Arsenic Exposure: Sources, Health Effects, and Protection
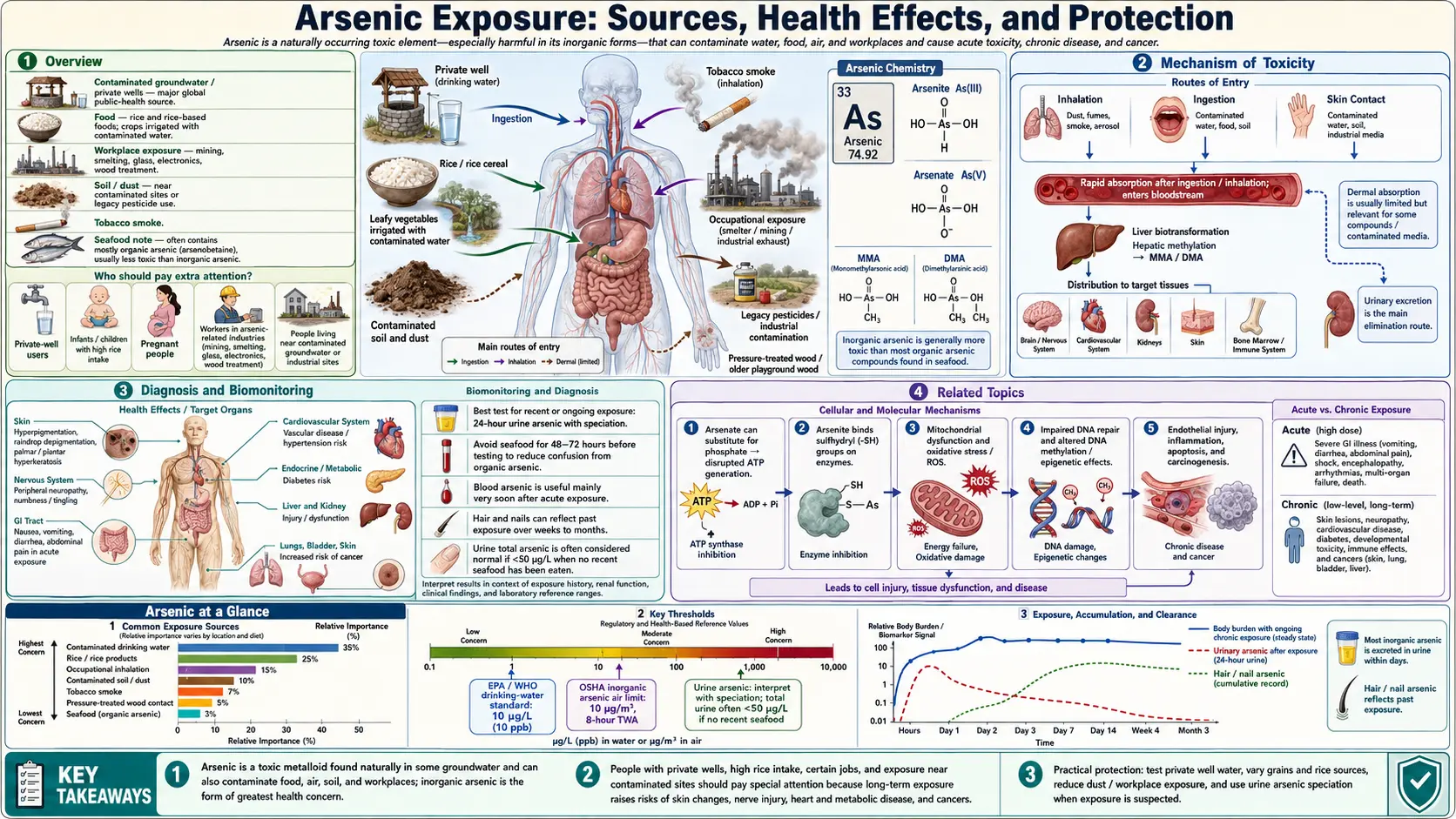



Arsenic is a naturally occurring metalloid with a long history of both medicinal use and toxic exposure. Today it ranks among the most pervasive environmental contaminants worldwide, affecting an estimated 200 million people through contaminated groundwater alone. Understanding its sources, how the body processes it, and how to reduce exposure is essential for informed health decisions.
