Fluoride in Water
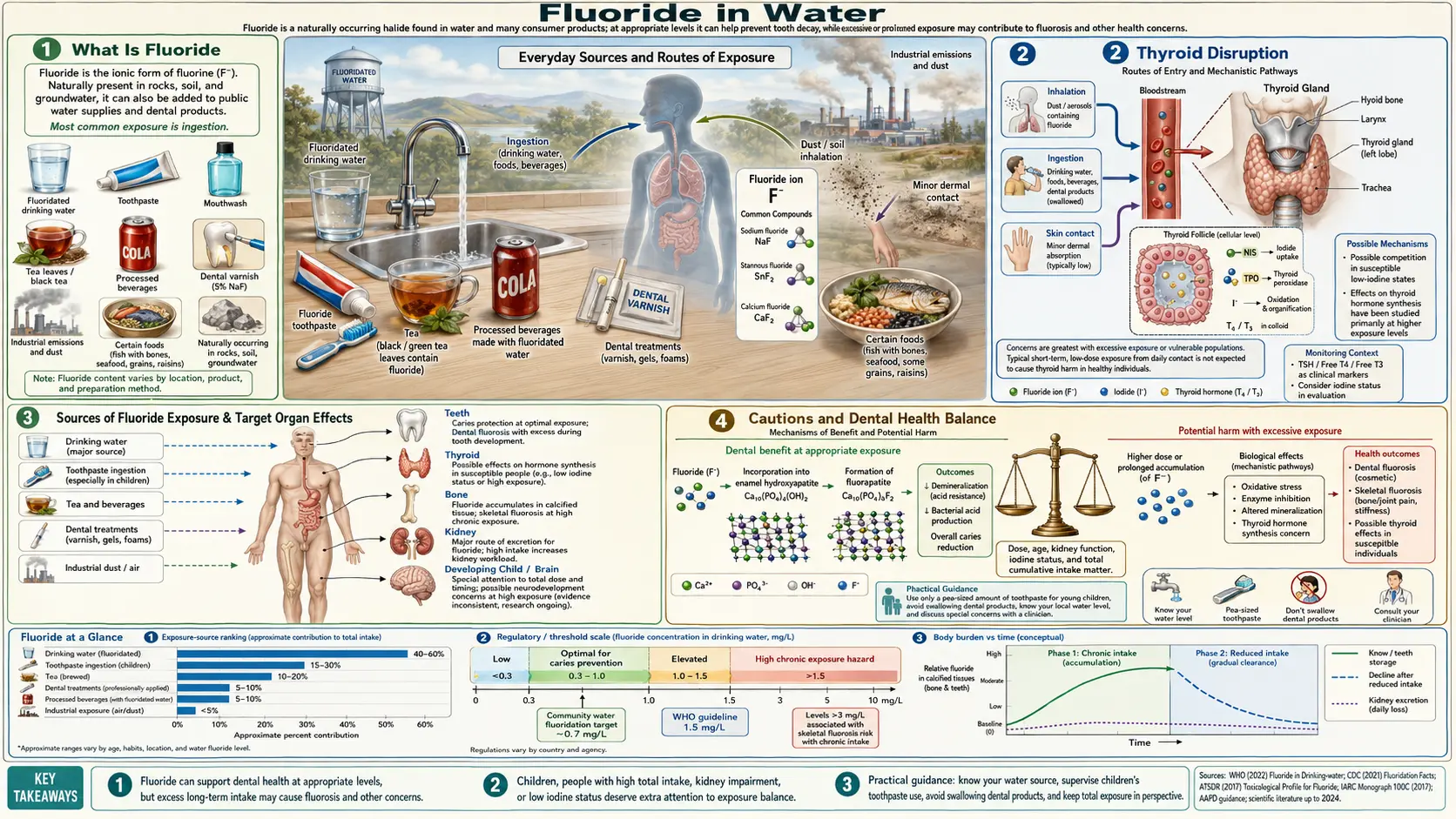

Fluoride is one of the most controversial substances deliberately added to public drinking water. Proponents argue it prevents tooth decay at minimal cost, while a growing body of scientific evidence raises serious concerns about its effects on the brain, thyroid, bones, kidneys, and pineal gland. From a naturopathic perspective, the mass addition of a pharmacologically active substance to the water supply, without individual dosage control, informed consent, or consideration of total exposure from all sources, represents a significant public health concern that deserves careful scrutiny.
