Mercury Toxicity: Sources, Health Effects, and Detoxification
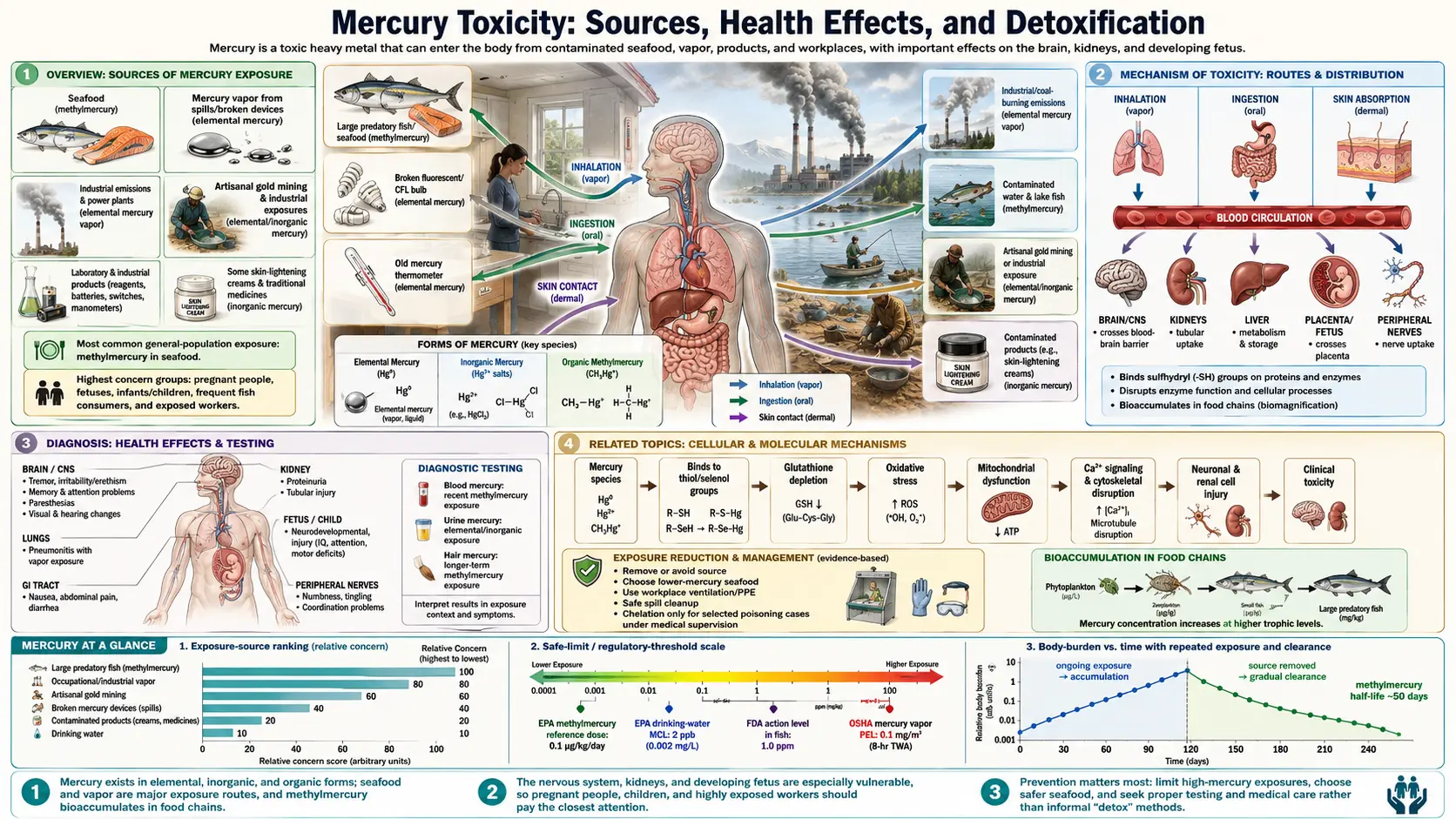


Table of Contents
- Overview
- Sources of Exposure
- Metabolism and Toxicokinetics
- Mechanism of Toxicity
- Health Effects
- Mercury in Seafood
- Dental Amalgam Controversy
- Diagnosis
- Treatment and Detoxification
- Prevention
- Related Topics
- References
- Connections
- Featured Videos
1. Overview
Mercury (Hg) is a naturally occurring heavy metal and one of the most extensively studied environmental neurotoxins. Unlike most metals, mercury exists as a liquid at room temperature and readily volatilizes, contributing to widespread atmospheric distribution. Human activities — particularly coal combustion, artisanal gold mining, and industrial processes — have approximately tripled the mercury burden in shallow ocean waters compared to pre-industrial levels.