Table of Contents
- Overview
- Sources of Exposure
- Metabolism and Toxicokinetics
- Mechanism of Toxicity
- Health Effects
- Arsenic in Drinking Water
- Arsenic in Food
- Diagnosis and Biomonitoring
- Treatment and Detoxification
- Prevention and Reduction Strategies
- Related Topics
- References
- Connections
- Featured Videos
1. Overview
Arsenic (chemical symbol As, atomic number 33) exists in multiple chemical forms that differ dramatically in toxicity. Organic arsenic compounds — such as arsenobetaine found in seafood — are largely nontoxic and are rapidly excreted. Inorganic arsenic, by contrast, is highly toxic and is the form responsible for most human health concerns. Inorganic arsenic occurs in two primary oxidation states: arsenite (As3+, or As(III)) and arsenate (As5+, or As(V)).
The International Agency for Research on Cancer (IARC) classifies inorganic arsenic as a Group 1 carcinogen — definitively carcinogenic to humans — with sufficient evidence for cancers of the skin, lung, and urinary bladder, and limited evidence for kidney and liver cancers.
Arsenic contamination represents a global public health crisis. Elevated concentrations occur naturally in groundwater across South and Southeast Asia, Latin America, and parts of the United States due to geological dissolution of arsenic-containing minerals. The WHO estimates that more than 200 million people are chronically exposed to arsenic above the 10 micrograms-per-liter (µg/L) guideline value. In countries such as Bangladesh and India, tens of millions rely on wells with arsenic levels exceeding safe thresholds by factors of ten or more, producing what has been called the largest mass poisoning in human history.
2. Sources of Exposure
Groundwater Contamination
Geological dissolution of arsenic-bearing minerals — particularly iron oxyhydroxides and sulfide minerals such as arsenopyrite — is the dominant natural source of groundwater arsenic. The Bangladesh and West Bengal crisis is the most severe documented case: surveys in the 1990s revealed that approximately 35–77 million people in Bangladesh alone were drinking water with arsenic above 50 µg/L (the then-national standard), with millions exposed to levels exceeding 200 µg/L. The crisis is attributed to the anaerobic dissolution of arsenic from aquifer sediments, accelerated by the large-scale installation of tube wells intended to provide microbiologically safe drinking water.
In the United States, arsenic hotspots include the western states (Nevada, Arizona, Utah, Montana, Idaho), New England (Maine, New Hampshire), and parts of the Midwest. An estimated 13 million Americans rely on private wells, which are unregulated at the federal level and may contain arsenic concentrations well above the EPA maximum contaminant level (MCL) of 10 µg/L.
Rice and Rice Products
Rice is the single most important dietary source of inorganic arsenic in populations that consume it as a staple. Rice is a semi-aquatic crop grown in flooded paddies, and it uniquely hyperaccumulates inorganic arsenic from soil and irrigation water via silicon transport channels. Rice bran and brown rice contain higher arsenic concentrations than white rice because arsenic concentrates in the outer bran layer. Rice cereals, rice cakes, rice crackers, and rice-based infant formula are significant exposure sources, particularly for infants and young children.
Apple Juice and Fruit Juices
Apple juice has been identified as a source of inorganic arsenic exposure in children. Concerns arose following FDA and Consumer Reports analyses showing that a significant proportion of apple juice samples exceeded 10 µg/L. In 2013 the FDA set an action level of 10 µg/L for inorganic arsenic in apple juice. The arsenic derives partly from historical use of lead arsenate pesticides on orchards and from arsenic in irrigation water.
Chicken (Historical Roxarsone Use)
For decades, the organoarsenic compound roxarsone (3-nitro-4-hydroxyphenylarsonic acid) was added to poultry feed in the United States to promote growth and control intestinal parasites. Studies found that roxarsone metabolized to inorganic arsenic in chicken tissue and was excreted in manure, contaminating soil and water. The FDA withdrew approval for roxarsone and related compounds between 2011 and 2015. Chicken sold after this period carries substantially lower arsenic levels, though historical litter application to agricultural land may persist as a legacy source.
