, thyroid, and endocrine system, particularly at higher doses. Its continued presence in processed foods, combined with cumulative exposure from cosmetics, pharmaceuticals, and food packaging, makes it an important additive to understand.
Sources
BHT is one of the most widely used synthetic antioxidants in the food supply. Major dietary sources include:
- Breakfast cereals — the single most common source of BHT exposure; many ready-to-eat cereals list BHT as a preservative for the fat content of the grain or added directly to the packaging
- Chewing gum — BHT is added to prevent oxidation of the gum base, a blend of synthetic polymers and fats
- Snack foods — potato chips, crackers, popcorn, and extruded snacks frequently contain BHT to extend shelf life of their fat content
- Butter and margarine — some products use BHT to retard rancidity during refrigerator storage
- Vegetable oils — edible oils, particularly those intended for long shelf life or high-temperature use, may contain BHT
- Beer — BHT is used in some brewing processes to prevent oxidative off-flavors
- Food packaging — BHT is incorporated into wax paper, plastic film, and cardboard packaging materials, from which it migrates into the food product (see Section 7)
- Cosmetics and personal care products — lipstick, moisturizers, sunscreens, and hair products frequently list BHT, contributing to dermal and oral exposure
- Rubber and plastics — BHT is used industrially as an antioxidant stabilizer in rubber goods and plastic products, contributing to background environmental exposure
Because BHT is fat-soluble, it accumulates in fatty tissues of the body. Biomonitoring studies have detected BHT and its metabolites in human adipose tissue, blood plasma, and breast milk, confirming meaningful systemic absorption from routine dietary and cosmetic exposure.
Mechanism of Toxicity
BHT's toxicity — to the extent it exists — stems primarily from its metabolic activation by cytochrome P450 enzymes (CYP450), particularly CYP1A1, CYP1A2, and CYP2B6. The key reactive intermediate is BHT-quinone methide (BHT-QM), a highly electrophilic species capable of forming covalent adducts with cellular proteins and DNA.
The metabolic pathway proceeds as follows:
- BHT is absorbed from the gastrointestinal tract and transported to the liver.
- Hepatic CYP450 enzymes oxidize BHT at the 4-methyl group, yielding BHT-alcohol and subsequently BHT-aldehyde.
- Further oxidation produces BHT-quinone methide, the reactive electrophile that can alkylate nucleophiles including glutathione, protein sulfhydryls, and DNA bases.
- In the lungs, a separate activation pathway — mediated by pulmonary CYP2B6 and peroxidases — generates reactive metabolites locally, explaining the species-specific lung toxicity observed in mice.
At high doses in animal models, BHT causes lung toxicity in mice specifically, characterized by hemorrhage, edema, and necrosis of the alveolar epithelium. This lung toxicity is highly species-specific and has not been demonstrated in rats, dogs, or monkeys, raising questions about its relevance to human risk.
Liver hypertrophy — enlargement of the liver accompanied by increased activity of drug-metabolizing enzymes — is observed in rodents given BHT-supplemented diets. This is interpreted as an adaptive rather than purely toxic response, though it indicates significant perturbation of hepatic function.
BHT has demonstrated thyroid effects in rodent studies, including reductions in circulating thyroid hormone levels and alterations in thyroid gland histology. The mechanism may involve induction of hepatic enzymes that accelerate thyroid hormone catabolism.
Critically, BHT has been classified as a tumor promoter rather than a tumor initiator in several animal models. This means BHT does not directly damage DNA to cause cancer initiation, but at high doses it can enhance the growth and progression of tumors already initiated by other carcinogens. The distinction is important for risk assessment, as tumor promotion is generally considered a threshold phenomenon with a no-observed-effect level.
Health Effects
The health effects of BHT in humans remain incompletely characterized because most evidence derives from animal studies at doses substantially higher than typical human exposure. Nevertheless, the following effects have been reported:
- Liver and kidney enlargement — consistently observed in rodents fed BHT-supplemented diets; associated with increased hepatic enzyme activity and altered lipid metabolism
- Lung hemorrhage in mice — severe at high doses (200–400 mg/kg body weight); highly species-specific and not observed in other mammals, limiting direct extrapolation to humans
- Thyroid disruption — reductions in serum T3 and T4 in rodents; potential concern for people with pre-existing thyroid dysfunction or iodine insufficiency
- Tumor promotion — enhancement of chemically initiated tumors in the bladder, thyroid, and liver in rodent bioassays at high BHT doses; not classified as a human carcinogen by IARC
- Developmental and reproductive effects — some studies report decreased litter size, altered pup development, and changes in reproductive organ weight at high doses; data are inconsistent across studies
- Allergic skin reactions — contact dermatitis and urticaria have been documented in humans exposed to BHT in cosmetics and food; cross-reactivity with BHA has been reported
- Endocrine-disrupting potential — in vitro studies have shown weak estrogenic and anti-androgenic activity; the relevance of these findings to dietary exposure levels remains under investigation
It is important to note that some research has suggested potential beneficial effects of BHT, including antiviral activity against enveloped viruses (herpes, influenza) and anti-tumor effects in certain models — illustrating the complexity of evaluating a compound with multiple biological activities across a wide dose range.