The debate over water fluoridation has intensified in recent years as peer-reviewed research from institutions including Harvard University and The Lancet has classified fluoride as a developmental neurotoxicant, placing it alongside lead, mercury, and arsenic. Meanwhile, 97% of Western Europe has chosen not to fluoridate its water, and several communities across North America have voted to end the practice. Understanding the full scope of fluoride exposure, its documented health effects, and practical strategies for reducing intake is essential for making informed decisions about your health.
Table of Contents
- What Is Fluoride
- History of Water Fluoridation
- The Dental Health Argument
- The Consent Controversy
- Neurotoxicity and IQ Reduction
- Thyroid Disruption
- Skeletal Fluorosis
- Pineal Gland Calcification
- Kidney Damage
- Sources of Fluoride Exposure
- Countries That Have Banned or Rejected Fluoridation
- How to Reduce Fluoride Exposure
- Fluoride-Free Toothpaste Alternatives
- Testing Your Water
- Cautions and Dental Health Balance
- Connections
- Featured Videos
What Is Fluoride
Fluoride is the ionic form of fluorine, the most electronegative and reactive of all elements on the periodic table. While fluoride occurs naturally in some water sources and mineral deposits, the forms of fluoride used in water treatment and dental products vary significantly in their origin, purity, and potential toxicity.
- Calcium fluoride (CaF2): This is the naturally occurring form found in soil, rocks, and some groundwater sources. It is relatively insoluble and poorly absorbed by the body. When fluoride advocates point to naturally occurring fluoride in water, they are typically referring to calcium fluoride. This form has the lowest toxicity profile of the common fluoride compounds
- Sodium fluoride (NaF): A synthetic compound originally produced as an industrial waste product of aluminum manufacturing. Sodium fluoride is the form used in most fluoride toothpastes and was the original compound used in early water fluoridation programs. It is far more bioavailable than calcium fluoride, meaning the body absorbs it more readily. Sodium fluoride is also used commercially as a pesticide and rat poison
- Hydrofluorosilicic acid (H2SiF6): Also known as fluorosilicic acid or hexafluorosilicic acid, this is the form of fluoride most commonly used in modern water fluoridation programs in the United States. It is an unpurified industrial waste product captured from the phosphate fertilizer manufacturing process. Before being sold to water treatment facilities, this substance would otherwise need to be disposed of as hazardous waste at significant cost. It contains trace contaminants including arsenic, lead, and other heavy metals. Approximately 90% of fluoridated water systems in the United States use hydrofluorosilicic acid
The distinction between these forms matters greatly. The claim that fluoridation simply adjusts naturally occurring fluoride levels to an optimal concentration obscures the fact that the industrial compounds added to water are chemically and toxicologically different from the calcium fluoride found in nature.
History of Water Fluoridation
The history of water fluoridation in the United States is intertwined with industrial interests, wartime research, and a public health paradigm that prioritized population-level intervention over individual choice.