CCA-Treated Wood
Chromated copper arsenate (CCA) was widely used to pressure-treat wood for outdoor applications — playground equipment, decks, picnic tables, and residential fencing — until the EPA phased it out for residential use in 2004. CCA-treated lumber is identifiable by its greenish tint. Children playing on CCA-treated structures can ingest arsenic through hand-to-mouth behavior after touching treated surfaces. Sanding, cutting, or burning CCA wood releases toxic arsenic-containing dust and fumes. Wood treated before the 2004 phase-out remains in service and continues to leach arsenic.
Occupational Exposure
Workers in several industries face elevated arsenic exposure. Smelting and refining of non-ferrous metals (copper, lead, zinc, gold) releases arsenic-containing dust and fumes. Semiconductor manufacturing uses arsine gas (AsH3) and gallium arsenide. Glass manufacturing historically used arsenic trioxide as a fining agent. Pesticide production and application (especially older arsenical herbicides such as MSMA) and wood preservative application are additional occupational routes. Coal combustion releases arsenic, creating exposure risk for power plant workers and nearby communities.
3. Metabolism and Toxicokinetics
Arsenite versus Arsenate
The two primary inorganic forms differ in cellular uptake mechanisms. Arsenate (As(V)) enters cells via phosphate transporters because it mimics phosphate structurally. Inside the cell it is rapidly reduced to arsenite. Arsenite (As(III)) enters via aquaporin channels (particularly AQP7 and AQP9) and through glucose transporters. Arsenite is generally considered more acutely toxic than arsenate because it binds more avidly to thiol (sulfhydryl) groups in proteins.
Methylation Pathway
After absorption, inorganic arsenic undergoes biomethylation in the liver — the primary detoxification pathway in humans. The stepwise process, using S-adenosylmethionine (SAM) as the methyl donor, proceeds as follows:
- Inorganic arsenite (iAs3+) is oxidized to arsenate (iAs5+), then reduced back to arsenite prior to methylation.
- First methylation yields monomethylarsonic acid (MMA5+), which is reduced to monomethylarsonous acid (MMA3+).
- Second methylation yields dimethylarsinic acid (DMA5+), which may be further reduced to dimethylarsinous acid (DMA3+).
The trivalent methylated intermediates — particularly MMA3+ and DMA3+ — are more reactive and more cytotoxic than the parent inorganic arsenic, complicating the simple view that methylation is purely detoxifying. DMA5+ is the major urinary excretion product in humans.
AS3MT Enzyme and Genetic Polymorphisms
The enzyme arsenic (+3 oxidation state) methyltransferase (AS3MT) catalyzes both methylation steps. Single nucleotide polymorphisms (SNPs) in the AS3MT gene significantly influence methylation efficiency. Individuals who are poor methylators retain higher proportions of inorganic arsenic and MMA in their tissues and urine, correlating with greater cancer risk in epidemiological studies. Folate status is critical because methylation requires adequate SAM; folate deficiency impairs arsenic methylation and increases toxicity.
Organ Distribution and Urinary Excretion
After gastrointestinal absorption (typically 70–90% for inorganic arsenic in solution), arsenic distributes to the liver, kidneys, lungs, and spleen. It binds to keratin-rich tissues — skin, hair, and nails — where it accumulates over weeks to months, making these tissues useful for retrospective biomonitoring. The primary excretion route is urine, where total arsenic is excreted as a mixture of inorganic arsenic (typically 10–30%), MMA (10–20%), and DMA (60–70%). Biological half-life of inorganic arsenic in blood is approximately 3–5 days; in hair and nails, months.
4. Mechanism of Toxicity
Oxidative Stress
Arsenic generates reactive oxygen species (ROS) including superoxide, hydrogen peroxide, and hydroxyl radicals, both directly and through inhibition of antioxidant enzymes such as glutathione peroxidase and superoxide dismutase. Depletion of reduced glutathione (GSH) — the cell's primary thiol-based antioxidant — amplifies oxidative damage to lipids, proteins, and DNA. Oxidative DNA damage, particularly 8-hydroxydeoxyguanosine (8-OHdG) formation, is a key mechanism underlying arsenic carcinogenesis.
