Ractopamine: The Growth Promoter Banned in 160 Countries
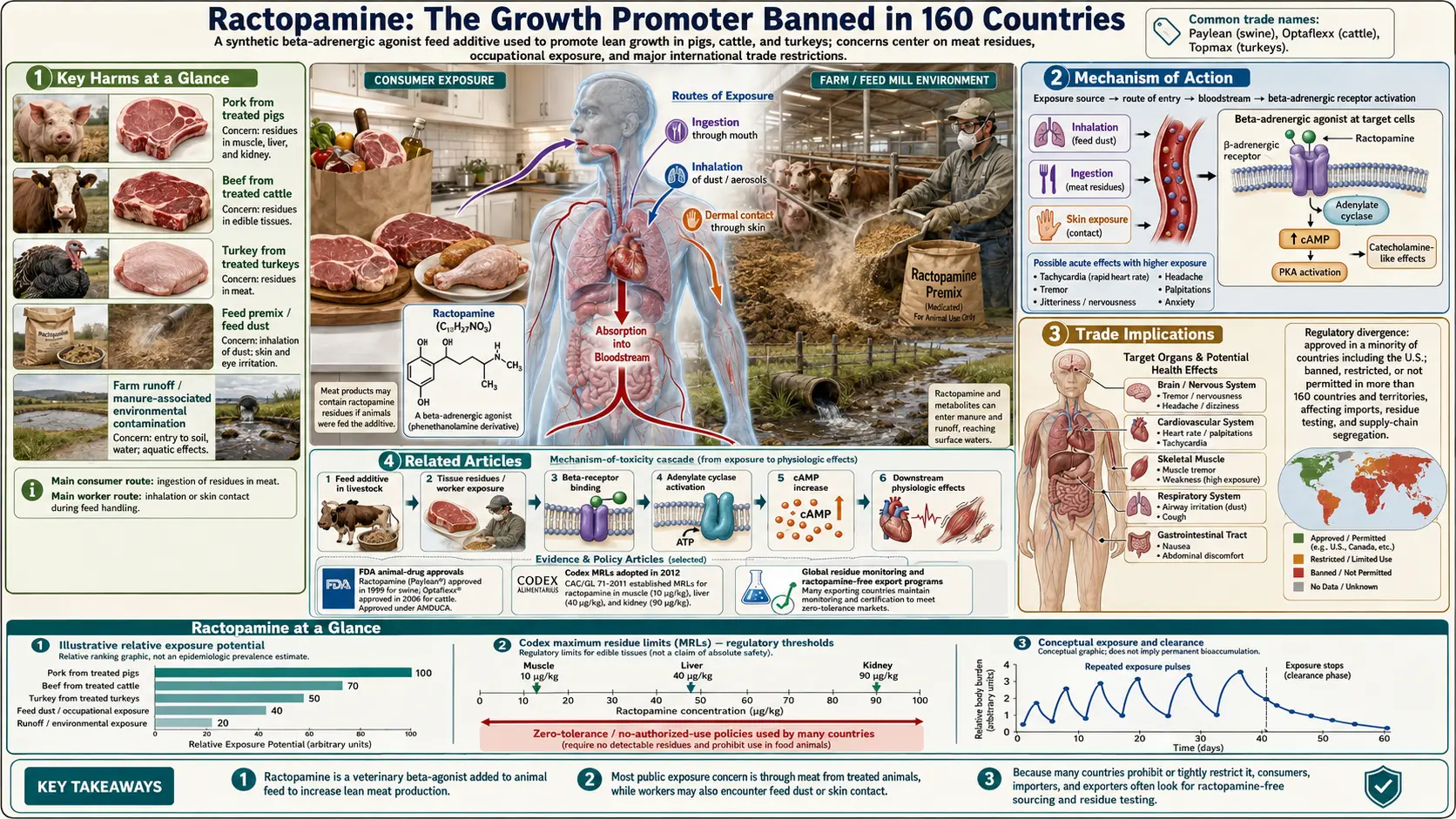
Ractopamine is a synthetic beta-adrenergic agonist drug administered to pigs, cattle, and turkeys in the final weeks before slaughter to shift the metabolism of the animal away from fat deposition and toward lean muscle growth. This process, known as nutrient repartitioning, produces leaner carcasses and larger loin-eye measurements, allowing producers to deliver more lean meat per animal at lower feed cost. Ractopamine was FDA-approved for pigs in 1999 and for cattle in 2003. It is sold under the brand names Paylean (for swine), Optaflexx (for cattle), and Topmax (for turkeys) by Elanco, the animal health division spun off from Eli Lilly. Despite US approval and continued use, ractopamine is banned or severely restricted in more than 160 countries, including every nation of the European Union, China, Russia, Taiwan, and dozens more. It is the single food additive at the center of more bilateral trade disputes than any other substance in modern food history.
