Oxidative Stress: Free Radicals, Antioxidants, and How to Test
Oxidative stress is the imbalance between the production of reactive oxygen species (ROS) and the body's capacity to neutralize them with antioxidant defenses. A small amount of ROS production is normal and necessary — cells use peroxide bursts for signaling, immune killing, and metabolic regulation. The trouble starts when ROS production chronically exceeds the antioxidant systems that contain them, damaging lipids, proteins, and DNA. Decades of research have linked sustained oxidative stress to cardiovascular disease, neurodegeneration, cancer initiation, accelerated aging, infertility, and chronic inflammation.
This page covers what oxidative stress actually is at the molecular level, the antioxidant systems that defend against it, the lifestyle and dietary inputs that change the balance, the supplements with the strongest evidence, and the lab tests that can quantify the damage.
Table of Contents
- What Oxidative Stress Is
- Sources of Reactive Oxygen Species
- The Body's Antioxidant Systems
- Consequences of Chronic Oxidative Stress
- Lifestyle Drivers and Reducers
- Dietary Antioxidants
- Supplements with the Strongest Evidence
- Testing Oxidative Stress in the Lab
- The Antioxidant Paradox
- A Practical Protocol
- Research Papers and References
- Connections
What Oxidative Stress Is
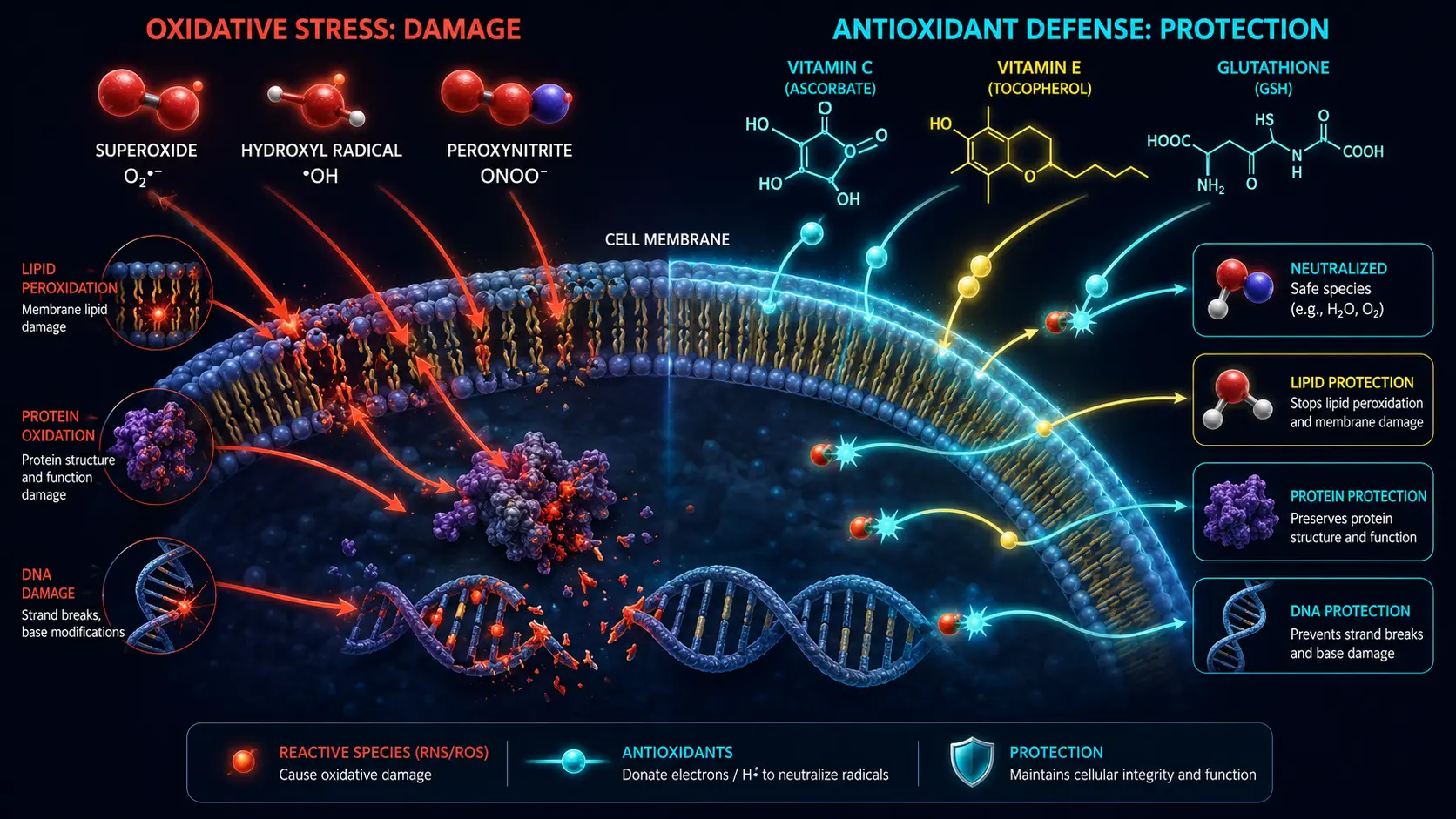
Reactive oxygen species are oxygen-containing molecules with an unpaired electron, making them chemically aggressive. The major species:
- Superoxide (O₂·⁻) — produced primarily as a byproduct of mitochondrial electron transport (about 1–2% of consumed oxygen leaks as superoxide)