Enzyme Inhibition
Arsenite's high affinity for thiol groups allows it to inhibit numerous enzymes whose activity depends on reduced cysteine residues. The most toxicologically significant target is the pyruvate dehydrogenase complex: arsenite binds to the lipoic acid cofactor of the E2 subunit (dihydrolipoamide acetyltransferase), blocking oxidative decarboxylation of pyruvate and halting entry of acetyl-CoA into the TCA cycle. This disrupts cellular energy production and explains the profound metabolic toxicity seen in acute arsenic poisoning. Alpha-ketoglutarate dehydrogenase is similarly inhibited, further impairing the TCA cycle.
DNA Repair Interference
Arsenic does not directly damage DNA as a classical mutagen but acts as a co-mutagen and DNA repair inhibitor. It inhibits nucleotide excision repair (NER), base excision repair (BER), and double-strand break repair, allowing DNA damage from other agents (including arsenic-generated ROS) to accumulate. Arsenic also inhibits poly(ADP-ribose) polymerase (PARP), a key DNA damage sensor and repair coordinator.
Epigenetic Changes
Chronic arsenic exposure induces widespread epigenetic alterations. These include global DNA hypomethylation (due to SAM depletion by arsenic methylation), gene-specific promoter hypermethylation silencing tumor suppressor genes, histone modification changes, and altered microRNA expression profiles. Epigenetic changes may be heritable across cell divisions and potentially across generations, explaining long-latency cancer development and transgenerational effects observed in animal models.
Angiogenesis Promotion
Paradoxically, the same arsenic trioxide used as a chemotherapeutic agent (in acute promyelocytic leukemia) promotes angiogenesis at lower, chronic exposure doses. Arsenic activates hypoxia-inducible factor 1-alpha (HIF-1α) and upregulates vascular endothelial growth factor (VEGF) expression, facilitating tumor vascularization and potentially contributing to the promotion phase of arsenic carcinogenesis in the lung and bladder.
5. Health Effects
Cancer
The evidence for arsenic-induced cancer is strongest for four sites, all classified as IARC Group 1:
- Skin cancer: The earliest and most visible marker of chronic arsenicosis. Both basal cell carcinoma and squamous cell carcinoma (Bowen's disease in situ progressing to invasive SCC) are dose-dependently increased. A landmark Taiwanese study found that populations drinking water with arsenic above 600 µg/L had bladder and kidney cancer rates 10–15 times higher than unexposed populations.
- Lung cancer: Strongly associated with both inhalation (occupational smelter workers) and ingestion (drinking water). A Chilean cohort experienced a 7-fold increase in lung cancer mortality in people exposed to high-arsenic drinking water during childhood, with effects persisting decades after exposure ceased.
- Bladder cancer: The most consistently observed cancer in drinking water studies globally. A U.S. ecological study estimated that reducing arsenic from 50 to 10 µg/L would prevent approximately 57 additional bladder cancer cases per million people per year.
- Kidney cancer: Elevated in multiple high-exposure populations, though with more variability than the above three sites.
Cardiovascular Disease — Blackfoot Disease
Taiwan's Blackfoot disease — a severe peripheral vascular disease causing gangrene of the extremities — was first described in southwestern Taiwan in the 1950s and is the most striking arsenic-related cardiovascular condition. Affecting populations drinking artesian well water with arsenic concentrations of 100–1,800 µg/L, it results from progressive occlusion of peripheral arteries. Beyond this extreme case, epidemiological evidence links arsenic exposure to increased risk of ischemic heart disease, carotid atherosclerosis, and QT prolongation at lower exposure levels common in the United States and Europe.
Diabetes Mellitus
Multiple epidemiological studies across different continents report associations between arsenic exposure and type 2 diabetes. Proposed mechanisms include arsenic's inhibition of glucose-stimulated insulin secretion in pancreatic beta cells, induction of insulin resistance in adipocytes and muscle cells via oxidative stress, and disruption of transcription factors (PPAR-gamma) involved in glucose metabolism. A prospective U.S. study (Strong Heart Study in American Indian populations) found significant associations between urinary arsenic and incident diabetes.