Mercury occurs in three chemically and toxicologically distinct forms, each with different exposure routes, bioavailability, and organ targets:
- Elemental mercury (Hg⁰): The metallic liquid form found in thermometers, barometers, and dental amalgam fillings. Poorly absorbed through the gut when ingested, but highly dangerous when inhaled as vapor. Converted to inorganic mercury in the body after absorption.
- Inorganic mercury (Hg²⁺): Mercuric and mercurous salts formed when elemental mercury reacts with other elements. Primarily affects the kidneys. Historically used in antiseptics, laxatives, and hat-making (giving rise to the phrase "mad as a hatter").
- Organic mercury (methylmercury, MeHg): The most neurotoxic form, produced by anaerobic bacteria that methylate inorganic mercury in aquatic sediments. Accumulates efficiently in fish and marine mammals and is the predominant form of human exposure through diet. Readily crosses both the blood-brain barrier and the placenta.
Minamata disease stands as the defining historical example of mercury mass poisoning. Between 1932 and 1968, the Chisso Corporation's chemical plant in Minamata, Japan, discharged an estimated 27–81 tonnes of methylmercury into Minamata Bay. Local fishing communities who consumed contaminated fish and shellfish developed a debilitating neurological syndrome characterized by sensory disturbances, ataxia, constriction of visual fields, hearing loss, and — in severe prenatal cases — profound cerebral palsy. More than 2,000 people were officially certified as having Minamata disease; thousands more were affected. A second outbreak occurred in the Agano River basin in Niigata prefecture in 1965. These tragedies directly shaped modern environmental mercury regulation and the 2013 Minamata Convention on Mercury.
In the global mercury cycle, natural sources (volcanic eruptions, weathering of mercury-bearing rocks, oceanic outgassing) release approximately 5,000–8,000 tonnes per year, while anthropogenic sources contribute a roughly equal or greater amount. Once emitted to the atmosphere as elemental mercury vapor, it can travel globally before being oxidized and deposited. Aquatic microorganisms then convert inorganic mercury to methylmercury, which enters food webs and undergoes biomagnification — concentrations increasing at each trophic level until reaching up to 10 million times ambient water concentrations in large predatory fish.
2. Sources of Exposure
Fish and Seafood
Dietary intake of fish and seafood is the dominant route of methylmercury exposure for the general population. Biomagnification means that large, long-lived, predatory species accumulate the highest concentrations. Shark, swordfish, king mackerel, tilefish, and bigeye tuna regularly exceed 1 part per million (ppm) mercury — the threshold at which the U.S. Food and Drug Administration (FDA) advises avoidance for vulnerable populations. Canned light tuna averages approximately 0.128 ppm, while salmon, sardines, and shrimp typically contain less than 0.05 ppm.
Biomagnification operates through several mechanisms: methylmercury binds tightly to muscle proteins, is not efficiently excreted, and accumulates over the lifetime of the organism. A swordfish that lives 9 years acquires mercury from the hundreds of smaller fish it consumes; a sperm whale feeding on those swordfish accumulates even higher burdens.
Dental Amalgam
Dental amalgam fillings are approximately 50% elemental mercury by weight, combined with silver, tin, copper, and zinc. Mercury vapor is continuously released from amalgam surfaces, particularly during chewing, tooth grinding, and consumption of hot liquids. Inhaled vapor is absorbed at roughly 80% efficiency through the lungs. Studies have found that individuals with multiple amalgam fillings have measurably higher blood and urine mercury levels than those without. The average amalgam filling releases an estimated 1–10 micrograms of mercury per day.
Coal-Burning Power Plants
Combustion of coal is the largest single anthropogenic source of mercury emissions globally, accounting for roughly 25–30% of total anthropogenic releases. Mercury present in coal is volatilized during combustion and emitted as gaseous elemental mercury, which then undergoes atmospheric oxidation, deposition, and methylation in aquatic systems. A single large coal-fired power plant can emit 50–200 pounds of mercury annually. Mercury emissions from power plants have been substantially reduced in countries with flue-gas desulfurization and activated carbon injection controls.
Artisanal and Small-Scale Gold Mining
Artisanal and small-scale gold mining (ASGM) is the world's largest source of anthropogenic mercury releases to the environment, estimated at 800–1,000 tonnes per year globally. Miners use liquid elemental mercury to form an amalgam with gold particles; the amalgam is then heated to evaporate the mercury, releasing vapor that is directly inhaled by workers and their families. ASGM affects an estimated 10–19 million miners in over 70 countries, predominantly in sub-Saharan Africa, South and Southeast Asia, and Latin America. Blood mercury levels in ASGM communities frequently exceed occupational exposure limits by orders of magnitude.