- Early observations (1900s-1930s): In the early twentieth century, dentist Frederick McKay noticed that residents of Colorado Springs with brown-stained teeth (dental fluorosis) had fewer cavities. Research by H.V. Churchill at the Aluminum Company of America (ALCOA) identified fluoride in the water as the cause of the staining. ALCOA, which produced massive quantities of fluoride waste from aluminum smelting, had a financial interest in finding a beneficial use for this industrial byproduct
- Grand Rapids experiment (1945): On January 25, 1945, Grand Rapids, Michigan became the first city in the world to add fluoride to its public water supply. The experiment was intended to run for 15 years with nearby Muskegon serving as an unfluoridated control city. However, Muskegon began fluoridating its water before the study was completed, compromising the control data. Despite this, the experiment was declared a success and fluoridation spread rapidly across the country
- Rapid adoption (1950s-1960s): The American Dental Association and the U.S. Public Health Service endorsed water fluoridation in 1950, before the Grand Rapids study was completed. By the mid-1960s, thousands of American communities had adopted fluoridation, often over the objections of citizens who questioned the safety and ethics of the practice
- Industrial connections: Declassified documents have revealed that much of the early fluoride safety research was conducted by scientists with ties to the Manhattan Project and the nuclear weapons industry, which used massive quantities of fluoride in uranium enrichment. There were significant financial incentives to establish fluoride as safe rather than acknowledge it as a toxic pollutant requiring expensive disposal and cleanup
The Dental Health Argument
The primary justification for water fluoridation has always been its purported ability to reduce tooth decay. While there is evidence that topical application of fluoride can strengthen tooth enamel, the case for systemic ingestion through drinking water is considerably weaker than commonly believed.
- Topical versus systemic: The original theory behind water fluoridation was that fluoride needed to be ingested during tooth development to be incorporated into the enamel structure. Modern dental research has largely abandoned this position, with the CDC itself acknowledging that the primary benefit of fluoride is topical, meaning it works on the surface of teeth after they have erupted. This raises the fundamental question of why fluoride needs to be swallowed at all
- Declining cavity rates everywhere: Tooth decay rates have declined dramatically in all developed countries over the past several decades, regardless of whether they fluoridate their water. Countries that do not fluoridate, such as Germany, Denmark, the Netherlands, and Japan, have experienced the same or greater reductions in cavities as fluoridated countries like the United States, suggesting that improved dental hygiene, nutrition, and access to dental care are the primary drivers
- Dental fluorosis: The CDC reports that approximately 65% of American adolescents now show signs of dental fluorosis, the white spots, streaks, and in severe cases pitting and brown staining of teeth caused by excessive fluoride exposure during tooth development. This dramatic increase from 10% in the 1950s indicates that total fluoride intake has far exceeded the levels originally intended
- Diminishing returns: Even studies that find a benefit from fluoridation report modest effects, typically a reduction of less than one cavity per person. The 2015 Cochrane Review, considered the gold standard for evidence-based analysis, found that the evidence supporting water fluoridation was of low quality and that most studies were conducted before the widespread availability of fluoride toothpaste, making their relevance to modern conditions questionable
The Consent Controversy
Perhaps the most fundamental objection to water fluoridation is the ethical issue of mass medication without individual consent. This concern is shared by many medical professionals, ethicists, and legal scholars regardless of their position on fluoride's safety.
- Mass medication without consent: Water fluoridation is the only instance in which a pharmacologically active substance is added to the public water supply for the purpose of treating a medical condition (tooth decay) in the population. Unlike chlorine, which is added to purify the water itself, fluoride is added to treat the people who drink it. This constitutes mass medication without individual informed consent
- No dose control: When fluoride is added to the water supply, there is no way to control the dose that any individual receives. A manual laborer who drinks two gallons of water per day receives a dramatically different dose than an elderly person who drinks two cups. Infants fed formula mixed with fluoridated water receive doses per body weight that far exceed recommended levels. There is no other medication administered this way
- Vulnerable populations: Water fluoridation cannot account for individual susceptibility. People with kidney disease, thyroid disorders, diabetes, and other conditions may be more vulnerable to fluoride's effects. Infants, the elderly, and those with nutritional deficiencies including iodine deficiency are at increased risk
- Violation of medical ethics: The principles of medical ethics require informed consent before administering any treatment. Water fluoridation violates this principle by forcing every person who drinks or bathes in public water to receive a dose of fluoride whether they want it or not and regardless of their individual health status
Neurotoxicity and IQ Reduction
The neurotoxic effects of fluoride have become one of the most active and concerning areas of fluoride research. A substantial and growing body of evidence links fluoride exposure to reduced cognitive function, particularly in children.