Neurological Effects
Acute high-dose arsenic causes a classic peripheral neuropathy — a painful, predominantly sensory, stocking-glove pattern neuropathy that may progress to motor involvement. Chronic lower-level exposure is associated with cognitive impairment, particularly in children. Studies in Bangladesh, India, and the United States show that children with elevated urinary arsenic perform worse on tests of intellectual function, verbal learning, and executive function. Arsenic crosses the blood-brain barrier and accumulates in several brain regions.
Skin Manifestations
Characteristic dermatological signs of chronic arsenicosis include:
- Melanosis: Hyperpigmentation (raindrop pigmentation) and hypopigmentation appearing in a mottled pattern on the trunk.
- Keratoses: Bilateral palmar and plantar hyperkeratosis, a hallmark of chronic arsenic exposure. Lesions may be punctate (hard, corn-like) or diffuse.
- Bowen's disease: Arsenic-induced intraepidermal squamous cell carcinoma in situ, presenting as slow-growing, scaly, erythematous plaques.
Dermatological signs typically appear after 5–10 years of exposure to arsenic concentrations above 100 µg/L and are used clinically as markers of chronic arsenicosis in endemic regions.
Reproductive Effects
Animal and epidemiological studies link arsenic to adverse reproductive outcomes. Associations include increased risk of spontaneous abortion, stillbirth, low birth weight, and preterm delivery in women with elevated arsenic exposure. Placental transfer of arsenic exposes the developing fetus, and cord blood arsenic correlates with maternal urinary arsenic. Early-life arsenic exposure has been associated with altered immune development and increased respiratory infections in infants.
6. Arsenic in Drinking Water
The World Health Organization (WHO) established a guideline value of 10 µg/L (10 parts per billion, ppb) for arsenic in drinking water in 1993, replacing the previous 50 µg/L standard. The 10 µg/L value was set as a practical limit reflecting analytical and treatment capabilities rather than a threshold below which no risk exists; the WHO acknowledges residual cancer risk at this level.
The U.S. Environmental Protection Agency (EPA) lowered the maximum contaminant level (MCL) for arsenic in public water systems from 50 µg/L to 10 µg/L, effective January 2006. The EPA estimated this rule would prevent 19–31 additional bladder and lung cancer cases per million people served by high-arsenic water systems, while acknowledging a residual lifetime cancer risk of approximately 1 in 300 at exactly 10 µg/L — far exceeding the customary 1 in 1,000,000 risk benchmark for environmental contaminants.
Countries Most Affected
Nations with documented large-scale groundwater arsenic contamination above WHO guidelines include Bangladesh, India (West Bengal, Jharkhand, Bihar, Uttar Pradesh, Assam), Pakistan, Nepal, Myanmar, Vietnam, Cambodia, China, Mongolia, Argentina, Chile, Mexico, and Hungary. The global scale of natural arsenic contamination in sedimentary aquifers is still being mapped; millions of people remain unaware they are drinking unsafe water.
Private Well Testing
In the United States, private wells are not regulated under the Safe Drinking Water Act. The EPA and CDC recommend that private well owners test their water at least once for arsenic, and more frequently in regions with known geological arsenic deposits. State health departments often provide lists of certified laboratories. Testing costs range from $15–$50 for total arsenic. Homeowners in high-risk areas (the Southwest, Mountain West, New England) should test regardless of water taste or appearance, as arsenic is colorless, odorless, and tasteless.
7. Arsenic in Food
Rice: Why It Accumulates Arsenic
Rice (Oryza sativa) accumulates inorganic arsenic more efficiently than other cereal grains for two reasons. First, rice is grown in flooded anaerobic paddies where soil arsenic is mobilized into porewater in the reduced arsenite form. Second, rice roots express silicon/nodulin 26-like intrinsic protein (NIP) aquaglyceroporins that facilitate arsenite uptake alongside silicic acid — arsenic "hijacks" a nutrient transporter. Once inside the plant, arsenic moves through the xylem to the grain.
Inorganic arsenic concentrations in rice typically range from 0.1–0.4 mg/kg dry weight, but can exceed 0.6 mg/kg in rice from highly contaminated regions. Brown rice consistently contains more inorganic arsenic than white rice because the bran layer — removed during milling — is where arsenic concentrates. Basmati rice (especially from India, Pakistan, and California) generally has lower arsenic than long-grain varieties from the southern United States (Arkansas, Louisiana, Mississippi, Texas), where historical cotton cultivation with arsenical pesticides has elevated soil arsenic.