Broken Thermometers and Compact Fluorescent Lamps
Traditional mercury thermometers contain approximately 0.5–3 grams of elemental mercury. While a single broken thermometer poses limited risk in a well-ventilated space, improper cleanup — particularly vacuuming, which disperses mercury as fine droplets — can lead to significant vapor contamination. Compact fluorescent lamps (CFLs) contain 1–5 milligrams of mercury, sufficient to cause brief localized vapor elevation when broken indoors. Proper ventilation and damp-wipe cleanup followed by sealed disposal are recommended for both scenarios.
3. Metabolism and Toxicokinetics
Absorption
The route and efficiency of mercury absorption depend critically on its chemical form. Methylmercury is absorbed with approximately 95% efficiency from the gastrointestinal tract, making dietary fish consumption an extremely efficient exposure route. It enters intestinal epithelial cells via amino acid transport systems, particularly as a cysteine conjugate that mimics methionine. Inorganic mercury is absorbed at only 7–15% efficiency from the gut. Elemental mercury vapor, when inhaled, crosses alveolar membranes and enters the bloodstream with approximately 80% efficiency due to its high lipid solubility.
Distribution and Tissue Binding
In blood, methylmercury partitions predominantly into red blood cells (approximately 90%), where it binds to hemoglobin. The whole-blood to plasma ratio is approximately 20:1. Inorganic mercury distributes primarily to the kidneys, which contain the highest concentrations of inorganic mercury in chronically exposed individuals. Methylmercury distributes widely to all tissues, with brain, liver, and kidney as major repositories.
Blood-Brain Barrier and Placental Transfer
Methylmercury's ability to cross the blood-brain barrier (BBB) is central to its neurotoxicity. The cysteine-methylmercury complex is transported across the BBB via the L-type neutral amino acid transporter (LAT1), exploiting the transporter's affinity for large neutral amino acids. Once in the brain, methylmercury undergoes slow demethylation to inorganic mercury, which accumulates in lysosomes and may persist in neural tissue for years to decades. The half-life of inorganic mercury in brain tissue is estimated at several years, far exceeding the approximately 70-day blood half-life of methylmercury.
Methylmercury also crosses the placenta efficiently and accumulates in fetal tissues at concentrations 70–100% higher than maternal blood levels. The developing fetal brain is particularly vulnerable because of rapid cell division, incomplete BBB function, and limited antioxidant defenses. Breast milk transmits methylmercury to nursing infants, though at lower concentrations than cord blood.
Selenium Binding
Selenium has a remarkably high affinity for mercury, forming insoluble mercury selenide (HgSe) complexes in tissues. This interaction is bidirectional: selenium can sequester mercury and reduce its toxicity, but mercury can simultaneously deplete biologically active selenium, impairing selenoprotein function. The selenium:mercury molar ratio in tissues is a key determinant of toxicological outcome — when selenium exceeds mercury molarwise, protective sequestration predominates; when mercury exceeds selenium, toxicity is enhanced.
Elimination and Half-Life
Methylmercury is primarily eliminated via biliary excretion into the gut, where intestinal bacteria partially demethylate it and the inorganic residue is excreted in feces. A fraction undergoes enterohepatic recirculation. The blood half-life of methylmercury is approximately 70 days in adults. Hair mercury reflects integrated blood mercury levels over time (approximately 1 cm of hair growth per month), making hair analysis useful for retrospective exposure reconstruction. Inorganic mercury is eliminated primarily in urine. Mercury in brain tissue turns over far more slowly, with half-lives estimated at 1–22 years for different brain regions.
4. Mechanism of Toxicity
Sulfhydryl Group Binding
Mercury's primary molecular mechanism of toxicity is its extraordinarily high affinity for sulfhydryl (thiol, -SH) groups on proteins and small molecules. Mercury forms stable, covalent bonds with cysteine residues, glutathione, and other thiol-containing compounds with binding constants many orders of magnitude higher than those of calcium or zinc. This non-selective binding inactivates hundreds of enzymes and structural proteins, including those critical for antioxidant defense, energy metabolism, DNA repair, and neurotransmission.
Selenium Depletion and Selenoprotein Inhibition
Mercury has an even higher affinity for selenium than for sulfur, forming extremely insoluble mercury selenide. This sequesters selenium away from selenoproteins, a family of 25 human proteins that include glutathione peroxidases (antioxidant enzymes), thioredoxin reductases (redox regulation), and iodothyronine deiodinases (thyroid hormone activation). Inhibition of these selenoproteins amplifies oxidative stress and disrupts thyroid hormone metabolism.