- Harvard meta-analysis (2012): A systematic review and meta-analysis conducted by researchers at the Harvard School of Public Health, published in Environmental Health Perspectives, analyzed 27 studies and found that children living in areas with high fluoride exposure had significantly lower IQ scores than those in low-fluoride areas. The average IQ difference was approximately 7 points, a difference with profound implications at the population level
- Lancet neurotoxicant classification: In 2014, Dr. Philippe Grandjean of Harvard and Dr. Philip Landrigan of Mount Sinai published a landmark review in The Lancet Neurology that classified fluoride as a developmental neurotoxicant, placing it in the same category as lead, mercury, arsenic, toluene, and polychlorinated biphenyls (PCBs). They noted that fluoride can cause neurodevelopmental harm at doses currently considered safe
- NIEHS-funded study (2017): A study funded by the U.S. National Institute of Environmental Health Sciences, published in Environmental Health Perspectives, followed mother-child pairs in Mexico and found that higher prenatal fluoride exposure was associated with lower IQ scores in children at ages 4 and 6-12. Each 0.5 mg/L increase in maternal urinary fluoride was associated with a 3.15-point decrease in IQ in offspring
- Canadian study (2019): A study published in JAMA Pediatrics examining Canadian mother-child pairs found that fluoride exposure during pregnancy was associated with lower IQ scores in boys. Mothers living in fluoridated communities had children with IQ scores averaging 4.5 points lower than those in non-fluoridated communities
- NTP review (2020-2024): The National Toxicology Program conducted a systematic review of fluoride's neurotoxicity and concluded that fluoride is presumed to be a cognitive neurodevelopmental hazard to humans, based on a consistent pattern of findings across human studies at exposure levels relevant to water fluoridation
- Mechanisms of neurotoxicity: Laboratory research has identified multiple mechanisms by which fluoride damages the brain, including oxidative stress, mitochondrial dysfunction, disruption of neurotransmitter systems, inhibition of enzymes critical to brain development, promotion of neuroinflammation, and interference with thyroid hormones essential for brain development
Thyroid Disruption
The thyroid gland is one of the organs most sensitive to fluoride exposure, and the connection between fluoride and thyroid dysfunction has been documented since the mid-twentieth century when fluoride compounds were actually prescribed as anti-thyroid medication.
- Fluoride as anti-thyroid agent: In the 1950s and 1960s, fluoride was used medically to treat hyperthyroidism (overactive thyroid) because of its ability to suppress thyroid function. The doses used therapeutically were comparable to the amounts that many people now receive from fluoridated water and other sources combined
- Iodine displacement: Fluoride belongs to the halogen group on the periodic table, along with iodine, chlorine, and bromine. Because fluoride is a smaller and more electronegative atom than iodine, it can displace iodine from receptor sites in the thyroid gland, effectively blocking the production of thyroid hormones. In populations with marginal iodine intake, this displacement effect is particularly damaging
- Hypothyroidism link: A 2018 study published in the journal Science of the Total Environment found that adults in fluoridated communities had nearly double the rate of hypothyroidism compared to those in non-fluoridated areas. A 2015 study in the Journal of Epidemiology and Community Health similarly found that fluoridated areas in England had significantly higher rates of hypothyroidism
- TSH elevation: Multiple studies have found that fluoride exposure is associated with elevated levels of thyroid-stimulating hormone (TSH), a marker indicating that the thyroid is underperforming and the pituitary gland is working harder to stimulate it
- Subclinical effects: Even subclinical thyroid dysfunction, where TSH is slightly elevated but still within the conventional reference range, has been associated with fatigue, weight gain, depression, cognitive impairment, elevated cholesterol, and increased cardiovascular risk. The widespread nature of fluoride exposure means even modest thyroid-suppressing effects could affect millions of people
Skeletal Fluorosis
Skeletal fluorosis is a bone disease caused by chronic excessive fluoride accumulation in the skeleton. While severe forms are most commonly seen in regions with naturally very high fluoride levels, emerging evidence suggests that milder forms may be more common in fluoridated countries than previously recognized.