Cooking Methods to Reduce Arsenic
- High water-to-rice ratio ("pasta method"): Cooking rice in a large excess of water (6–10 parts water per 1 part rice) and draining can reduce inorganic arsenic by 40–60%, though some water-soluble vitamins and minerals are also lost.
- Pre-soaking: Soaking rice overnight and discarding the water before cooking removes approximately 20–30% of inorganic arsenic.
- Percolation cooking: Allowing water to continuously pass through rice (a method tested by researchers) achieves up to 85% arsenic reduction.
- Rinsing: Rinsing raw rice before cooking removes surface starch but has minimal effect on inorganic arsenic embedded in the grain matrix.
The FDA recommends that infants receive varied grain cereals rather than rice cereal exclusively, and that pregnant women moderate rice consumption, particularly from high-arsenic varieties.
Apple Juice and Baby Food
The FDA's action level for inorganic arsenic in apple juice is 10 µg/L. Testing by Consumer Reports (2011–2013) found that approximately 10% of apple juice samples exceeded this threshold. Apple juice is a significant exposure source for young children who consume it daily. The FDA has also proposed action levels for inorganic arsenic in infant rice cereal (100 µg/kg), acknowledging the disproportionate exposure of infants relative to body weight.
Seaweed: Hijiki Advisory
Hijiki (Sargassum fusiforme), a brown seaweed popular in Japanese cuisine, contains exceptionally high concentrations of inorganic arsenic — typically 40–100 mg/kg dry weight, several orders of magnitude above other seaweeds. Canada, the UK, Australia, and New Zealand have issued advisories recommending avoidance of hijiki. Other seaweeds (nori, kombu, wakame) contain predominantly organic arsenosugars, which are considered far less toxic. Japan, where hijiki is widely consumed, has not issued a formal advisory but acknowledges the inorganic arsenic content.
8. Diagnosis and Biomonitoring
Urinary Arsenic (Speciated)
Urine is the preferred matrix for assessing recent arsenic exposure. Total urinary arsenic, however, is confounded by dietary intake of organic seafood arsenicals (arsenobetaine, arsenocholine, arsenosugars), which are rapidly excreted and toxicologically irrelevant. Speciated urinary arsenic — measuring inorganic arsenic plus its methylated metabolites (MMA and DMA) separately from organic seafood arsenicals — is the gold standard. Results are expressed as micrograms per gram creatinine to correct for urine dilution. Reference ranges vary by laboratory; the 95th percentile for U.S. adults (NHANES) for speciated inorganic arsenic plus metabolites is approximately 5–10 µg/g creatinine. Elevated values reflect exposure within the prior few days to weeks.
Hair and Nail Analysis
Hair and fingernails incorporate arsenic during growth, providing a historical record of exposure. Arsenic concentration in hair reflects exposure approximately 1–4 months prior (based on hair growth rate of ~1 cm/month); the distal end of a long hair sample represents older exposure. Normal hair arsenic is below 1 µg/g dry weight; values above 1–3 µg/g suggest elevated chronic exposure. External contamination is a significant analytical challenge — arsenic from shampoo, dust, or medicinal hair products can adsorb onto the hair shaft and must be removed by standardized washing protocols before analysis. Fingernail arsenic reflects a 3–6 month exposure window.
Blood Arsenic
Blood arsenic reflects very recent (acute) exposure and clears within days to weeks of cessation of exposure. It is useful for diagnosing acute arsenic poisoning but has limited utility for assessing chronic low-level exposure, where urine is far more informative. Blood arsenic is expressed as micrograms per liter (µg/L); normal values in unexposed populations are generally below 2 µg/L.
9. Treatment and Detoxification
Chelation Therapy
For acute arsenic poisoning, chelation therapy is the primary medical intervention. British Anti-Lewisite (BAL, dimercaprol) was developed in World War II as an antidote to arsenic-based chemical weapons and remains useful for severe acute poisoning, administered intramuscularly. DMSA (meso-2,3-dimercaptosuccinic acid; succimer) is an oral chelator approved in the United States for lead poisoning in children and used off-label for arsenic; it forms stable, water-soluble complexes with arsenic that are renally excreted. DMPS (2,3-dimercaptopropane-1-sulfonate) is used in Europe and has shown efficacy in arsenic poisoning. Chelation for chronic low-level arsenic exposure is not established as effective and is not recommended outside of clinical trials.