Oxidative Stress
Mercury dramatically increases reactive oxygen species (ROS) production through multiple mechanisms: inhibition of glutathione peroxidase and thioredoxin reductase, depletion of glutathione itself (mercury binds glutathione directly), and impairment of mitochondrial electron transport. The resulting oxidative stress damages cellular lipids, proteins, and DNA. Neuronal cells, which have high metabolic rates and relatively low antioxidant capacity, are especially vulnerable to mercury-induced oxidative damage.
Mitochondrial Dysfunction
Mitochondria are critical targets of mercury toxicity. Mercury inhibits complexes I, II, III, and IV of the electron transport chain, uncouples oxidative phosphorylation, disrupts mitochondrial membrane potential, and triggers mitochondrial permeability transition. The result is ATP depletion, impaired calcium homeostasis, and initiation of intrinsic apoptotic pathways. Neurons are particularly dependent on mitochondrial ATP production and are thus highly susceptible.
Microtubule Disruption
Methylmercury disrupts neuronal cytoskeletal architecture by inhibiting tubulin polymerization. Tubulin contains multiple cysteine residues whose thiol groups are targeted by mercury. Disruption of microtubules impairs axonal transport (critical for delivering mitochondria and synaptic vesicles to nerve terminals), neuronal migration during development, and synaptic function. This mechanism is thought to contribute significantly to the characteristic cerebellar and sensory neuron damage of methylmercury poisoning.
Neuronal Apoptosis
The convergence of oxidative stress, mitochondrial dysfunction, microtubule disruption, and direct inhibition of antiapoptotic proteins triggers programmed cell death in neurons. Mercury activates both the intrinsic (mitochondrial) and extrinsic apoptotic pathways, as well as caspase-independent cell death mechanisms. In the developing brain, mercury-induced apoptosis can eliminate irreplaceable neurons during critical periods of synaptogenesis and circuit formation, producing permanent functional deficits.
5. Health Effects
Neurotoxicity
The nervous system is the primary target of both methylmercury and elemental mercury vapor. The classic triad of chronic elemental mercury vapor poisoning includes tremor (initially fine intention tremor of the hands, progressing to whole-body involvement), erethism (an unusual neuropsychiatric complex of excessive shyness, emotional lability, insomnia, and memory disturbances), and gingivitis. Methylmercury poisoning, as seen in Minamata disease, produces a distinct syndrome dominated by:
- Sensory disturbances: Bilateral paresthesias (numbness and tingling) of the hands, feet, and perioral region, reflecting preferential damage to sensory neurons.
- Cerebellar ataxia: Unsteady gait, dysarthria (slurred speech), and intentional tremor resulting from Purkinje cell damage in the cerebellum.
- Constriction of visual fields: Bilateral concentric narrowing of the visual field ("tunnel vision") due to damage to the calcarine cortex; a hallmark of Minamata disease.
- Sensorineural hearing loss: Damage to the auditory cortex and cochlear nerve.
Cognitive and Psychiatric Effects
Chronic low-level mercury exposure, even below the threshold for classical neurotoxic symptoms, has been associated with measurable deficits in memory, attention, processing speed, and fine motor coordination. Occupational studies of mercury-exposed workers document dose-dependent reductions in neuropsychological test performance. Several population studies have found associations between blood or hair mercury levels and subtle cognitive deficits in adults.
Prenatal and Developmental Effects
The fetal brain is far more sensitive to methylmercury than the adult brain. The Faroe Islands prospective cohort study, led by Philippe Grandjean, followed children born to women who consumed pilot whale meat (which contains high methylmercury concentrations) during pregnancy. Children with higher cord blood mercury levels demonstrated dose-dependent deficits in attention, language, verbal memory, and visuospatial function at ages 7 and 14, with effects detectable at cord blood levels below 20 µg/L. A parallel cohort in the Seychelles Islands, where fish consumption is very high but the species consumed are lower in mercury, showed less consistent findings, underscoring the importance of species-specific mercury levels and the potential confounding benefit of omega-3 fatty acids in fish.
Cardiovascular Effects
Epidemiological studies have examined associations between mercury exposure and cardiovascular disease, particularly among fish-consuming populations. The Finnish prospective study by Virtanen and colleagues found that high hair mercury levels (reflecting high intake of freshwater fish from mercury-contaminated lakes) were independently associated with increased risk of acute myocardial infarction. Proposed mechanisms include mercury-induced oxidative stress in vascular endothelium, impairment of antioxidant selenoproteins, and direct cardiotoxic effects. However, the cardiovascular benefit of omega-3 fatty acids in marine fish may partially counteract mercury's adverse effects, complicating epidemiological interpretation.
