Manganese – Essential Trace Mineral for Human Health
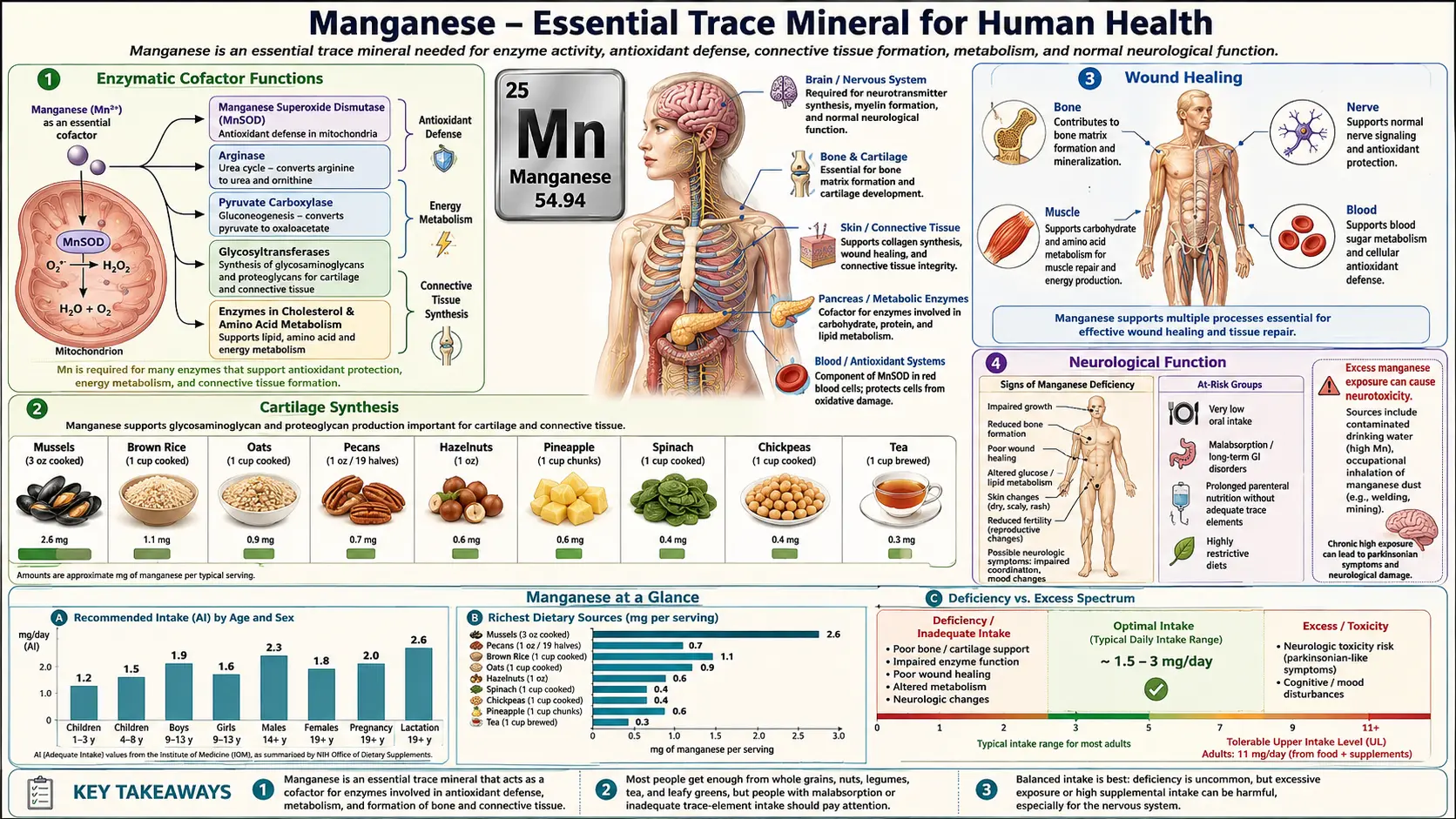
Manganese is an essential trace mineral that serves as a critical cofactor for numerous enzymes involved in bone formation, antioxidant defense, amino acid metabolism, carbohydrate metabolism, and reproductive function. The human body contains approximately 10 to 20 milligrams of manganese, concentrated primarily in the bones, liver, kidneys, and pancreas. The adequate intake (AI) for manganese is 2.3 milligrams per day for adult men and 1.8 milligrams per day for adult women, with a tolerable upper intake level (UL) of 11 milligrams per day.