Table of Contents
- Overview
- Key Harms at a Glance
- Regulatory History (Safety)
- Mechanism of Action
- Health Effects
- Residues in Meat (Exposure Routes)
- Trade Implications
- Animal Welfare
- Alternatives (How to Avoid)
- Related Articles
- Research Papers and References
- Connections
- Featured Videos
Key Harms at a Glance
- Cardiovascular effects — ractopamine residues can produce tachycardia, palpitations, tremor, and elevated blood pressure in sensitive consumers via beta-adrenergic activation.
- Banned in 160+ countries — the European Union, China, Russia, Taiwan, and most other jurisdictions prohibit ractopamine as a feed additive and bar imports of ractopamine-fed meat.
- Narrow safety margin — the single human volunteer study used by the FDA enrolled only six subjects, one of whom withdrew; JECFA's acceptable daily intake has been contested.
- Residues in pork and beef — measurable residues persist in muscle, liver, and kidney of treated animals at slaughter.
- Animal welfare harms — injuries, inability to walk, and "downer pigs" are documented in slaughterhouse records for ractopamine-fed animals.
- Especially concerning in cardiovascular disease — beta-agonist exposure is contraindicated in patients with arrhythmias or ischemic heart disease.
1. Overview
- Drug class — beta-adrenergic agonist (beta-agonist), specifically acting on both beta-1 and beta-2 adrenergic receptors. The beta-agonist drug class includes medications used to treat asthma and heart conditions in humans (e.g., salbutamol/albuterol, clenbuterol). Ractopamine is structurally related to these drugs.
- Brand names and species — Paylean (swine), Optaflexx (cattle), Topmax (turkeys). All three formulations contain ractopamine hydrochloride as the active ingredient, manufactured by Elanco Animal Health (formerly a division of Eli Lilly and Company).
- Manufacturer — Eli Lilly conducted the original research and sponsored the FDA new animal drug applications. The animal health portfolio, including ractopamine, was spun off into Elanco Animal Health, which completed its separation from Eli Lilly and became an independent company in 2019.
- Mechanism of growth effect — ractopamine acts as a nutrient repartitioning agent by stimulating beta-adrenergic receptors in muscle and adipose (fat) tissue, increasing protein synthesis in muscle and simultaneously stimulating lipolysis (fat breakdown) in adipose tissue. The net result is accelerated lean muscle accumulation and reduced fat deposition without a proportional increase in feed intake.
- Production benefit — lean meat yield increases of 5 to 10 percent have been documented in clinical trials. In swine, studies have shown increases of 24 to 34 percent in loin eye area and reductions in back fat of 5 to 10 percent compared to untreated controls receiving the same feed.
- FDA approvals — Paylean (swine) was approved by the FDA's Center for Veterinary Medicine on December 22, 1999. Optaflexx (cattle) was approved on August 22, 2003. Topmax (turkeys) was approved in 2008. All three approvals established pre-slaughter withdrawal periods and maximum use concentrations.
- Administration — ractopamine is mixed into animal feed and administered in the final 28 to 42 days of the finishing period before slaughter. It is not injected; it is consumed daily in feed.
2. Regulatory History
United States: Approval and Limited Oversight
- FDA approval process — Eli Lilly's new animal drug applications were supported by safety studies conducted primarily by or on behalf of the company. The FDA reviewed these studies and concluded ractopamine was safe for animals and that residues in edible tissues did not pose a human health risk at the concentrations arising from approved use.
- Human safety basis — the FDA's human food safety assessment was based primarily on studies submitted by Eli Lilly. Critics noted that human pharmacological data came from a very small number of subjects (as few as 6 in some studies) and that long-term safety data were not required before approval.