Renal Effects
The kidney is the primary target organ for inorganic mercury. Proximal tubular cells, which are responsible for reabsorbing filtered mercury, accumulate high concentrations of inorganic mercury and are selectively damaged. Manifestations range from subclinical proteinuria and tubular enzymuria to acute tubular necrosis in severe poisoning cases. Nephrotic syndrome via immune complex glomerulonephritis has also been described. Urine mercury and N-acetyl-beta-D-glucosaminidase (NAG) are sensitive biomarkers of mercury-induced renal tubular damage.
Immunological Effects
Mercury can trigger autoimmune reactions through multiple mechanisms, including modification of self-proteins (creating novel antigenic epitopes), activation of autoreactive T-lymphocytes, and polyclonal B-cell activation. Animal studies consistently demonstrate that mercury exposure induces autoimmune glomerulonephritis and thyroiditis. Human epidemiological data are less conclusive but suggest possible associations with autoimmune thyroid disease, lupus-like syndromes, and multiple sclerosis in genetically susceptible individuals.
Endocrine Disruption
Mercury disrupts the endocrine system through several pathways. Inhibition of selenoprotein iodothyronine deiodinases impairs conversion of T4 to the active thyroid hormone T3, potentially contributing to hypothyroidism-like symptoms. Mercury binds to thyroid hormone receptor binding sites and can interfere with glucocorticoid signaling. Studies of occupationally and environmentally exposed populations have found associations between mercury exposure and altered thyroid hormone levels, menstrual irregularities, and reduced testosterone levels in men.
6. Mercury in Seafood
FDA/EPA Fish Advisory Categories
The U.S. FDA and EPA jointly issue fish consumption advisories, with particular guidance for pregnant women, women who may become pregnant, breastfeeding mothers, and young children — groups most vulnerable to methylmercury's neurodevelopmental effects. The current advisory (updated 2024) categorizes commercial fish into three groups based on average methylmercury concentrations:
- Best choices (eat 2–3 servings per week): Salmon, sardines, herring, anchovies, canned light tuna, tilapia, catfish, cod, shrimp, oysters, scallops, clams, crab, trout. These typically contain less than 0.1 ppm mercury.
- Good choices (eat 1 serving per week): Albacore (white) tuna, halibut, mahi-mahi, grouper, carp, yellowfin tuna, monkfish, bluefish, Chilean sea bass. These typically contain 0.1–0.3 ppm mercury.
- Choices to avoid (highest mercury levels): Shark, swordfish, king mackerel, orange roughy, bigeye tuna, marlin, tilefish from the Gulf of Mexico. These typically contain more than 0.3–1.0 ppm mercury.
Omega-3 Benefit vs. Mercury Risk Calculus
The public health calculus of fish consumption is complicated by the well-established cardiovascular and neurodevelopmental benefits of omega-3 fatty acids (EPA and DHA) present in the same fatty fish that may contain mercury. For the general adult population, the cardiovascular benefits of regular fish consumption (2+ servings per week) are considered to outweigh mercury risks when lower-mercury species are chosen. For pregnant women, the developmental benefits of DHA for fetal brain and eye development argue for continued fish consumption — but restricted to low-mercury species and quantities within advisory limits. Avoiding fish entirely during pregnancy to minimize mercury exposure may paradoxically harm fetal neurodevelopment by eliminating an important DHA source.
Selenium:Mercury Molar Ratio Hypothesis
Researchers including Nicholas Ralston have proposed that the molar ratio of selenium to mercury in fish flesh is a better predictor of net health effects than mercury concentration alone. Fish with selenium:mercury molar ratios greater than 1 (meaning selenium exceeds mercury on a mole-for-mole basis) may provide net neuroprotection because the excess selenium is available to support selenoprotein function after mercury is sequestered as mercury selenide. Oceanic fish like tuna and salmon tend to have high selenium:mercury ratios (often 10:1 or greater), while freshwater fish from mercury-contaminated lakes may have ratios below 1. Pilot whale meat, consumed in the Faroe Islands, has an unusually low selenium:mercury ratio, which may explain the potent neurotoxic effects observed in Faroese children despite mercury levels not dramatically higher than those in some ocean fish.
Safest Fish Choices
For individuals wishing to maximize omega-3 intake while minimizing mercury exposure, the following species consistently offer the most favorable profiles:
- Wild Alaskan salmon: High EPA/DHA, very low mercury (<0.01 ppm average), high selenium.
- Sardines: Exceptionally high EPA/DHA per calorie, very low mercury (short-lived, low trophic level), high selenium and calcium.
- Atlantic mackerel (not king mackerel): High omega-3, low mercury.
- Anchovies: Very low mercury, high omega-3.
- Arctic char: Similar profile to salmon with low mercury levels.