- Hydrogen peroxide (H₂O₂) — less reactive but highly diffusible; functions as a signaling molecule at low levels
- Hydroxyl radical (•OH) — the most damaging; formed when H₂O₂ reacts with iron or copper (Fenton reaction)
- Peroxynitrite (ONOO⁻) — from superoxide and nitric oxide; particularly damaging to mitochondria and proteins
- Singlet oxygen (¹O₂) — formed in skin from UV exposure and from neutrophil activity
Reactive nitrogen species (RNS), reactive sulfur species, and lipid peroxidation products (4-HNE, malondialdehyde) operate alongside ROS and are sometimes lumped under the broader term "reactive species" or "redox stress." A balanced cell handles all of these continuously through a layered set of antioxidant defenses.
Sources of Reactive Oxygen Species
Endogenous (internal)
- Mitochondrial respiration — the largest single source; complexes I and III leak superoxide
- NADPH oxidase (NOX) — in neutrophils, macrophages, and vascular cells; deliberate ROS production for signaling and antimicrobial defense
- Xanthine oxidase — uric acid metabolism; major source after ischemia and reperfusion
- Cytochrome P450 enzymes — drug and xenobiotic metabolism (especially alcohol and acetaminophen)
- Peroxisomal β-oxidation — processing very-long-chain fatty acids
- Inflammation — activated immune cells generate large ROS bursts to kill pathogens; chronic inflammation means chronic ROS
Exogenous (external)
- Smoking — the most concentrated single oxidative insult most adults can voluntarily eliminate
- Air pollution — PM2.5, ozone, nitrogen oxides
- UV and ionizing radiation — sunlight, medical imaging, cosmic rays
- Heavy metals — lead, mercury, cadmium, arsenic catalyze Fenton chemistry
- Pesticides and industrial solvents