- No mandatory testing — unlike many veterinary drug residues, USDA does not conduct routine systematic testing of US meat for ractopamine residues. Testing rates have historically been below 1 percent of pork production, creating a monitoring gap that other countries have cited as a reason they cannot accept US pork.
European Union Ban
- EU Directive 96/22/EC — the EU banned the administration of beta-agonists (including ractopamine) to food-producing animals for growth promotion under Council Directive 96/22/EC, enacted in 1996. This predated ractopamine's US approval. The directive was strengthened by Directive 2003/74/EC and later replaced by Regulation (EU) No 470/2009. The ban is absolute: no EU member state may approve any beta-agonist for growth promotion, and no meat from animals treated with ractopamine may be legally marketed within the EU.
- Scientific basis — the European Food Safety Authority (EFSA) reviewed ractopamine as part of its assessment of the Codex Alimentarius MRL proposal and concluded in 2009 that a safe dietary exposure could not be established for certain population subgroups, particularly people with pre-existing cardiovascular conditions, and that existing data were insufficient to set a health-based guidance value.
China
- 2002 ban — China banned ractopamine in 2002 following multiple incidents of consumer illness attributed to ractopamine residues in pork. Chinese health authorities documented hundreds of hospitalizations linked to beta-agonist-contaminated pork products, including incidents involving clenbuterol (a similar banned drug) and ractopamine. The ban covers all beta-agonists in food animal production.
- Trade leverage — China is the world's largest pork market and a major importer of US beef. China's ractopamine ban has been a persistent irritant in US–China agricultural trade negotiations. China has used the ban as leverage in broader trade discussions, and the US has pressed China to accept Codex standards as a basis for market access.
Russia
- Import ban — Russia suspended imports of US pork and beef products multiple times between 2012 and 2014 over ractopamine concerns, most notably in February 2013. Russia's veterinary and phytosanitary authority (Rosselkhoznadzor) cited the absence of verified ractopamine-free certification from US exporters as grounds for the suspensions. The ban contributed to significant losses for US pork exporters.
Taiwan
- 2012 protests — Taiwan had banned ractopamine in pork since 2006. In 2012, the Taiwan government proposed allowing ractopamine residues in beef imports under Codex MRL standards. The proposal triggered large public protests and street demonstrations, reflecting intense consumer concern about ractopamine safety. Tens of thousands of demonstrators took to the streets in what became known as the "ractopamine protests." Taiwan's parliament voted to ban imports of beef with any detectable ractopamine residue, creating a legal standoff with the US government.
- 2020 reversal — after years of US trade pressure, Taiwan's president Tsai Ing-wen announced in August 2020 that Taiwan would allow imports of US pork and beef containing ractopamine residues within Codex MRL limits, a decision seen as a political concession to secure a bilateral trade framework with the US. The decision provoked renewed protests and legislative challenges.
Codex Alimentarius: The Most Contested Vote in Codex History
- JECFA evaluation — the Joint FAO/WHO Expert Committee on Food Additives (JECFA) evaluated ractopamine in 2004 and 2010. The 2010 JECFA assessment established an acceptable daily intake (ADI) of 0–1 microgram per kilogram of body weight per day and proposed Maximum Residue Limits (MRLs) for muscle, fat, liver, and kidney of pigs and cattle. The assessment was based substantially on data provided by Elanco and was contested by the EU and other nations as scientifically inadequate.
- 2012 Codex vote — at the 35th session of the Codex Alimentarius Commission in Rome in July 2012, member states voted on whether to adopt the JECFA-proposed MRLs for ractopamine. The vote was 69 in favor to 67 against, with 7 abstentions — the narrowest vote margin in Codex history. The US, Canada, and a number of developing nations voted in favor; the EU, China, Russia, and most Asian and African nations voted against. Critics argued the vote was a political result rather than a scientific consensus, given the near-even split among member states.
- Significance — the adoption of Codex MRLs for ractopamine gave the US a basis to argue at the WTO that countries banning ractopamine-treated meat imports were acting inconsistently with international food safety standards. Countries that maintain more restrictive standards than Codex must justify them with their own scientific risk assessments under WTO rules.
3. Mechanism of Action
Understanding the pharmacology of ractopamine is essential to evaluating both its intended effects in animals and its potential adverse effects in humans who consume residues in meat.