Table of Contents
- Enzymatic Cofactor Functions
- Bone Formation
- Cartilage Synthesis
- Antioxidant Defense
- Amino Acid and Carbohydrate Metabolism
- Wound Healing
- Reproductive Function
- Neurological Function
- Connections
- Featured Videos
Enzymatic Cofactor Functions
Manganese is distinguished by its role as either a direct component of metalloenzymes or as an activating cofactor for a broad array of enzyme classes. Unlike some trace minerals that are associated with only a few enzymes, manganese participates in the catalytic activity of oxidoreductases, transferases, hydrolases, lyases, isomerases, and ligases.
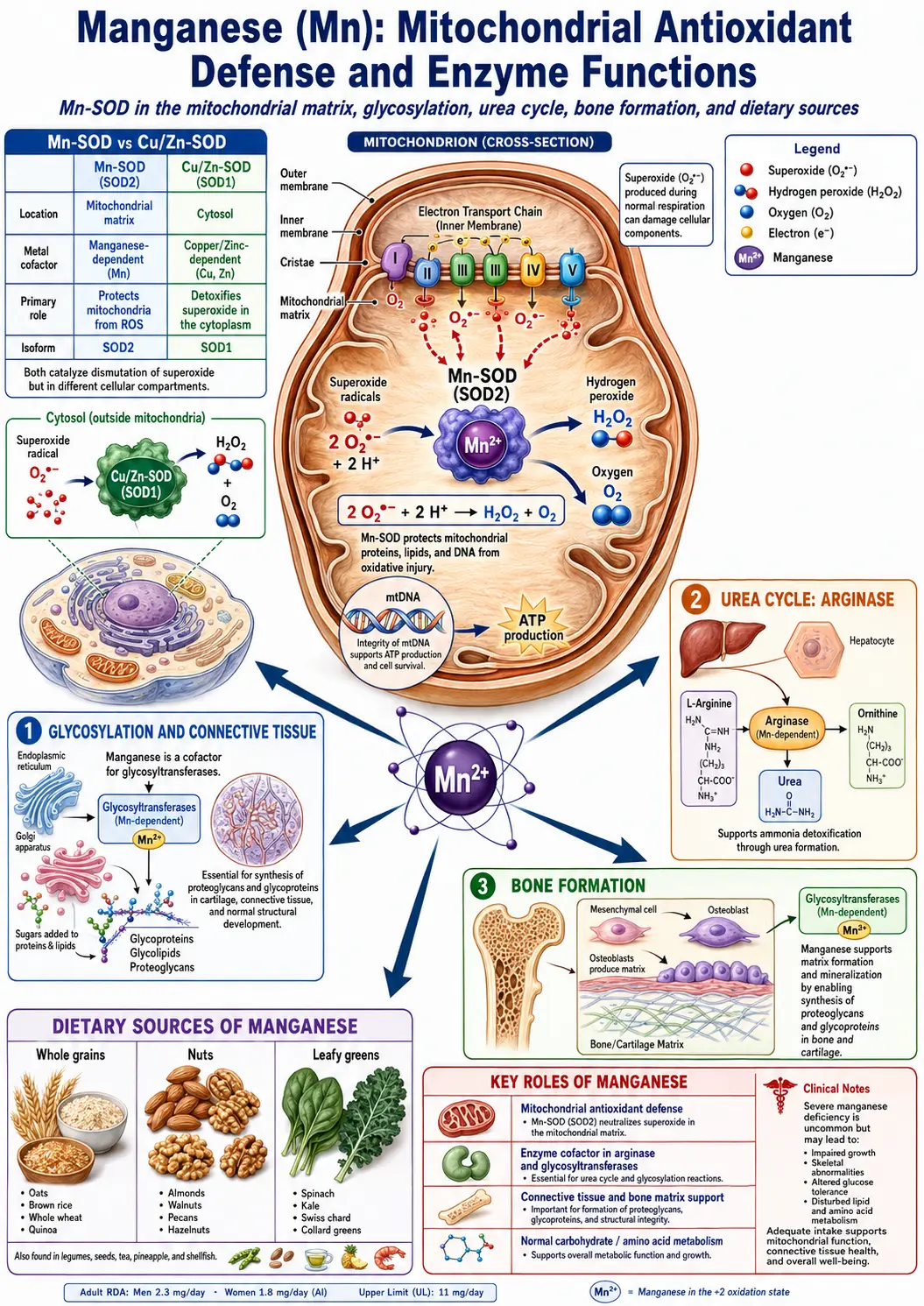
- Arginase: Arginase is a manganese-containing metalloenzyme that catalyzes the final step of the urea cycle, converting L-arginine to L-ornithine and urea. This reaction is essential for the detoxification of ammonia generated during amino acid catabolism. Each arginase molecule contains two manganese ions in its active site that are required for catalytic function. Without adequate manganese, urea cycle efficiency is compromised, potentially leading to ammonia accumulation.