BHT vs BHA
BHT and BHA are structural analogs — both are alkylated phenols — and are frequently used together, but they have meaningfully different safety profiles:
- Structure — BHA (butylated hydroxyanisole) contains a methoxy group at the 2- or 3-position of the phenol ring, while BHT has two tert-butyl groups and a methyl group. This structural difference confers distinct metabolic pathways and biological activities.
- Carcinogenicity classification — BHA is classified as IARC Group 2B (possibly carcinogenic to humans) based on evidence of forestomach tumors in rodents — a tissue humans do not possess. BHT is not classified by IARC, reflecting less concern about carcinogenic potential despite its tumor-promoting activity in some models.
- Lung effects — BHT is more studied for pulmonary toxicity than BHA; BHA's primary concern is its carcinogenic designation and potential estrogenic activity.
- Synergistic antioxidant action — when combined, BHA and BHT regenerate each other's antioxidant capacity through electron transfer, making the combination more effective than the sum of the parts. This synergism is exploited commercially to reduce the total amount of each compound needed.
- Regulatory differences — BHA is more restricted in some jurisdictions (notably Japan has banned BHA in certain food categories). BHT generally faces fewer restrictions, though both are under periodic review in the EU.
Both compounds are fat-soluble, bioaccumulative, and contribute to cumulative synthetic antioxidant burden when both are present in the diet simultaneously.
Regulatory Status
BHT occupies an unusual regulatory position: it is widely approved yet subject to increasing scrutiny:
- United States (FDA) — BHT holds GRAS (Generally Recognized as Safe) status. It is permitted at a maximum of 0.02% of the fat or oil content of a food product, or 200 ppm. The GRAS affirmation dates to the 1970s and has not been comprehensively re-evaluated using modern toxicological standards.
- European Union — Approved as E321 with an Acceptable Daily Intake (ADI) of 0–0.3 mg/kg body weight per day (EFSA, 2012). Use is permitted but has declined as food manufacturers respond to consumer demand for cleaner labels.
- Codex Alimentarius — The joint FAO/WHO expert committee (JECFA) has established an ADI of 0–0.3 mg/kg body weight/day, consistent with EU limits.
- IARC classification — BHT is not listed in any IARC carcinogen group. This distinguishes it from BHA (Group 2B) and reflects the determination that available evidence is insufficient to classify BHT as a human carcinogen.
- NTP (National Toxicology Program) — The NTP has conducted extensive studies on BHT. Findings include evidence of lung toxicity in mice and tumor promotion in rodents, but BHT is not listed in the NTP's Report on Carcinogens.
- UK — The Food Standards Agency has noted BHT as a food additive of interest for further monitoring, particularly regarding cumulative exposure from food and cosmetics.
A growing number of food manufacturers, particularly in the natural and organic sector, have voluntarily removed BHT in response to consumer preference rather than regulatory requirement.
The Packaging Problem
One of the least understood aspects of BHT exposure is its presence in food packaging materials. BHT is incorporated into wax coatings on cardboard cereal boxes, plastic food-wrap films, and other food contact materials specifically to prevent the packaging itself from degrading. This use is technologically distinct from its direct addition to food.
The critical issue is migration: BHT diffuses from the packaging matrix into the food product over time, especially when the food has a high fat content (which dissolves BHT from the packaging) or when packaging is stored for extended periods. The rate of migration increases with temperature, fat content, and storage duration.
From a regulatory and labeling standpoint, this creates a transparency problem. When BHT migrates into food from packaging, it is considered an indirect food additive rather than a directly added ingredient. In many jurisdictions, including the United States, BHT migrating from packaging is not required to appear on the ingredient label of the food product. Consumers reading ingredient labels therefore cannot determine whether BHT is present if it arrived via packaging migration rather than direct addition.
This pathway is likely responsible for a meaningful fraction of total dietary BHT exposure, particularly for consumers who eat large amounts of packaged breakfast cereals, crackers, and snack foods — foods that may not list BHT in their ingredients but whose packaging contains it.
Alternatives
The food industry has access to several effective natural alternatives to BHT that provide antioxidant protection without the toxicological concerns associated with synthetic phenolic antioxidants:
- Tocopherols (Vitamin E) — mixed tocopherols, particularly alpha- and gamma-tocopherol, are highly effective fat-soluble antioxidants that perform the same function as BHT in most applications. They are approved as food additives (E306–E309) and carry a well-established safety profile as essential nutrients.