- Beta-adrenergic receptor agonism — ractopamine binds to and activates beta-1 and beta-2 adrenergic receptors, which are components of the sympathetic nervous system. Beta-1 receptors are predominantly located in the heart and kidney; beta-2 receptors are found in smooth muscle of the bronchi, blood vessels, uterus, and gastrointestinal tract, as well as in skeletal muscle and adipose tissue.
- Structural similarity to asthma medications — ractopamine is structurally similar to salbutamol (albuterol), the most commonly prescribed short-acting bronchodilator for asthma. Clenbuterol, another beta-agonist that is banned worldwide as a growth promoter but is misused illegally, is in the same drug class. The fact that ractopamine belongs to a pharmacological class whose human drugs carry cardiovascular warnings is central to the safety debate.
- Cardiovascular effects — beta-1 receptor activation in the heart produces positive chronotropic effects (increased heart rate, tachycardia) and positive inotropic effects (increased force of contraction). In humans, pharmacological doses of beta-1 agonists cause tachycardia, palpitations, and elevated blood pressure. The question for food safety is whether ractopamine residues in meat are sufficient to produce these effects in consumers, particularly those with pre-existing cardiovascular disease.
- Muscle protein synthesis — beta-2 receptor stimulation in skeletal muscle activates cAMP-dependent pathways that upregulate protein synthesis and downregulate protein degradation (proteolysis). The net effect is hypertrophy of muscle fibers, particularly fast-twitch (Type II) fibers, producing the enlarged lean muscle mass seen in treated animals.
- Lipolysis in adipose tissue — simultaneous beta-receptor stimulation in fat tissue activates hormone-sensitive lipase, releasing fatty acids from triglyceride stores. This reduces fat deposition and mobilizes stored fat, contributing to the leaner body composition seen in treated animals.
- Duration of action — ractopamine has a relatively short half-life in animals; however, it is administered continuously in feed throughout the finishing period, maintaining a persistent pharmacological effect for weeks before slaughter. Residues accumulate in edible tissues, with the highest concentrations in liver, kidney, and gastrointestinal tract rather than muscle.
4. Health Effects
Cardiovascular Effects in Humans
- Known pharmacology — as a beta-adrenergic agonist, ractopamine's drug class is inherently cardiovascular-active. Human drugs in the same class carry prescribing warnings about tachycardia, palpitations, hypertension, and risk of cardiac arrhythmia. The human safety question for ractopamine is one of dose: are residue levels in meat sufficient to cause these effects?
- Limited human studies — only a small number of human pharmacokinetic and pharmacodynamic studies of ractopamine have been conducted, most involving very small numbers of healthy adult volunteers and using doses higher than dietary residue exposures. These studies demonstrated measurable cardiovascular effects at pharmacological doses but could not reliably characterize the risk from chronic low-level dietary exposure.
- EFSA's assessment gap — the European Food Safety Authority (EFSA), in its 2009 opinion on ractopamine, concluded that available human data were insufficient to establish a health-based guidance value and that existing studies could not rule out cardiovascular risk in sensitive subpopulations — particularly individuals with cardiovascular disease, the elderly, and children. This scientific gap was the formal basis for the EU's refusal to adopt the Codex MRLs.
FDA Adverse Event Reports
- 218,000+ pig adverse event reports — the FDA Adverse Event Reporting System for veterinary products recorded more adverse events for ractopamine (Paylean) than for any other livestock drug on the market. By 2011, over 218,000 reports of adverse events in pigs had been filed, including reports of trembling, staggering, inability to rise, inability to walk, hyperactivity, death, and cardiovascular stress. This figure represented the highest adverse event burden in the FDA veterinary adverse event database for any single drug.
- Cattle adverse events — adverse events for Optaflexx in cattle included similar stress-related signs: agitation, inability to rise, leg disorders, and deaths. The sheer number of reports led consumer groups and veterinary scientists to petition the FDA to re-evaluate the approval.
- FDA response — rather than withdrawing approval, the FDA required Elanco to add a Cautionary Statement to Paylean's label in 2002, warning about signs of stress in treated pigs. The label addition acknowledged the adverse event problem while leaving the drug on the market.