Dietary Selenium Protection
Selenium exhibits a well-documented protective relationship against arsenic toxicity. The two elements form an insoluble selenide-arsenide complex in tissues, reducing the biological activity of both. Animal studies consistently show that selenium supplementation reduces arsenic-induced carcinogenesis, oxidative stress, and genotoxicity. Epidemiological evidence in Bangladesh found that higher selenium nutritional status was associated with reduced arsenicosis severity and lower cancer risk in arsenic-exposed populations. Selenium-rich foods include Brazil nuts, seafood, organ meats, and whole grains.
Folate and Methylation Support
Because arsenic methylation depends on SAM-derived methyl groups, adequate folate status is critical for efficient arsenic detoxification. Folate deficiency impairs the one-carbon metabolism cycle that regenerates SAM, slowing arsenic methylation and increasing retention of the more toxic inorganic forms. Clinical studies in Bangladesh showed that folate supplementation increased the proportion of arsenic excreted as DMA and reduced the proportion of the more toxic MMA. Adequate intake of vitamin B12, riboflavin, and choline — all involved in one-carbon metabolism — may similarly support arsenic methylation efficiency.
Chlorella
Chlorella (Chlorella vulgaris), a freshwater green microalga, has demonstrated arsenic-binding properties in animal and in vitro studies. Its cell wall contains sporopollenin-like compounds capable of adsorbing heavy metals including arsenic in the gastrointestinal tract, potentially reducing absorption. Animal studies show reduced arsenic tissue accumulation and improved oxidative stress markers with chlorella supplementation. Human clinical evidence remains limited, but chlorella is used as a supportive nutraceutical in some integrative protocols for heavy metal exposure. Chlorella should be sourced from certified suppliers to ensure it is free of contamination.
10. Prevention and Reduction Strategies
Water Filtration
Multiple point-of-use filtration technologies effectively remove arsenic from drinking water:
- Reverse osmosis (RO): The most effective household technology, removing 90–95% or more of arsenic (both arsenite and arsenate). Requires pre-filtration to protect the membrane and produces a reject water stream (approximately 3–4 gallons of waste per gallon of treated water).
- Activated alumina: An adsorptive media that is highly effective for arsenate (As(V)), removing 95%+ under optimal pH (5.5–6.0). Less effective for arsenite, so oxidation of As(III) to As(V) by chlorination or aeration prior to treatment significantly improves performance. Used in both point-of-use and community-scale systems.
- Iron-based media: Granular ferric hydroxide and similar materials adsorb arsenic effectively at near-neutral pH and are widely used in community water treatment globally.
- Distillation: Highly effective but energy-intensive.
- Standard pitcher filters (activated carbon): Not effective for arsenic removal and should not be relied upon in arsenic-affected areas.
Point-of-use filters require regular maintenance and filter replacement per manufacturer specifications; exhausted media can release arsenic back into treated water.
Rice Preparation
As described in Section 7, cooking rice with excess water and draining reduces inorganic arsenic by 40–60%. Diversifying grain intake — substituting quinoa, millet, buckwheat, oats, and other grains for some rice servings — meaningfully reduces cumulative arsenic exposure without sacrificing dietary variety. For infants, the FDA recommends varied grain cereals rather than an exclusive reliance on rice-based products.
Food Diversification
Reducing reliance on any single arsenic-accumulating food — rice, apple juice, or certain seafood — lowers total dietary exposure. Preferring basmati rice from India or California over U.S. long-grain varieties reduces arsenic intake. Choosing low-arsenic juice alternatives (grape, orange, pineapple) for children, particularly as a primary beverage, reduces daily arsenic load. Reading test results from FDA's Total Diet Study and advocacy organization monitoring (Consumer Reports, Environmental Working Group) provides current data on arsenic in specific food products.
11. Related Topics
12. References
- IARC Working Group. Arsenic, Metals, Fibres, and Dusts. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Vol. 100C. Lyon: IARC; 2012. Available at: https://monographs.iarc.fr/wp-content/uploads/2018/06/mono100C-6.pdf
- Smedley PL, Kinniburgh DG. A review of the source, behaviour and distribution of arsenic in natural waters. Applied Geochemistry. 2002;17(5):517–568.