- Heat-treated and processed foods — advanced glycation end-products (AGEs), oxidized lipids, acrylamide
- Excessive iron — high serum ferritin, hemochromatosis, and excess iron supplementation
- Excessive alcohol — CYP2E1 induction and acetaldehyde
The Body's Antioxidant Systems
The body has three layered antioxidant defenses:
Enzymatic antioxidants (the first responders)
- Superoxide dismutase (SOD) — converts superoxide to hydrogen peroxide; copper/zinc-dependent (cytoplasm) and manganese-dependent (mitochondria)
- Catalase — converts hydrogen peroxide to water and oxygen; iron-dependent
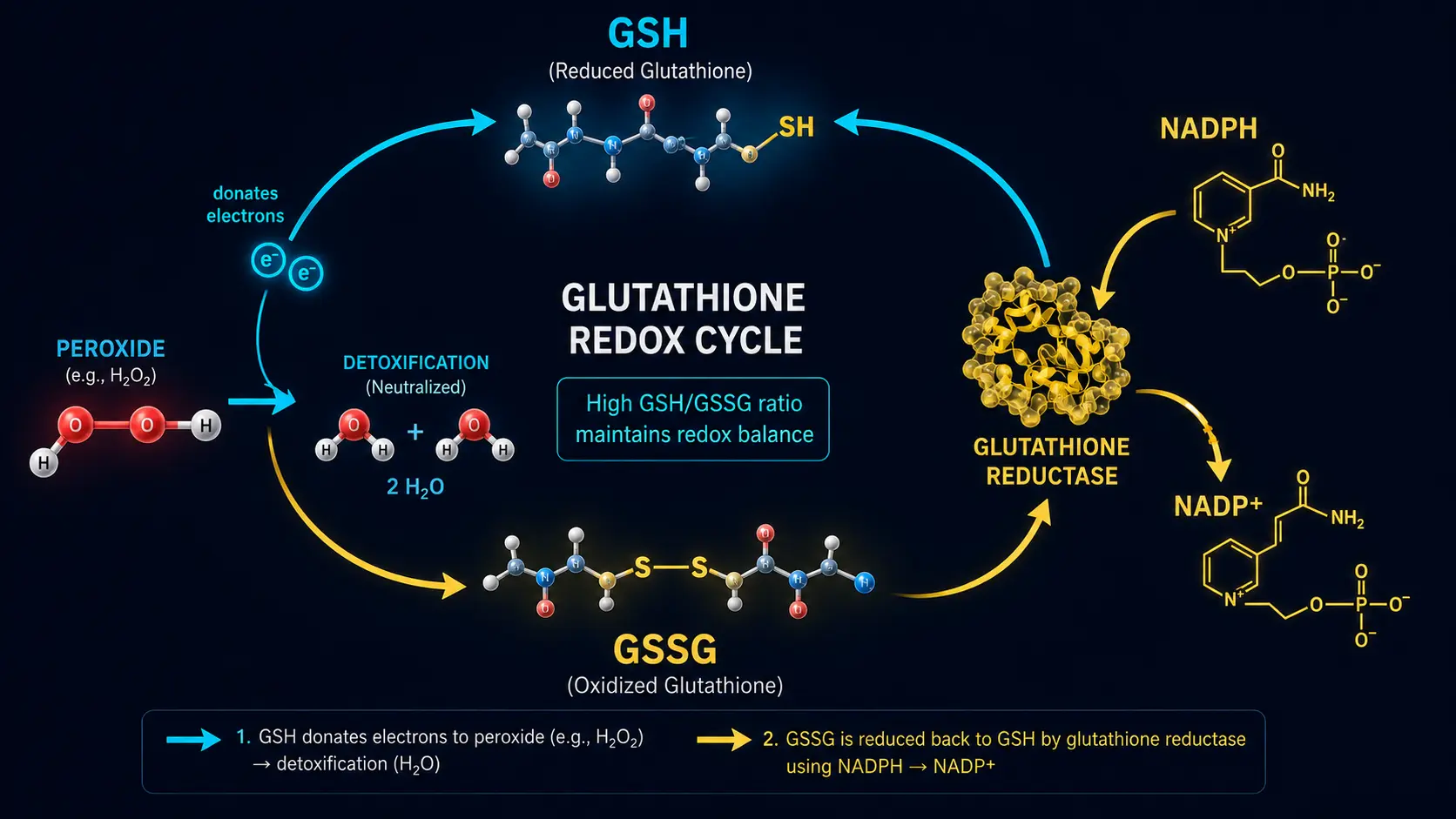
- Glutathione peroxidase — converts hydrogen peroxide to water using glutathione; selenium-dependent
- Glutathione reductase — recycles oxidized glutathione (GSSG) back to reduced glutathione (GSH) using NADPH
- Thioredoxin reductase — another major redox-cycling enzyme
Small-molecule antioxidants (the heavy lifters)
- Glutathione (GSH) — the cell's primary non-enzymatic antioxidant; tripeptide of glycine, cysteine, glutamate
- Vitamin C (ascorbate) — water-soluble; primary antioxidant in plasma and cytoplasm
- Vitamin E (alpha-tocopherol) — lipid-soluble; primary antioxidant in cell membranes
- Coenzyme Q10 (ubiquinol) — mitochondrial electron transport and lipid antioxidant
- Uric acid — major plasma antioxidant; the upside of moderate uric acid
- Bilirubin — underrated lipid-phase antioxidant
- Alpha-lipoic acid — both water- and lipid-soluble; recycles other antioxidants
Mineral cofactors
Selenium, zinc, copper, manganese, and iron are all required cofactors for the antioxidant enzymes above. Deficiency in any of them impairs the system.