- Mechanism: Fluoride is a bone-seeking element. Approximately 50% of absorbed fluoride is deposited in bones and teeth, where it accumulates over a lifetime. As fluoride replaces the hydroxyl groups in hydroxyapatite (the mineral component of bone), it forms fluorapatite, which makes bones more dense but also more brittle
- Stages of skeletal fluorosis: The condition progresses through stages, beginning with joint pain and stiffness (often misdiagnosed as arthritis) and advancing to calcification of ligaments, osteosclerosis (abnormal bone hardening), and in severe cases, crippling skeletal deformity with compression of the spinal cord
- Preclinical stage: The preclinical stage of skeletal fluorosis, characterized by vague joint and bone pain, is virtually indistinguishable from common arthritis and is rarely investigated for fluoride as a potential cause. Researchers have suggested that many cases of unexplained joint and bone pain in fluoridated populations may represent undiagnosed preclinical skeletal fluorosis
- Bone fracture risk: Several studies have found associations between fluoride exposure and increased risk of hip fractures in the elderly, possibly because fluoride makes bones more dense but simultaneously more brittle and less flexible
- Endemic in high-fluoride regions: Skeletal fluorosis affects millions of people in India, China, and parts of Africa where naturally occurring fluoride in groundwater reaches high concentrations. These populations provide sobering examples of what chronic fluoride accumulation does to the human skeleton over decades
Pineal Gland Calcification
The pineal gland, a small endocrine gland in the center of the brain that produces melatonin and regulates sleep-wake cycles, is a major site of fluoride accumulation in the body.
- Fluoride accumulation: Research by Jennifer Luke at the University of Surrey found that the pineal gland accumulates more fluoride than any other soft tissue in the body, with concentrations comparable to those found in bones and teeth. The calcified deposits (corpora arenacea) within the pineal gland act as a magnet for fluoride
- Melatonin disruption: Luke's research in animal studies demonstrated that fluoride accumulation in the pineal gland was associated with reduced melatonin production and earlier onset of puberty. Melatonin is the body's primary sleep hormone and a powerful antioxidant with roles in immune function, cancer protection, and aging
- Sleep implications: Given the pineal gland's role in regulating circadian rhythm and sleep patterns, fluoride-induced pineal gland dysfunction could contribute to the epidemic of sleep disorders affecting modern populations. Impaired melatonin production affects not only sleep quality but also immune surveillance, mood regulation, and overall hormonal balance
- Accelerated calcification: While some pineal calcification occurs naturally with aging, excessive fluoride exposure may accelerate this process. Calcification of the pineal gland has been associated with reduced melatonin output, disrupted sleep patterns, and in some research, increased risk of neurodegenerative diseases including Alzheimer's disease
Kidney Damage
The kidneys are the primary organs responsible for excreting fluoride from the body, making them particularly vulnerable to its toxic effects. Individuals with impaired kidney function are at heightened risk because they retain a larger proportion of ingested fluoride.
- Renal fluoride handling: The kidneys filter and excrete approximately 50% of ingested fluoride. In individuals with reduced kidney function, whether from chronic kidney disease, diabetes, aging, or other conditions, the rate of fluoride excretion is significantly decreased, leading to higher blood levels and greater tissue accumulation
- Direct nephrotoxicity: Animal studies and human case reports have documented fluoride's direct toxic effects on kidney tubular cells. Chronic exposure to fluoride concentrations found in fluoridated water has been shown to cause oxidative stress, inflammation, and structural damage to kidney tissue in laboratory animals
- Chronic kidney disease population: An estimated 37 million Americans have chronic kidney disease, and millions more have reduced kidney function due to aging, diabetes, or hypertension. These individuals cannot efficiently excrete fluoride and may accumulate toxic levels from the same water supply that is considered safe for the general population
- No safe level established: There is no consensus on a safe fluoride level for individuals with compromised kidney function. The National Kidney Foundation withdrew its endorsement of water fluoridation in 2008, citing insufficient evidence of safety for kidney patients
- Dialysis concerns: Dialysis patients are especially vulnerable because of the large volume of water used in hemodialysis. Although dialysis water is typically treated to remove fluoride, the cumulative exposure from drinking fluoridated water between treatments remains a concern
Sources of Fluoride Exposure
One of the most overlooked aspects of the fluoridation debate is the total cumulative fluoride exposure from all sources. When fluoridation was first introduced in 1945, drinking water was essentially the only source of fluoride in the diet. Today, fluoride exposure comes from numerous sources, many of which did not exist when the "optimal" water fluoridation level was established.