- Argos M, Kalra T, Rathouz PJ, et al. Arsenic exposure from drinking water, and all-cause and chronic-disease mortalities in Bangladesh (HEALS): a prospective cohort study. Lancet. 2010;376(9737):252–258.
- Wasserman GA, Liu X, Parvez F, et al. Water arsenic exposure and children's intellectual function in Araihazar, Bangladesh. Environmental Health Perspectives. 2004;112(13):1329–1333.
- Navas-Acien A, Sharrett AR, Silbergeld EK, et al. Arsenic exposure and cardiovascular disease: a systematic review of the epidemiologic evidence. American Journal of Epidemiology. 2005;162(11):1037–1049.
- Tseng WP. Effects and dose-response relationships of skin cancer and Blackfoot disease with arsenic. Environmental Health Perspectives. 1977;19:109–119.
- Steinmaus C, Moore LE, Shipp M, et al. Genetic polymorphisms in MTHFR 677 and 1298, GSTM1 and T1, and metabolism of arsenic. Journal of Toxicology and Environmental Health Part A. 2007;70(2):159–170.
- Gamble MV, Liu X, Ahsan H, et al. Folate and arsenic metabolism: a double-blind, placebo-controlled folic acid-supplementation trial in Bangladesh. American Journal of Clinical Nutrition. 2006;84(5):1093–1101.
- Karagas MR, Punshon T, Sayarath V, et al. Association of rice and rice-product consumption with arsenic exposure early in life. JAMA Pediatrics. 2016;170(6):609–616.
- Cubadda F, Jackson BP, Cottingham KL, Van Horne YO, Kurzius-Spencer M. Human exposure to dietary inorganic arsenic and other arsenic species: State of knowledge, gaps and uncertainties. Science of the Total Environment. 2017;579:1228–1239.
- Carey M, Meharg C, Williams P, et al. Global sourcing of low-inorganic arsenic rice grain. Exposure and Health. 2020;12:711–719.
- Flanagan SV, Johnston RB, Zheng Y. Arsenic in tube well water in Bangladesh: health and economic impacts and implications for arsenic mitigation. Bulletin of the World Health Organization. 2012;90(11):839–846.
- Nair AR, Degheselle O, Smeets K, Van Kerkhove E, Cuypers A. Cadmium-induced pathologies: where is the oxidative balance lost (or not)? International Journal of Molecular Sciences. 2013;14(3):6116–6143.
- Ratnaike RN. Acute and chronic arsenic toxicity. Postgraduate Medical Journal. 2003;79(933):391–396.
- Mandal BK, Suzuki KT. Arsenic round the world: a review. Talanta. 2002;58(1):201–235.
- Navas-Acien A, Silbergeld EK, Pastor-Barriuso R, Guallar E. Arsenic exposure and prevalence of type 2 diabetes in US adults. JAMA. 2008;300(7):814–822.
- Vahter M. Mechanisms of arsenic biotransformation. Toxicology. 2002;181–182:211–217.
- Bhattacharya P, Ramanathan AL, Mukherjee AB, et al. Arsenic in groundwater of the Bengal Delta Plain aquifers in Bangladesh and West Bengal, India. Journal of the Bangladesh Academy of Sciences. 2006. Available at:.
- Uchino T, Roychowdhury T, Roychowdhury A, Tokunaga H, Ando M. Contamination of arsenic in groundwater in the Jessore district of Bangladesh. Chemosphere. 2006;62(1):101–109.
- Yoshida T, Yamauchi H, Fan Sun G. Chronic health effects in people exposed to arsenic via the drinking water: dose-response relationships in review. Toxicology and Applied Pharmacology. 2004;198(3):243–252.
Connections
- Heavy Metals
- Selenium
- Chlorella
- Pesticides
- Lead
- Cadmium
- Mercury
- Cancer
- Bladder Cancer
- Cardiovascular Disease
- Diabetes
- Glutathione
- Oxidative Stress
- Detoxification
- Detox Protocols
- Spirulina
- Zinc
- Fluoride