7. Dental Amalgam Controversy
Composition and Mercury Vapor Release
Dental amalgam ("silver fillings") is composed of approximately 50% elemental mercury, 25–35% silver, and smaller proportions of tin, copper, and zinc. The alloy is stable under normal conditions, but mercury vapor is continuously released, particularly during chewing, tooth brushing, clenching, and consumption of hot beverages. Estimates of daily mercury release range from 1 to 10 micrograms per filling per day, with higher estimates for those with many fillings who chew vigorously. Absorbed amalgam-derived mercury contributes to detectable blood and urine mercury levels — studies consistently find that amalgam carriers have roughly twice the urine mercury levels of non-carriers.
FDA 2020 Advisory for Sensitive Populations
In 2020, the U.S. FDA updated its guidance to recommend avoiding dental amalgam, when possible, in specific high-risk populations: pregnant women and women planning pregnancy, nursing mothers, children under 6 years, people with kidney impairment, people with neurological conditions, and people with known hypersensitivity to mercury or other amalgam components. The FDA stopped short of recommending removal of existing amalgam in asymptomatic patients, noting that drilling out intact fillings releases a brief pulse of mercury vapor and that the net health benefit of removal is uncertain.
European Restrictions
The European Union has pursued more aggressive amalgam restrictions. Since 2018, dental amalgam has been prohibited for use in children under 15 and in pregnant or breastfeeding women across EU member states. Sweden and Norway banned amalgam entirely as early as 2008–2009. The EU's broader goal is a complete phase-down of dental amalgam use, aligned with the Minamata Convention's objective of reducing mercury in products.
Alternatives
Composite resin (tooth-colored plastic) fillings are the primary alternative to amalgam for most posterior restorations and do not contain mercury. Glass ionomer cements, ceramic inlays, and gold alloy restorations are additional options. Modern composite resins have improved durability, with longevity approaching that of amalgam in well-placed restorations. The higher cost of composite versus amalgam remains a barrier to access in some healthcare systems.
8. Diagnosis
Blood Mercury
Whole-blood mercury reflects recent methylmercury exposure (primarily from fish consumption) and is the preferred biomarker for acute or ongoing dietary exposure assessment. Blood mercury predominantly represents methylmercury (as organic mercury in red blood cells). The CDC reference range for blood mercury in U.S. adults is <5.8 µg/L (95th percentile). The EPA's reference dose for methylmercury corresponds to a blood level of approximately 5.8 µg/L. Values above 20–50 µg/L are associated with early subclinical neurotoxic effects; levels above 200 µg/L may produce overt neurological signs.
Urine Mercury
Urine mercury primarily reflects inorganic mercury burden — from dental amalgam vapor or past exposure to inorganic mercury compounds. It is the preferred biomarker for occupational exposure monitoring and for tracking the efficacy of amalgam removal. Urine mercury is typically reported as micrograms per gram creatinine (µg/g Cr) to correct for urine concentration. Values above 35 µg/g Cr are considered elevated in occupational settings; values above 100 µg/g Cr warrant clinical evaluation.
Hair Mercury
Hair mercury provides a retrospective record of methylmercury exposure, since mercury is incorporated into the hair shaft in proportion to blood mercury levels at the time of growth. Approximately 1 cm of hair represents one month of exposure history. Hair mercury is the preferred biomarker for epidemiological studies of prenatal methylmercury exposure (using maternal hair collected during or after pregnancy) and for evaluating chronic dietary patterns. Normal reference values are generally <1 µg/g; values above 2 µg/g suggest elevated dietary methylmercury intake.
Provocation Testing Controversy
Provocation (challenge) testing involves administering a chelating agent (typically DMSA or DMPS) and measuring urinary mercury excretion over the subsequent 6–24 hours. Proponents argue this reveals a "body burden" of mercury not detected by baseline urine testing. However, major toxicology organizations — including the American College of Medical Toxicology and the CDC — do not recommend provocation testing for mercury, citing several concerns: it lacks validated reference ranges, mobilized mercury may redistribute to sensitive tissues (including the brain), it has not been shown to correlate with clinical toxicity, and it can produce abnormal-appearing results in unexposed individuals. Provocation testing is widely used in integrative and alternative medicine settings but is not supported by mainstream toxicological evidence.
9. Treatment and Detoxification
Medical Chelation Therapy
Chelation therapy is the cornerstone of treatment for clinically significant mercury poisoning, using agents that form stable, water-soluble complexes with mercury that are then excreted in urine or bile:
- DMSA (succimer, 2,3-dimercaptosuccinic acid): An oral dithiol chelator approved by the FDA for pediatric lead poisoning and widely used for mercury. Effective for both inorganic and methylmercury, with relatively favorable safety profile. Standard dosing is 10 mg/kg orally three times daily for 5 days, repeated in cycles. Side effects include gastrointestinal upset, transient liver enzyme elevation, and skin rash.