China Poisoning Incidents
- Beta-agonist food poisonings — China documented hundreds of cases of human illness attributed to beta-agonist residues in pork between 1998 and 2006. Symptoms reported included tachycardia, palpitations, tremors, dizziness, headache, nausea, and vomiting — consistent with the pharmacological effects of beta-agonist ingestion. While many Chinese incidents involved clenbuterol (a more potent and longer-acting banned beta-agonist), ractopamine was also implicated and its pharmacological mechanism is similar.
- Zhangzhou incident (2006) — approximately 330 people were hospitalized in Zhangzhou, Fujian Province after consuming pork contaminated with ractopamine. This incident accelerated enforcement of China's beta-agonist ban and contributed to China's refusal to accept Codex MRLs for ractopamine.
5. Residues in Meat
The presence of ractopamine residues in commercially sold US meat is not disputed. The disagreements are about testing frequency, residue levels, and whether those levels pose health risks to consumers.
- Tissue distribution — ractopamine residues are highest in liver and kidney, followed by the gastrointestinal tract, with lower concentrations in skeletal muscle. The Codex MRLs established in 2012 set different limits for each tissue type: 10 μg/kg for swine muscle, 40 μg/kg for swine liver, 90 μg/kg for swine kidney, 10 μg/kg for bovine muscle, 40 μg/kg for bovine liver.
- Withdrawal periods — the FDA-approved label for Paylean specifies a zero-day pre-slaughter withdrawal period for pigs. This means pigs may be slaughtered on the same day they last received ractopamine in their feed, maximizing residue levels in edible tissues at time of slaughter. Optaflexx for cattle also specifies a short withdrawal period (28 days). The zero-day pork withdrawal period is particularly significant because residues will be at maximum concentrations.
- USDA testing rate — the USDA Food Safety and Inspection Service (FSIS) tests less than 1 percent of US pork for ractopamine residues under its National Residue Program. Consumer and industry groups have noted this is inadequate to provide meaningful assurance of residue compliance, particularly given the zero-day withdrawal period.
- Liver and organ meat exposure — consumers who regularly eat organ meats (liver, kidney) from pigs or cattle receive substantially higher ractopamine exposure than those who consume only muscle meat. Given that organ meats are consumed in higher proportions by some demographic groups (low-income consumers, certain immigrant communities), residue exposure is not uniformly distributed across the population.
- Export certification problem — because most US pigs are not segregated by ractopamine treatment status and testing is minimal, US pork exporters have faced difficulties providing ractopamine-free certification required by importing countries. Some large pork producers have developed ractopamine-free supply chains specifically for export markets, while domestic supply may still contain residues.
6. Trade Implications
Ractopamine is at the center of some of the most consequential food safety trade disputes in recent history. The US has consistently treated the issue as a trade barrier question rather than a food safety question, while importing countries have framed it as consumer protection.
- Russia–US pork disputes — Russia suspended US pork imports on multiple occasions beginning in 2012, citing failure to provide ractopamine-free certification. The Rosselkhoznadzor stated that US veterinary certificates did not guarantee that pork was free from ractopamine residues and that Russia could not accept pork without such guarantees. The US State Department and USTR pressed Russia to accept Codex MRLs as the basis for trade; Russia declined, citing its own risk assessment authority.
- China beef and pork restrictions — China's ban on ractopamine has been a persistent barrier to US beef and pork exports since 2002. US agricultural negotiators have repeatedly pressed China to liberalize access for ractopamine-treated products, and ractopamine featured in the Phase One trade deal negotiations under the Trump administration (2018–2019). China agreed to allow imports of US beef containing ractopamine residues within Codex limits in 2020 as part of trade deal commitments, but implementation has been inconsistent.
- Taiwan protests and policy reversal — Taiwan's 2020 decision to accept ractopamine in US pork and beef was widely seen as a political quid pro quo for improved US–Taiwan relations rather than a science-based food safety decision. Taiwan's public health community and consumer groups opposed the change, arguing that the EFSA assessment (finding insufficient data to establish safe levels) was more scientifically rigorous than the JECFA assessment used to support the Codex MRLs.
- WTO implications — the narrow 2012 Codex vote adopting MRLs gave the US a legal foundation to challenge import bans at the WTO under the Agreement on Sanitary and Phytosanitary Measures (SPS Agreement). The SPS Agreement requires WTO members maintaining food safety measures more restrictive than Codex standards to justify them with their own risk assessments. The EU, with its comprehensive EFSA opinion, is better positioned to defend its ban than some smaller countries.