- Tap water: Fluoridated municipal water typically contains 0.7 mg/L of fluoride (reduced from the previous range of 0.7-1.2 mg/L in 2015). This includes water used for drinking, cooking, and food preparation. Foods prepared with fluoridated water, including restaurant meals, commercially processed foods, and beverages, contribute to total intake
- Toothpaste: Standard fluoride toothpaste contains 1,000-1,500 ppm of fluoride. A pea-sized amount contains approximately 0.25 mg of fluoride. Young children frequently swallow toothpaste, and even adults absorb some fluoride through the oral mucosa during brushing
- Tea: The tea plant (Camellia sinensis) is a hyperaccumulator of fluoride, absorbing it from soil and concentrating it in its leaves. Black tea can contain 1-6 mg of fluoride per liter, with older leaves and lower-quality teas containing the highest concentrations. Heavy tea drinkers can receive several milligrams of fluoride daily from tea alone
- Pesticides: Cryolite (sodium aluminum fluoride) is a fluoride-based pesticide widely used on grapes, citrus fruits, potatoes, and other crops. Sulfuryl fluoride is used as a fumigant for food storage and processing facilities. Residues of these fluoride-containing pesticides on food contribute to dietary fluoride intake
- Teflon cookware: Polytetrafluoroethylene (PTFE), marketed as Teflon, contains fluoride in its polymer structure. When Teflon-coated cookware is heated to high temperatures, it can release fluoride-containing compounds including perfluorooctanoic acid (PFOA) and other perfluorinated chemicals
- Pharmaceuticals: Approximately 20-30% of all pharmaceuticals contain fluorine in their molecular structure. Fluoxetine (Prozac), the popular antidepressant, contains three fluorine atoms per molecule. Other fluorinated medications include ciprofloxacin (antibiotic), fluticasone (steroid inhaler), voriconazole (antifungal), and many others. While the fluorine in these drugs is organically bound, some is metabolized and released as inorganic fluoride in the body
- Processed beverages: Soft drinks, juices, beer, wine, and other beverages manufactured with fluoridated water contain fluoride. Infant formula reconstituted with fluoridated tap water delivers fluoride doses per body weight that greatly exceed recommended levels for infants
- Dental treatments: Professional fluoride treatments, fluoride varnishes, fluoride mouth rinses, and fluoride supplements prescribed for children all add to total fluoride exposure
- Mechanically deboned meat: The mechanical process used to separate meat from bones in processed meat products can introduce fluoride from bone particles into the final product
Countries That Have Banned or Rejected Fluoridation
Despite the insistence of dental associations in the United States, Canada, and Australia that water fluoridation is safe and effective, the vast majority of the world's developed nations have chosen not to fluoridate their water supplies.