- DMPS (2,3-dimercaptopropane-1-sulfonate): Available by prescription in Germany and other European countries; used off-label in the United States. Administered orally or intravenously. Generally considered more potent than DMSA for inorganic mercury and particularly effective for elemental mercury vapor poisoning. Also used in provocation testing, though this application is controversial.
- BAL (British Anti-Lewisite, dimercaprol): The oldest mercurial chelator, developed during World War II. Administered by intramuscular injection. Now largely superseded by DMSA and DMPS due to more adverse effects, including hypertension and nephrotoxicity. Still used for severe acute poisoning when IV therapy is required and DMPS is unavailable.
Chelation should be guided by clinical assessment and confirmed elevated mercury levels. It is not recommended for asymptomatic individuals with borderline exposures or as a preventive measure.
Selenium Co-supplementation
Given mercury's depletion of bioactive selenium, co-supplementation with selenium is a rational adjunct to chelation therapy in mercury-poisoned individuals. Selenium as selenomethionine or sodium selenite may help restore selenoprotein activity and augment endogenous mercury sequestration. However, the optimal dose and form of selenium supplementation in mercury poisoning have not been established in controlled clinical trials, and excessive selenium supplementation carries its own toxicity risk (selenosis). Doses in the range of 100–200 micrograms per day as selenomethionine are commonly used clinically.
N-Acetyl Cysteine (NAC)
N-acetyl cysteine is a precursor to glutathione, the primary intracellular antioxidant and a key mercury-binding molecule. NAC supplementation increases cellular glutathione levels, potentially improving mercury sequestration and reducing oxidative stress. Some animal studies demonstrate that NAC augments mercury excretion. It is commonly used as an adjunct in functional medicine protocols for mercury detoxification, though robust human clinical trial data are lacking. Doses range from 600 to 1,800 mg per day in divided doses.
Alpha-Lipoic Acid
Alpha-lipoic acid (ALA) is a dithiol antioxidant that can chelate mercury in vitro and has been shown to mobilize mercury in animal models. ALA is both water- and fat-soluble, giving it access to compartments unavailable to purely water-soluble chelators. However, controversy exists about ALA's safety in mercury-burdened individuals: the Cutler protocol cautions that ALA's ability to cross the blood-brain barrier may redistribute mercury into the brain if used incorrectly. ALA is widely used in integrative medicine settings for mercury and other heavy metal detoxification, but evidence from controlled human trials is sparse.
Chlorella and Cilantro Protocol
The combination of Chlorella pyrenoidosa (a green microalgae) and cilantro (Coriandrum sativum) leaf extract is promoted in naturopathic medicine as a natural mercury detoxification protocol. Chlorella is theorized to bind mercury in the gut (reducing enterohepatic recirculation) and to mobilize heavy metals from tissues. Cilantro is claimed to mobilize mercury from the central nervous system. Animal studies show some mercury-binding activity by chlorella, and small human studies report modest reductions in fecal and urine mercury. However, the clinical evidence base is limited, and this protocol should not be used as a substitute for medical chelation in individuals with significant mercury toxicity.
Dietary Fiber
Dietary fiber can reduce methylmercury absorption by binding mercury in the gut and decreasing enterohepatic recirculation. Certain types of fiber — particularly pectin (from fruits and vegetables) and fiber from whole grains — have demonstrated mercury-binding capacity in vitro and animal studies. High-fiber diets are a reasonable general health measure that may provide modest ancillary benefit in reducing mercury absorption from dietary sources.
10. Prevention
The Minamata Convention on Mercury
The Minamata Convention on Mercury, adopted in 2013 and entering into force in 2017, is a global treaty under the United Nations Environment Programme (UNEP) that aims to protect human health and the environment from anthropogenic mercury emissions and releases. Key provisions include: phasing out or banning mercury in products (including thermometers, batteries, fluorescent lamps, and dental amalgam); controlling emissions from coal-fired power plants, cement production, and non-ferrous metal smelting; restricting mercury use in artisanal gold mining; and eliminating mercury from specific manufacturing processes. As of 2024, over 140 countries have ratified the convention. The treaty is named for Minamata, Japan, the site of the world's most infamous mercury mass poisoning.
Fish Selection Strategies
Practical strategies for minimizing methylmercury exposure through dietary choices include:
- Prioritizing short-lived, low-trophic-level species (sardines, anchovies, herrings, farmed oysters, clams) that accumulate far less mercury than large predatory fish.
- Limiting consumption of high-mercury species (shark, swordfish, tilefish, king mackerel, bigeye tuna) to no more than once per month, or avoiding them entirely during pregnancy.