- Manganese superoxide dismutase (MnSOD): MnSOD is a mitochondrial antioxidant enzyme that catalyzes the dismutation of superoxide radicals (O2-) into hydrogen peroxide (H2O2) and molecular oxygen. This reaction is the first and most critical step in the mitochondrial antioxidant defense system, as superoxide is the primary reactive oxygen species generated during oxidative phosphorylation. MnSOD contains one manganese ion per subunit and is essential for cellular survival; genetic knockout of MnSOD in animal models is lethal.
- Pyruvate carboxylase: Pyruvate carboxylase is a manganese-dependent enzyme that catalyzes the carboxylation of pyruvate to oxaloacetate, a critical anaplerotic reaction that replenishes tricarboxylic acid (TCA) cycle intermediates. This enzyme is also essential for gluconeogenesis, as oxaloacetate is the starting substrate for the synthesis of glucose from non-carbohydrate precursors in the liver and kidneys. Manganese is required at the active site for the biotin-dependent carboxylation mechanism.
- Glutamine synthetase: This enzyme, highly expressed in astrocytes of the brain, catalyzes the conversion of glutamate and ammonia to glutamine. It requires manganese for catalytic activity and plays a vital role in neurotransmitter recycling, ammonia detoxification in the central nervous system, and nitrogen metabolism.
- Glycosyltransferases: Manganese activates a family of glycosyltransferase enzymes that catalyze the transfer of sugar moieties to growing polysaccharide and glycoprotein chains. These enzymes are essential for the synthesis of proteoglycans, glycosaminoglycans, and glycoproteins, which are critical structural components of connective tissues including cartilage, bone matrix, and skin.
Bone Formation
Manganese plays an indispensable role in skeletal development and the maintenance of bone health throughout life. Its contribution to bone formation operates through several biochemical mechanisms that affect both the organic and inorganic components of bone tissue.
- Proteoglycan and glycosaminoglycan synthesis: Manganese-activated glycosyltransferases are essential for the synthesis of chondroitin sulfate, keratan sulfate, and other glycosaminoglycans that form the proteoglycan ground substance of bone and cartilage matrix. These molecules provide the structural framework upon which mineralization occurs and confer resilience and compressive strength to skeletal tissues.
- Osteoblast activity: Manganese supports the function of osteoblasts, the bone-forming cells responsible for synthesizing and depositing new bone matrix. Adequate manganese availability is necessary for osteoblasts to produce the full complement of matrix proteins and proteoglycans required for proper bone formation.
- Bone mineral density: Epidemiological studies have identified associations between low manganese intake and reduced bone mineral density, particularly in postmenopausal women. While manganese deficiency alone is unlikely to cause osteoporosis, suboptimal manganese status may contribute to impaired bone quality as a cofactor in the multifactorial etiology of osteoporosis.
- Skeletal abnormalities in deficiency: Animal studies have consistently demonstrated that manganese deficiency during development produces severe skeletal abnormalities, including shortened and thickened limb bones, joint deformities, and impaired endochondral ossification. These findings underscore the critical importance of manganese during periods of active skeletal growth.
Cartilage Synthesis
The synthesis and maintenance of healthy cartilage tissue is heavily dependent on manganese-requiring enzymatic processes. Cartilage is a specialized connective tissue that cushions joints, shapes the external ear and nose, and forms the structural template for long bone development through endochondral ossification.
- Chondroitin sulfate production: Chondroitin sulfate is a major glycosaminoglycan component of cartilage extracellular matrix. Its synthesis requires multiple manganese-activated glycosyltransferases that sequentially add sugar residues to the growing polysaccharide chain. Manganese deficiency impairs chondroitin sulfate production, leading to cartilage that is thinner, less resilient, and more susceptible to mechanical damage.
- Aggrecan assembly: Aggrecan is the predominant proteoglycan in articular cartilage, consisting of a core protein decorated with numerous chondroitin sulfate and keratan sulfate side chains. The assembly of this complex molecule depends on manganese-dependent glycosyltransferases. Aggrecan's ability to attract and retain water molecules gives cartilage its characteristic compressive stiffness and shock-absorbing properties.