- 97% of Western Europe does not fluoridate: Only Ireland and parts of the United Kingdom and Spain fluoridate water in Western Europe. The rest of Europe, including Germany, France, Italy, the Netherlands, Belgium, Austria, Switzerland, Sweden, Norway, Denmark, and Finland, have either never adopted fluoridation or discontinued the practice after reviewing the evidence
- Countries that stopped fluoridation: Germany, the Netherlands, Sweden, Finland, Japan, and many others tried water fluoridation and subsequently stopped it, citing concerns about safety, efficacy, ethics, or the availability of better alternatives for dental health
- Israel: Israel mandated water fluoridation in 2002 but reversed the mandate in 2014 after a government-appointed committee reviewed the scientific literature and concluded that the evidence did not support the practice
- China and India: Rather than adding fluoride to water, China and India spend significant resources removing naturally occurring fluoride from drinking water because of the devastating health effects of skeletal fluorosis and other fluoride-related diseases in their populations
- Dental health comparisons: The World Health Organization data on tooth decay rates show no consistent advantage for fluoridated countries. Many non-fluoridating European countries have equal or better dental health outcomes compared to the fluoridated United States, suggesting that fluoridation is not a necessary component of effective dental public health
How to Reduce Fluoride Exposure
Reducing fluoride exposure requires understanding which filtration methods actually remove fluoride and which do not. Many people assume their water filter removes fluoride when it does not.
- Reverse osmosis (RO): Reverse osmosis filtration is one of the most effective methods for removing fluoride from drinking water, typically removing 90-95% of fluoride along with many other contaminants. Countertop and under-sink RO systems are widely available and range from moderately to highly affordable depending on the system
- Activated alumina filters: Activated alumina is a highly porous form of aluminum oxide that has a strong affinity for fluoride. Filters using activated alumina can remove 90% or more of fluoride from water. These filters need to be replaced regularly to maintain effectiveness
- Distillation: Water distillation effectively removes fluoride by boiling water and collecting the steam, which leaves fluoride and other dissolved solids behind. Home water distillers are available and produce very pure water, though the process is slow and energy-intensive
- Bone char filters: Bone char carbon, made from animal bones heated to high temperatures, has been used for centuries to remove fluoride from water. It works through both adsorption and ion exchange, removing up to 90% of fluoride
- Carbon filters do NOT remove fluoride: Standard activated carbon filters, including popular pitcher filters such as Brita and PUR, refrigerator filters, and most faucet-mounted filters, do not remove fluoride from water. These filters are effective at removing chlorine, some organic compounds, and improving taste, but fluoride passes through carbon filtration virtually unaffected. This is one of the most common misconceptions about water filtration
- Boiling does not help: Boiling water does not remove fluoride. In fact, boiling concentrates fluoride by reducing the volume of water through evaporation while the fluoride remains behind
- Spring water: Many natural spring water brands contain very low fluoride levels, though concentrations vary by source. Check the water quality report for any bottled water brand to verify fluoride content
- Shower and bath filtration: Fluoride can be absorbed through the skin during bathing and showering. Whole-house filtration systems with activated alumina or bone char media can reduce fluoride exposure from all household water sources
Fluoride-Free Toothpaste Alternatives
For those seeking to reduce fluoride exposure while still maintaining excellent dental health, several effective fluoride-free toothpaste options exist, with hydroxyapatite-based formulations leading the way in evidence-based alternatives.
- Hydroxyapatite (HAp) toothpaste: Hydroxyapatite is the mineral that makes up 97% of tooth enamel and 70% of dentin. Nano-hydroxyapatite toothpaste works by depositing a layer of this biocompatible mineral directly onto teeth, filling microscopic enamel defects and creating a protective barrier. Multiple clinical studies have found hydroxyapatite toothpaste to be comparable to fluoride toothpaste in preventing cavities and remineralizing early enamel lesions. Japan has used hydroxyapatite in oral care products since the 1980s
- Xylitol: This naturally occurring sugar alcohol has been extensively studied for its ability to reduce cavity-causing bacteria. Xylitol inhibits the growth of Streptococcus mutans, the primary bacterium responsible for tooth decay. Many fluoride-free toothpastes include xylitol as an active ingredient
- Baking soda: Sodium bicarbonate has a long history of use in dental care. It creates an alkaline environment that inhibits bacterial growth, gently removes surface stains, and neutralizes acids that cause enamel erosion
- Oil pulling: The traditional Ayurvedic practice of swishing oil (typically coconut or sesame oil) in the mouth for 10-20 minutes has been shown in some studies to reduce bacterial load, plaque, and gingivitis. While not a toothpaste replacement, it can be a valuable addition to a fluoride-free oral care routine
- Diet and nutrition: From a naturopathic perspective, the most important factor in dental health is diet. Reducing sugar and processed carbohydrate intake, ensuring adequate fat-soluble vitamins (A, D, E, and K2), and maintaining sufficient mineral intake (calcium, phosphorus, magnesium) support tooth remineralization from within
Testing Your Water
Knowing the fluoride level in your water is the first step toward making informed decisions about your exposure.