- Using resources such as the FDA/EPA "Advice about Eating Fish" or the Monterey Bay Aquarium Seafood Watch guide, which integrate mercury data with sustainability considerations.
- For recreational anglers, following state-issued fish consumption advisories for local water bodies, which often reflect locally elevated mercury levels in freshwater species.
Amalgam Avoidance
For individuals with ongoing concerns about mercury from dental sources, practical prevention measures include:
- Requesting composite resin or ceramic restorations when new fillings are needed.
- For existing amalgam fillings, avoiding replacement in the absence of dental necessity, as drilling produces a pulse of mercury vapor and particulate.
- If amalgam removal is elected (e.g., for individuals with documented sensitivity), seeking a dentist who uses rubber dam isolation, high-volume evacuation, and sectioning techniques to minimize exposure during removal.
- Ensuring adequate selenium status through diet (Brazil nuts, seafood, organ meats) to support selenoprotein-mediated mercury detoxification.
11. Related Topics
12. References
- Grandjean P, Landrigan PJ. Neurobehavioural effects of developmental toxicity. Lancet Neurol. 2014;13(3):330–338.
- Clarkson TW, Magos L. The toxicology of mercury and its chemical compounds. Crit Rev Toxicol. 2006;36(8):609–662.
- Mergler D, Anderson HA, Chan LHM, et al. Methylmercury exposure and health effects in humans: a worldwide concern. Ambio. 2007;36(1):3–11.
- Grandjean P, Weihe P, White RF, et al. Cognitive deficit in 7-year-old children with prenatal exposure to methylmercury. Neurotoxicol Teratol. 1997;19(6):417–428.
- Myers GJ, Davidson PW, Cox C, et al. Prenatal methylmercury exposure from ocean fish consumption in the Seychelles child development study. Lancet. 2003;361(9370):1686–1692.
- Salonen JT, Seppänen K, Nyyssönen K, et al. Intake of mercury from fish, lipid peroxidation, and the risk of myocardial infarction and coronary, cardiovascular, and any death in eastern Finnish men. Circulation. 1995;91(3):645–655.
- Ralston NVC, Ralston CR, Blackwell JL 3rd, Raymond LJ. Dietary and tissue selenium in relation to methylmercury toxicity. Neurotoxicology. 2008;29(5):802–811.
- Virtanen JK, Voutilainen S, Rissanen TH, et al. Mercury, fish oils, and risk of acute coronary events and cardiovascular disease, coronary heart disease, and all-cause mortality in men in eastern Finland. Arterioscler Thromb Vasc Biol. 2005;25(1):228–233.
- Zahir F, Rizwi SJ, Haq SK, Khan RH. Low dose mercury toxicity and human health. Environ Toxicol Pharmacol. 2005;20(2):351–360.
- Bernhoft RA. Mercury toxicity and treatment: a review of the literature. J Environ Public Health. 2012;2012:460508.
- U.S. Food and Drug Administration. Advice About Eating Fish: For Women Who Are or Might Become Pregnant, Breastfeeding Mothers, and Young Children. FDA; 2024. fda.gov
- U.S. FDA. Dental Amalgam Fillings: Information for Specific Groups. FDA; 2020. fda.gov
- Harada M. Minamata disease: methylmercury poisoning in Japan caused by environmental pollution. Crit Rev Toxicol. 1995;25(1):1–24.
- Bhattacharya S. The facts about penicillamine in treating Wilson's disease. J Clin Diagn Res. 2016;10(4):OE01–OE03.
- Crinnion WJ. Environmental medicine, part 4: Pesticides — biologically persistent and ubiquitous toxins. Altern Med Rev. 2000;5(5):432–447. PMID: 11056413
- Rooney JPK. The retention time of inorganic mercury in the brain — a systematic review of the evidence. Toxicol Appl Pharmacol. 2014;274(3):425–435.
- United Nations Environment Programme. Global Mercury Assessment 2018. UNEP; 2018. unep.org
- Selin NE. Global biogeochemical cycling of mercury: a review. Annu Rev Environ Resour. 2009;34:43–63.
- Bjørklund G, Aaseth J, Ajsuvakova OP, et al. Molecular interaction between mercury and selenium in neurotoxicity. Coord Chem Rev. 2017;332:30–37.
- Genchi G, Sinicropi MS, Carocci A, Lauria G, Catalano A. Mercury exposure and heart diseases. Int J Environ Res Public Health. 2017;14(1):74.
Connections
- Lead
- Selenium
- Chlorella
- Heavy Metals
- Arsenic
- Cadmium
- Tuna
- Salmon
- Sardines
- Cysteine
- Glutathione
- NAC
- Detoxification
- Omega-3 Fatty Acids
- Zinc
- Hypertension
- Oxidative Stress
- Detox Protocols