- 160+ countries banned — the breadth of the international ban — encompassing the EU's 27 member states, China, Russia, the United Kingdom, most of East Asia, most of Latin America, and the majority of Africa — reflects a global regulatory consensus that ractopamine's safety profile is insufficiently established for use in food animal production. The US, Canada, Mexico, Brazil, and Australia are among the small minority of countries that permit its use.
7. Animal Welfare
The animal welfare consequences of ractopamine use are among the most extensively documented aspects of the drug's adverse effects. The FDA's own adverse event database and the drug's own label warnings make clear that ractopamine causes significant animal suffering.
- Stress behaviors in pigs — ractopamine-treated pigs exhibit increased stress behaviors including aggression, hyperactivity, restlessness, and vocalizations associated with distress. Research by Marchant-Forde et al. (2003) in the Journal of Animal Science documented significantly increased activity and aggression in ractopamine-treated swine, consistent with the pharmacological effect of sustained beta-adrenergic stimulation (a state resembling a chronic fight-or-flight response).
- Musculoskeletal injury — the rapid muscle hypertrophy induced by ractopamine can outpace the development of supporting skeletal structures, leading to increased rates of lameness, fractures, and joint disorders. The disproportionate muscle mass relative to bone density creates structural vulnerabilities. FDA adverse event reports documented thousands of cases of pigs collapsing, breaking limbs, or becoming unable to rise during transport to slaughter.
- Downer animals — "downer" pigs and cattle — animals that become unable to stand or walk — were among the most commonly reported adverse events in the FDA adverse event database for ractopamine. Downer animals suffer significantly and create animal welfare problems at loading facilities and slaughterhouses.
- Cardiovascular stress during transport — the combination of ractopamine-induced cardiovascular stimulation and the physical and psychological stress of transport to slaughter can produce transport mortality and pre-slaughter deaths. Industry studies have documented that ractopamine-treated pigs have higher transport mortality rates than untreated pigs.
- FDA label warnings — the current Paylean label explicitly warns: "Not for use in breeding swine" and includes cautionary language about use in pigs experiencing stress, lameness, or injury. The label instructs producers to "observe animals carefully" and to "provide adequate space and access to water." The label also warns against "sudden changes in management" during ractopamine treatment. These warnings acknowledge that the drug places animals in a physiologically stressed state that is incompatible with normal handling conditions.
- Comparison with EU practices — European pork producers, who have operated without ractopamine since the EU ban, do not report competitive disadvantage from its absence. EU swine welfare standards are more broadly protective and the production system does not rely on pharmaceutical shortcuts to maximize lean yield.
8. Alternatives
The US pork and beef industries have operated with ractopamine for over two decades, but a substantial portion of world meat production demonstrates that high-quality, competitive meat can be produced without beta-agonist growth promoters.
- Ractopamine-free production for export — several major US pork producers have developed parallel ractopamine-free supply chains specifically to service export markets that require certification. Smithfield Foods, Tyson Foods, and Cargill all maintain ractopamine-free programs for designated export lines, demonstrating that ractopamine-free production at industrial scale is operationally feasible in the US.
- EU and organic production — the European Union produces high-quality pork and beef competitively without any beta-agonist growth promoters. EU regulations prohibit not just ractopamine but all beta-agonist growth promoters under Directive 96/22/EC. EU producers achieve comparable lean yields through genetics, nutrition, and management rather than pharmacological intervention.
- Genetic selection — modern pig genetics (particularly Pietrain, Hampshire, and Duroc crosses used in high-lean production systems) have been selected for lean muscle yield to a degree that substantially reduces or eliminates the productivity gap between ractopamine-treated and untreated animals. Genetic improvement has been the primary productivity driver in non-ractopamine markets.
- Nutritional optimization — precision feeding programs that match amino acid intake (particularly lysine) to the animal's actual protein deposition capacity at each stage of the growth curve can approach the lean yield benefits of ractopamine without pharmaceutical intervention. Phase feeding programs are widely used in EU and Canadian production.