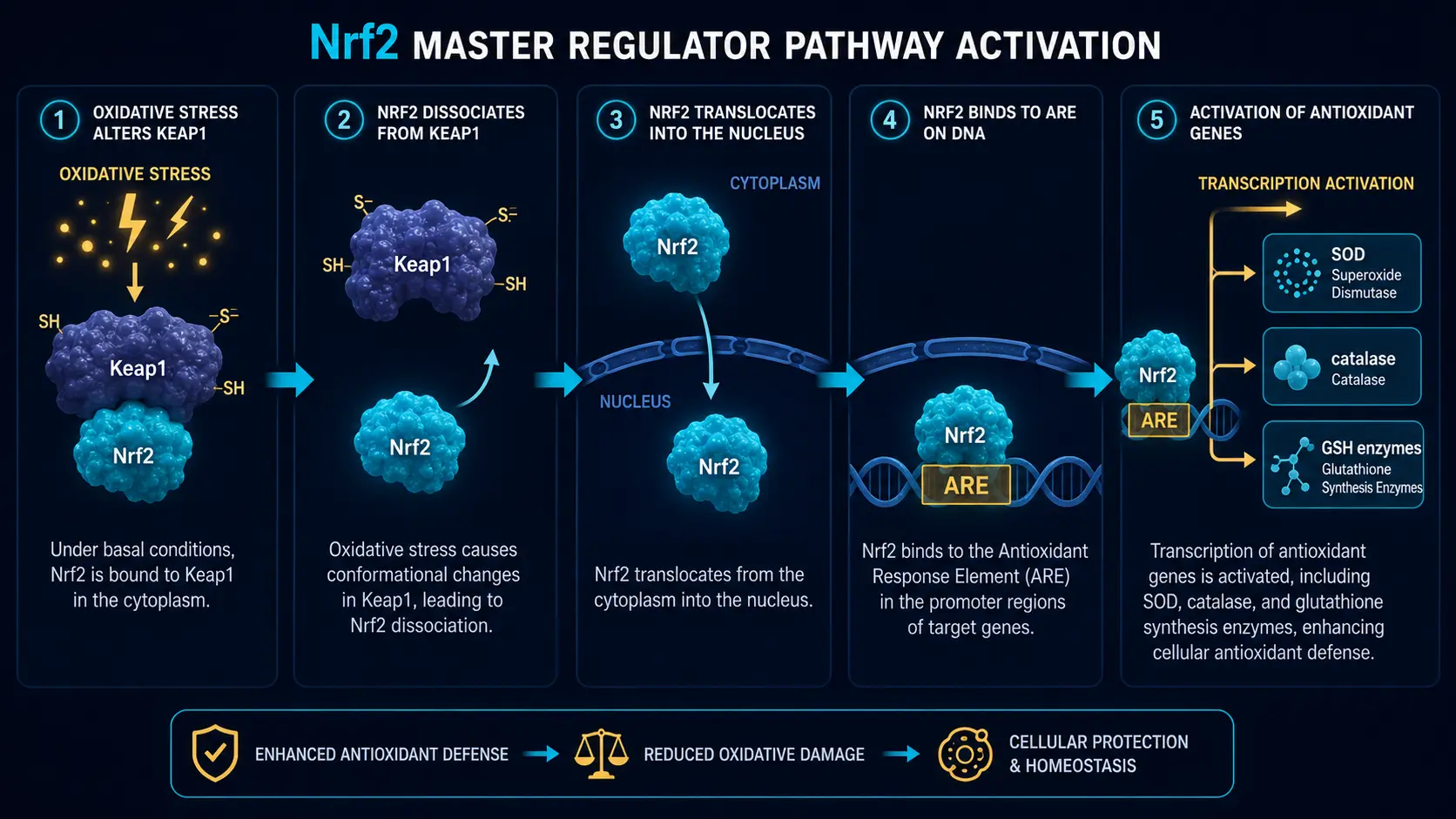
Master regulators
The Nrf2 transcription factor is the central conductor: when oxidative stress rises, Nrf2 enters the nucleus and switches on the genes for SOD, catalase, glutathione peroxidase, glutathione synthesis enzymes, and roughly 200 other antioxidant and detoxification genes. Nrf2 activation is the single most important mechanism by which dietary polyphenols (sulforaphane in broccoli, curcumin, EGCG in green tea, resveratrol) confer their oxidative-stress benefits.