- Rosemary extract — contains carnosic acid and carnosol, potent antioxidants that have been shown to match or exceed BHT's effectiveness in protecting vegetable oils and fats from oxidation. Approved in the EU as E392.
- Green tea catechins — epigallocatechin gallate (EGCG) and related polyphenols from green tea extract are emerging as effective antioxidants in food applications, with additional health-promoting properties.
- Ascorbic acid (Vitamin C) and ascorbyl palmitate — water- and fat-soluble forms of Vitamin C, respectively, serve as antioxidant synergists that help regenerate tocopherols and directly scavenge oxygen.
These alternatives are increasingly preferred by manufacturers targeting health-conscious consumers. The primary barriers to universal adoption are cost (natural antioxidants are typically more expensive than BHT) and performance variability across different food matrices and processing conditions.
Research Papers and References
Key research papers and authoritative sources. Links resolve to DOI, PubMed, or the issuing agency.
- Lanigan RS, Yamarik TA. Final report on the safety assessment of BHT. Int J Toxicol. 2002;21 Suppl 2:19-94.
- Witschi HP. Enhanced tumour development by butylated hydroxytoluene (BHT) in the liver, lung and gastro-intestinal tract. Food Chem Toxicol. 1986;24(10-11):1127-1130.
- Thompson DC, Perera K, London R. Quinone methide formation from para isomers of methylphenol (cresol), ethylphenol, and isopropylphenol. Chem Res Toxicol. 1995;8(1):55-60.
- Verhagen H, Schilderman PA, Kleinjans JC. Butylated hydroxyanisole in perspective. Chem Biol Interact. 1991;80(2):109-134.
- Ito N, Hirose M, Fukushima S, et al. Studies on antioxidants: their carcinogenic and modifying effects on chemical carcinogenesis. Food Chem Toxicol. 1986;24(10-11):1071-1082.
- EFSA Panel on Food Additives and Nutrient Sources. Re-evaluation of butylated hydroxytoluene BHT (E 321) as a food additive. EFSA J. 2012;10(3):2588.
- National Toxicology Program. Toxicology and carcinogenesis studies of butylated hydroxytoluene (CAS No. 128-37-0) in F344/N rats and B6C3F1 mice. NTP TR 459. 1998.
- Gharavi N, El-Kadi AO. tert-Butylhydroquinone is a novel aryl hydrocarbon receptor ligand. Drug Metab Dispos. 2005;33(3):365-372.
- Oikawa S, Nishino K, Oikawa S, et al. Oxidative DNA damage and apoptosis induced by metabolites of butylated hydroxytoluene. Biochem Pharmacol. 1998;56(3):361-370.
- Malkinson AM. The genetic basis of susceptibility to lung tumors in mice. Toxicology. 1989;54(3):241-271.
- Kahl R, Kappus H. Toxicology of the synthetic antioxidants BHA and BHT in comparison with the natural antioxidant vitamin E. Z Lebensm Unters Forsch. 1993;196(4):329-338.
- Strickland PT, Kensler TW. Modulation of aflatoxin B1-induced genotoxicity in mice by butylated hydroxytoluene. Mutat Res. 1981;88(3):261-270.
- van Esch GJ. Toxicology of toluene diisocyanate. Arch Toxicol Suppl. 1986;9:136-144.
- Bauer AK, Dwyer-Nield LD, Malkinson AM. High BHT and low NNK-induced lung tumor multiplicities in mice are associated with decreased levels of oxidized DNA bases. Exp Lung Res. 2000;26(7):491-507.
- Nair B. Final report on the safety assessment of benzyl alcohol, benzoic acid, and sodium benzoate. Int J Toxicol. 2001;20 Suppl 3:23-50.
- Botterweck AA, Verhagen H, Goldbohm RA, et al. Intake of butylated hydroxyanisole and butylated hydroxytoluene and stomach cancer risk. Am J Clin Nutr. 2000;72(5):1150-1158.
- Robards K, Prenzler PD, Tucker G, et al. Phenolic compounds and their role in oxidative processes in fruits. Food Chem. 1999;66(4):401-436.
- Yamamoto T, Imai T, Hasegawa R, et al. Effects of combined administration of antioxidants on the rat thyroid. Toxicol Lett. 1995;75(1-3):161-168.
- PubMed — BHT butylated hydroxytoluene toxicity search
Connections
- Food Additives
- BHA
- TBHQ
- Sodium Benzoate
- Sulfites
- BHA and BHT
- Trans Fats
- Vitamin E
- Cancer
- Rosemary
- Vitamin C
- Glutathione
- Carrageenan
- Brominated Vegetable Oil
- Azodicarbonamide
- Preservatives
- Potassium Bromate
- Ractopamine