- Chondrocyte function: Chondrocytes, the cells responsible for maintaining cartilage, require manganese for the synthesis and secretion of extracellular matrix components. Manganese also supports chondrocyte proliferation during cartilage growth and repair.
- Joint health implications: The dependence of cartilage integrity on manganese has led to interest in manganese supplementation as part of nutritional support for joint health, particularly in conditions such as osteoarthritis where cartilage degradation exceeds synthesis.
Antioxidant Defense
Manganese occupies a central position in the cellular antioxidant defense system, primarily through its role in manganese superoxide dismutase (MnSOD), but also through additional antioxidant mechanisms.
- Mitochondrial protection via MnSOD: Mitochondria are the primary source of intracellular reactive oxygen species (ROS), generated as byproducts of the electron transport chain during aerobic respiration. MnSOD, located exclusively in the mitochondrial matrix, intercepts superoxide radicals before they can damage mitochondrial DNA, proteins, and lipid membranes. The hydrogen peroxide produced by MnSOD is subsequently detoxified by glutathione peroxidase and catalase.
- Prevention of oxidative damage: By maintaining MnSOD activity, adequate manganese intake protects against oxidative damage to lipids (lipid peroxidation), proteins (carbonyl formation), and nucleic acids (8-hydroxy-2-deoxyguanosine formation). Cumulative oxidative damage to these macromolecules is implicated in aging, neurodegeneration, cardiovascular disease, and cancer.
- Non-enzymatic antioxidant activity: Free manganese ions (Mn2+) can function as non-enzymatic scavengers of superoxide and other reactive oxygen species. While this capacity is modest compared to MnSOD, it may provide supplementary antioxidant protection in cellular compartments where the free manganese concentration is appreciable.
- Interaction with other antioxidant systems: MnSOD functions as part of an integrated antioxidant network that includes copper-zinc superoxide dismutase (CuZnSOD) in the cytoplasm, catalase in peroxisomes, and the glutathione system. Adequate manganese status ensures that the mitochondrial component of this network functions optimally, complementing antioxidant defenses in other cellular compartments.
Amino Acid and Carbohydrate Metabolism
Manganese participates in the metabolism of both amino acids and carbohydrates through its roles as an enzyme cofactor and metalloenzyme component.
- Urea cycle and nitrogen disposal: Through its role in arginase, manganese is essential for the conversion of nitrogen waste from amino acid catabolism into urea for excretion by the kidneys. This function is critical for preventing the toxic accumulation of ammonia, particularly during periods of high protein intake or increased amino acid turnover.
- Gluconeogenesis: Manganese-dependent pyruvate carboxylase initiates the gluconeogenic pathway by converting pyruvate to oxaloacetate. This pathway is essential for maintaining blood glucose levels during fasting, prolonged exercise, and other states of glycogen depletion. Phosphoenolpyruvate carboxykinase (PEPCK), another key gluconeogenic enzyme, is also activated by manganese.
- TCA cycle anaplerosis: The pyruvate carboxylase reaction replenishes oxaloacetate in the TCA cycle, which is necessary to sustain the cycle when intermediates are drawn off for biosynthetic purposes such as amino acid synthesis, heme production, and gluconeogenesis. Without adequate manganese, the TCA cycle can become depleted of intermediates, impairing aerobic energy production.
- Amino acid synthesis: Manganese-activated enzymes participate in the biosynthesis of several non-essential amino acids, including proline and serine, and in the transamination reactions that interconvert amino acids. These processes are important for protein synthesis, collagen production, and neurotransmitter metabolism.
- Isocitrate dehydrogenase: Manganese serves as a cofactor for isocitrate dehydrogenase, which catalyzes the oxidative decarboxylation of isocitrate to alpha-ketoglutarate in the TCA cycle. This reaction generates NADH for the electron transport chain and produces a key intermediate for amino acid metabolism.
Wound Healing
Manganese contributes to wound healing through multiple mechanisms related to collagen synthesis, immune function, and antioxidant defense.
- Collagen production: Manganese activates prolidase, an enzyme that recycles proline from degraded collagen for use in new collagen synthesis. Proline and its hydroxylated form, hydroxyproline, are the most abundant amino acids in collagen, constituting approximately one-third of its residues. Adequate manganese is therefore necessary for efficient collagen turnover and deposition at wound sites.
- Extracellular matrix remodeling: The glycosaminoglycans and proteoglycans synthesized by manganese-dependent glycosyltransferases are essential components of the provisional wound matrix. This matrix provides a scaffold for cell migration, proliferation, and differentiation during the proliferative phase of wound healing.