Consequences of Chronic Oxidative Stress
- Lipid peroxidation — oxidation of cell membrane lipids; produces 4-HNE and malondialdehyde (MDA); a major driver of atherosclerosis (oxidized LDL drives plaque formation)
- Protein damage — oxidation of cysteine, methionine, tyrosine residues; protein carbonylation; aggregates implicated in Alzheimer's and Parkinson's
- DNA damage — 8-oxo-deoxyguanosine (8-OHdG) is the most measured oxidative DNA lesion; misrepair drives mutation and cancer
- Mitochondrial dysfunction — ROS damages the mitochondria that produce them, creating a self-amplifying loop
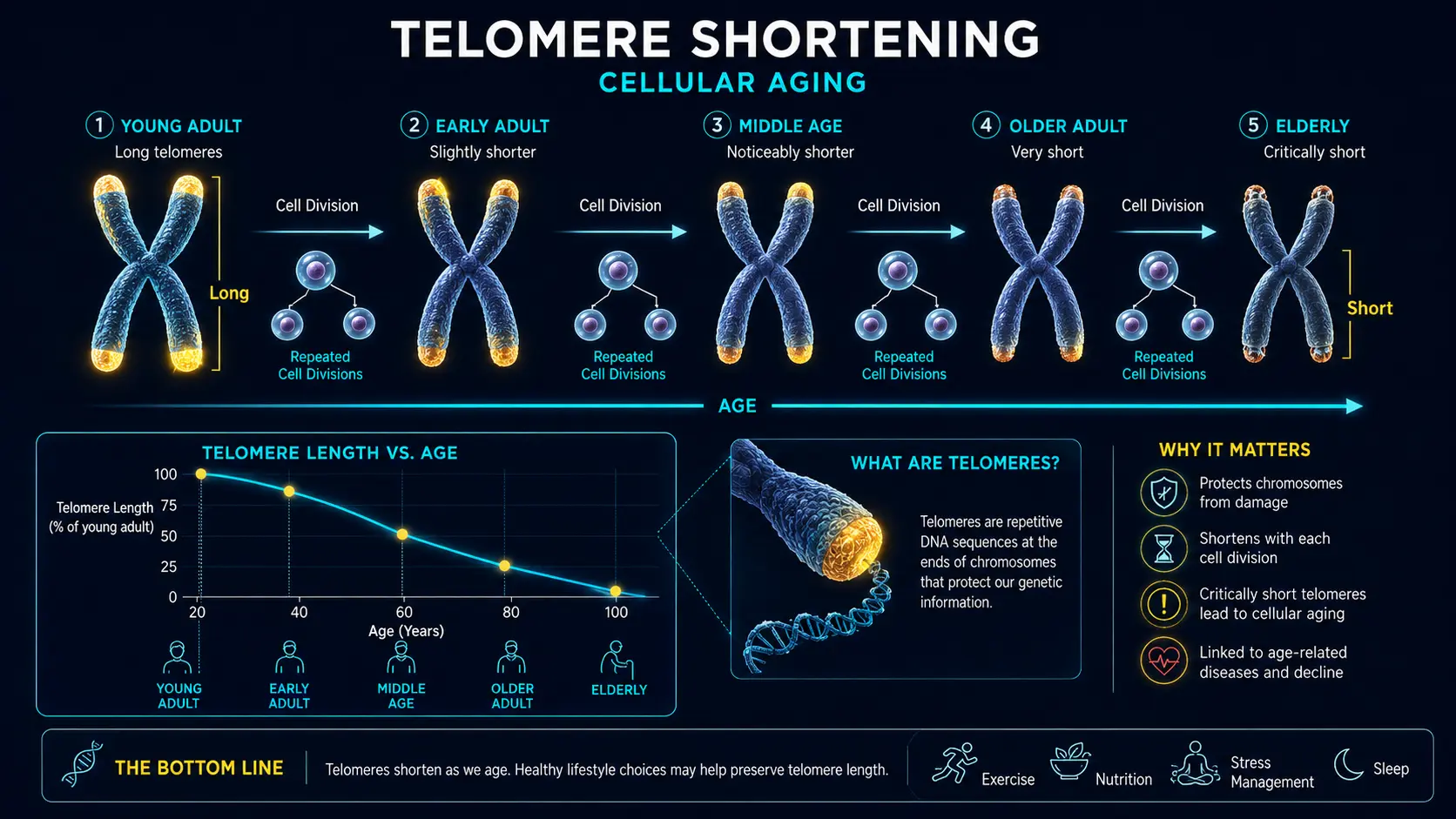
- Telomere shortening — accelerated aging
- Inflammation — oxidative stress activates NF-κB, the master inflammatory transcription factor; inflammation creates more ROS — the inflammation-oxidation loop
Conditions where oxidative stress is a documented contributor: cardiovascular disease, type 2 diabetes, non-alcoholic fatty liver disease, COPD, age-related macular degeneration, infertility (sperm DNA damage), Alzheimer's, Parkinson's, ALS, multiple sclerosis, and many cancers.
Lifestyle Drivers and Reducers
Things that increase oxidative stress
- Smoking and second-hand smoke
- Excess alcohol (more than 1 drink/day women, 2 drinks/day men)
- Chronic poor sleep (< 6 hours)
- Sedentary lifestyle
- Visceral obesity
- Chronic psychological stress (cortisol & catecholamine effects)
- Excessive endurance training without adequate recovery
- High-glycemic, ultra-processed Western diet
- Industrial seed oils oxidized at high heat
- Air pollution and indoor PM2.5 (e.g., gas stoves without ventilation)
Things that reduce oxidative stress
- Regular moderate exercise — increases mitochondrial biogenesis and Nrf2 activation, net antioxidant effect
- Adequate sleep (7–9 hours)
- Caloric restriction or time-restricted eating
- A diet rich in colorful plants, polyphenols, and cruciferous vegetables
- Heat exposure (sauna) and cold exposure — mild stressors that upregulate antioxidant defenses
- Smoking cessation
- Air filtration, especially in urban or wildfire environments
Dietary Antioxidants
Whole-food antioxidants outperform isolated supplements in nearly every long-term outcome study. Highest-impact food categories:
- Cruciferous vegetables — broccoli, kale, cabbage, cauliflower, Brussels sprouts; activate Nrf2 via sulforaphane
- Berries — blueberries, raspberries, blackberries, strawberries; anthocyanins, ellagic acid
- Dark leafy greens — spinach, kale; carotenoids, chlorophyll, polyphenols