- Consumer Confidence Reports: All public water systems in the United States are required to publish annual water quality reports, also known as Consumer Confidence Reports (CCRs). These reports include fluoride levels and are typically available on your water utility's website or by request
- Home testing kits: Inexpensive fluoride test strips and colorimetric test kits are available for home use. While not as precise as laboratory analysis, they can give a reasonable estimate of fluoride concentration in your water. Digital fluoride meters provide more accurate readings
- Laboratory testing: For the most accurate results, send a water sample to a certified water testing laboratory. Many labs offer comprehensive water quality panels that test for fluoride along with other contaminants including heavy metals, pesticides, and microorganisms. The EPA provides a list of certified laboratories by state
- Well water testing: If you rely on well water, testing is especially important because naturally occurring fluoride levels in groundwater vary dramatically by region. Some wells contain no measurable fluoride while others in certain geological areas may contain fluoride levels well above the EPA's maximum contaminant level of 4 mg/L
- Total exposure assessment: When evaluating your fluoride exposure, remember that drinking water is only one source. Consider your intake from tea, toothpaste, processed foods and beverages, medications, and dental treatments to estimate your total daily fluoride intake
Cautions and Dental Health Balance
While the evidence of fluoride's potential harms warrants serious attention and proactive steps to reduce unnecessary exposure, it is important to approach this topic with nuance and balance.
- Dental health matters: Tooth decay remains a significant health problem, particularly in underserved communities with limited access to dental care. Dismissing all cavity prevention measures without providing effective alternatives would be irresponsible. The goal should be to maintain excellent dental health while minimizing unnecessary fluoride exposure
- Topical benefits acknowledged: The topical application of fluoride to tooth surfaces does appear to have some protective effect against cavities. The objection from a naturopathic perspective is not necessarily to all fluoride use in dentistry but rather to the indiscriminate systemic exposure through the water supply and the lack of informed consent
- Individual risk assessment: The decision about fluoride exposure should be made individually based on personal health history, risk factors, and informed evaluation of the evidence. People with thyroid conditions, kidney disease, or iodine deficiency may have stronger reasons to minimize exposure than others
- Children require special consideration: Given the evidence linking fluoride to neurodevelopmental harm, particular care should be taken to minimize fluoride exposure in pregnant women, infants, and young children. The American Academy of Pediatrics recommends that infant formula be prepared with low-fluoride water
- Comprehensive oral health approach: Rather than relying solely on fluoride for cavity prevention, a comprehensive approach should include reducing sugar intake, maintaining good oral hygiene, ensuring adequate nutrition for tooth mineralization, using hydroxyapatite-based dental products, and receiving regular dental care
- Stay informed: The science on fluoride continues to evolve rapidly. Major court cases, including a 2024 federal lawsuit against the EPA, ongoing NTP reviews, and new epidemiological studies are continually adding to our understanding of fluoride's effects on human health. Staying current with the evidence is essential for making informed health decisions
Connections
- Lead
- Arsenic
- Mercury
- Heavy Metals
- Pesticides
- Iodine
- Calcium
- Thyroid Disorders
- Kidney Disease
- Kidney Function
- Arthritis
- Diabetes
- Depression
- Alzheimers Disease
- Hypertension
- Oxidative Stress
- Fatigue
- Oral Microbiome