- Inflammatory phase support: MnSOD plays a protective role during the inflammatory phase of wound healing by limiting oxidative damage caused by the respiratory burst of activated neutrophils and macrophages. While the respiratory burst is necessary for pathogen killing, excessive ROS production can damage surrounding healthy tissue and delay wound closure.
- Angiogenesis: Manganese may support the angiogenic process (formation of new blood vessels) that is essential for delivering oxygen and nutrients to the healing wound bed. Adequate antioxidant defense through MnSOD protects newly forming endothelial cells from oxidative damage during this vulnerable process.
Reproductive Function
Manganese is essential for normal reproductive function in both males and females, with deficiency producing significant reproductive impairment in animal models.
- Steroid hormone synthesis: Manganese is required for the synthesis of cholesterol, the precursor to all steroid hormones including estrogen, progesterone, and testosterone. Manganese-dependent enzymes participate in the mevalonate pathway of cholesterol biosynthesis, and deficiency can impair the production of reproductive hormones.
- Ovarian function: In female animals, manganese deficiency has been associated with impaired ovulation, irregular estrous cycles, and reduced fertility. These effects are attributed to inadequate steroid hormone production and impaired follicular development.
- Testicular function: Manganese deficiency in male animals leads to testicular degeneration, reduced spermatogenesis, and decreased testosterone production. The antioxidant role of MnSOD is particularly important in the testes, where high rates of cell division and oxygen consumption generate substantial ROS that must be neutralized to protect developing spermatozoa.
- Embryonic development: Manganese is critical during embryonic development for skeletal formation, central nervous system development, and organogenesis. Severe manganese deficiency during pregnancy in animal models results in skeletal malformations, ataxia, and increased embryonic mortality.
Neurological Function
Manganese has a complex relationship with neurological function. While it is essential for normal brain metabolism and neurotransmitter regulation, excessive accumulation in the brain is neurotoxic. Maintaining appropriate manganese homeostasis is therefore critical for neurological health.
- Glutamine synthetase in astrocytes: Manganese-dependent glutamine synthetase in brain astrocytes is essential for recycling the excitatory neurotransmitter glutamate. After glutamate is released into the synaptic cleft and reuptaken by astrocytes, glutamine synthetase converts it to glutamine, which is then transported back to neurons for reconversion to glutamate. This glutamate-glutamine cycle is fundamental to excitatory neurotransmission.
- Neurotransmitter metabolism: Manganese influences the synthesis and metabolism of several neurotransmitters, including dopamine, serotonin, and gamma-aminobutyric acid (GABA). Manganese-dependent enzymes participate in the biosynthetic pathways for these neurotransmitters, and alterations in manganese status can affect neurotransmitter balance.
- Mitochondrial function in neurons: Neurons are highly dependent on mitochondrial energy production, and MnSOD is essential for protecting neuronal mitochondria from oxidative damage. Impaired MnSOD function contributes to mitochondrial dysfunction, which is a recognized pathological feature of neurodegenerative diseases including Parkinson's disease, Alzheimer's disease, and amyotrophic lateral sclerosis.
- Manganism (toxicity): Chronic exposure to excessive manganese, particularly through inhalation in occupational settings such as mining, welding, and battery manufacturing, can lead to manganism, a neurotoxic condition resembling Parkinson's disease. Symptoms include tremor, rigidity, bradykinesia, psychiatric disturbances, and cognitive impairment. Manganism results from manganese accumulation in the basal ganglia, particularly the globus pallidus, where it disrupts dopaminergic neurotransmission. This toxicity highlights the importance of maintaining manganese intake within the recommended range.
- Dietary sources and balance: Manganese is found abundantly in whole grains, nuts, seeds, legumes, leafy green vegetables, tea, and pineapple. Most individuals consuming a varied diet obtain adequate manganese from food sources. Supplementation should be approached with caution and should not exceed the tolerable upper intake level of 11 milligrams per day for adults, given the potential for neurotoxicity at high doses.
Connections
- Manganese Benefits Deep Dive
- Manganese and Bone Formation
- Manganese MnSOD Antioxidant Defense
- Manganese for Wound Healing
- Manganese for Blood Sugar
- Osteoporosis
- Copper
- Zinc
- Magnesium
- Calcium
- Silicon
- Boron
- Phosphorus
- Vitamin D3
- Vitamin K
- Collagen
- Proline
- Arthritis
- Parkinson's Disease
- Copper-Iron Dysregulation
- Oxidative Stress
- Spinach