- Tomatoes — lycopene; bioavailability boosted by cooking with olive oil
- Tea, especially green — EGCG; Green Tea
- Coffee — chlorogenic acid and other polyphenols; the largest single source of polyphenols in most Western diets
- Dark chocolate (≥ 70%) — flavanols
- Spices — turmeric (curcumin), cinnamon, oregano, cumin (gram-for-gram, the most concentrated polyphenol source available)
- Olive oil, extra-virgin — oleocanthal and oleuropein
- Nuts & seeds — walnuts, almonds; vitamin E, polyphenols
- Fatty fish — salmon, sardines; omega-3s reduce inflammatory ROS production
Supplements with the Strongest Evidence
- N-Acetylcysteine (NAC) — precursor to glutathione; well-studied for liver protection, COPD, fertility, mental health
- Glutathione (oral or liposomal) — direct GSH supplementation; bioavailability variable
- Alpha-lipoic acid — 300–600 mg/day; recycles vitamins C and E; used in diabetic neuropathy
- Vitamin C — 500–2000 mg/day; modest baseline benefit, larger benefit in smokers and those eating few plants
- Vitamin E (mixed tocopherols) — 200–400 IU/day; avoid pure alpha-tocopherol at high doses
- Selenium — 100–200 mcg/day; required for glutathione peroxidase; do not exceed 400 mcg total
- CoQ10 (ubiquinol) — 100–300 mg/day; mitochondrial support; especially valuable in statin users
- Quercetin — 250–500 mg/day; mast-cell stabilizer with antioxidant properties
- Curcumin — 500–1000 mg/day with piperine or phytosomal form for absorption
- Milk Thistle (silymarin) — for hepatic oxidative stress
Testing Oxidative Stress in the Lab
No single oxidative-stress lab test is universally accepted. The most clinically informative markers:
- 8-OHdG (urine or serum) — oxidized DNA marker; best-validated single index of cumulative oxidative DNA damage
- F2-isoprostanes (urine) — lipid peroxidation; gold-standard research marker, increasingly available clinically
- Malondialdehyde (MDA) — older lipid peroxidation marker; less specific
- 4-HNE (4-hydroxynonenal) — lipid peroxidation; research-grade
- GSH/GSSG ratio — the ratio of reduced to oxidized glutathione; a falling ratio indicates rising oxidative stress
- Total antioxidant capacity (TAC) — reasonable screening but non-specific
- Oxidized LDL — specific to vascular oxidative stress; useful in cardiovascular workup
- Inflammatory markers — hs-CRP and others — not direct oxidative-stress markers but tightly linked downstream
- Homocysteine — indicates methylation and B-vitamin status; elevated levels promote oxidative stress