- Longer growing periods — in the absence of growth promoters, animals can reach the same final slaughter weight with a modest increase in days on feed. The economic cost of this increase is partially offset by avoiding the cost of ractopamine itself, lower veterinary costs from reduced drug-associated morbidity, and access to premium export and domestic markets that exclude ractopamine-treated products.
- Consumer demand — consumer demand for ractopamine-free meat is growing in the United States, driven by both the international ban narrative and general consumer trends toward minimally processed, antibiotic-free, and hormone-free meat products. Major retailers including Whole Foods Market prohibit ractopamine in their meat supply chains, and several natural pork brands market ractopamine-free as a selling point.
9. Related Articles
- Food Additives Overview
- Banned Additives: US vs. the World
- rBGH/rBST: Recombinant Bovine Growth Hormone in US Dairy
10. Research Papers and References
Key research papers, government dossiers, and authoritative sources. Links resolve to DOI, PubMed, or the official agency document.
- US Food and Drug Administration. NADA 140-863 (Paylean) Original Approval. Center for Veterinary Medicine; December 22, 1999. FDA ADAFDA
- US Food and Drug Administration. NADA 141-204 (Optaflexx) Approval Summary. Center for Veterinary Medicine; August 22, 2003. FDA Animal Veterinary
- European Food Safety Authority (EFSA). Scientific opinion on the assessment of the potential hazards to human health and the environment of ractopamine. EFSA Journal. 2009;7(9):1244.
- Joint FAO/WHO Expert Committee on Food Additives (JECFA). Toxicological evaluation of certain veterinary drug residues in food: ractopamine. WHO Technical Report Series 966. Geneva: WHO; 2012. WHO publication
- Codex Alimentarius Commission. Report of the 35th Session. Rome, July 2–7, 2012. ALINORM 12/35/REP. FAO; 2012.
- Marchant-Forde JN, Lay DC Jr, Pajor EA, Richert BT, Schinckel AP. The effects of ractopamine on the behavior and physiology of finishing pigs. J Anim Sci. 2003;81(2):416–422.
- Carr SN, Rincker PJ, Killefer J, Baker DH, Ellis M, McKeith FK. Effects of different cereal grains and ractopamine hydrochloride on performance, carcass characteristics, and fat quality in late-finishing pigs. J Anim Sci. 2005;83(1):223–230.
- Council of the European Union. Council Directive 96/22/EC concerning the prohibition on the use in stockfarming of certain substances having a hormonal or thyrostatic action and of beta-agonists. Official Journal of the European Communities. 1996;L 125:3–9.
- Lawrence BV, Schinckel AP, Adeola O, Cera K. Impact of ractopamine on pig growth performance and indices of physiological well-being. J Anim Sci. 2002;80(10):2690–2696.
- Aalhus JL, Schaefer AL, Murray AC, Jones SD. The effect of ractopamine on myofibre distribution and morphology and their relation to meat quality in swine. Meat Sci. 1992;31(4):397–409.
- Weber TE, Richert BT, Belury MA, Gu Y, Enright K, Schinckel AP. Evaluation of the effects of dietary fat, conjugated linoleic acid, and ractopamine on growth performance, pork quality, and fatty acid profiles in genetically lean gilts. J Anim Sci. 2006;84(3):720–732.
- Smith DJ. The pharmacokinetics, metabolism, and tissue residues of beta-adrenergic agonists in livestock. J Anim Sci. 1998;76(1):173–194.
- Huang B, Du X, Li Y, Yu A, Xu L. Residues of ractopamine and clenbuterol in swine tissues and their implication for food safety. Food Chem. 2013;141(3):2116–2122.
- Gonzalez-Esquerra R, Leeson S. Effects of menhaden oil and flaxseed in broiler diets on sensory quality and lipid composition of poultry meat. Br Poult Sci. 2000;41(4):481–488.
- National Pork Board. Pork Quality Assurance Plus Program Guidelines. Des Moines, IA: National Pork Board; 2015. pork.org
- USDA Food Safety and Inspection Service. 2019 National Residue Program Data. Washington, DC: USDA FSIS; 2020. FSIS publication
Connections
- Food Additives
- rBGH
- Banned Additives
- Pork
- Beef
- Hypertension
- Atrial Fibrillation
- Cardiovascular Disease
- Asthma
- Sodium Nitrite
- Glyphosate
- Organ Meats
- Arrhythmia
- BHA
- BHT