For most outpatients, the highest-yield test combination is hs-CRP + homocysteine + GGT + serum ferritin + oxidized LDL. Direct oxidative markers (8-OHdG, F2-isoprostanes) are useful in research and select clinical situations but are not first-line.
The Antioxidant Paradox
An important caution: high-dose isolated-antioxidant supplementation has not consistently reduced cardiovascular events, cancer, or mortality in large randomized trials. Some studies have shown increased mortality with high-dose vitamin E, beta-carotene (in smokers), and selenium combined with vitamin E.
The likely explanation: ROS are not purely "bad." Cells use them for signaling, immune defense, exercise adaptation, and tumor surveillance. Wiping them out with mega-dose antioxidants disrupts these signals. The strategies that consistently work are the ones that upregulate the body's own antioxidant systems rather than flooding the system with exogenous antioxidants:
- Polyphenol-rich whole foods (Nrf2 activators)
- Regular moderate exercise (mitochondrial biogenesis)
- Heat and cold exposure (hormetic stress)
- Adequate sleep
- Smoking cessation, alcohol moderation
- Targeted supplementation in the presence of documented deficiency or specific clinical indications (NAC for COPD, CoQ10 for statin users, alpha-lipoic acid for diabetic neuropathy)
A Practical Protocol
For someone wanting to address oxidative stress without overcomplicating:
- Eliminate the largest sources first — smoking, excessive alcohol, ultra-processed food. Address these and most of the work is done.
- Move daily — 30 minutes of brisk walking is a robust Nrf2 activator
- Sleep 7–9 hours — non-negotiable
- Eat the rainbow — 30 different plants per week; aim for cruciferous and dark berries daily
- Spice every meal — turmeric, oregano, cinnamon, cloves are the densest dietary polyphenol sources
- Three cups of tea or coffee per day
- Targeted supplements only if indicated: NAC for liver/lung concerns, CoQ10 for statin users, magnesium for sleep, vitamin D for documented deficiency
- Test if a baseline number would change behavior — hs-CRP, homocysteine, ferritin, oxidized LDL
Research Papers and References
- Oxidative stress and ROS — PubMed search
- Nrf2 master regulator — PubMed search
- Glutathione redox marker — PubMed search
- F2-isoprostanes lipid peroxidation — PubMed search
- 8-OHdG DNA oxidation marker — PubMed search
- NAC and glutathione clinical trials — PubMed search
- High-dose vitamin E and mortality — PubMed search
- Exercise hormesis & mitochondria — PubMed search
- Sulforaphane and Nrf2 — PubMed search
- Oxidized LDL and atherosclerosis — PubMed search