Ovarian Cysts
Ovarian cysts are fluid-filled or partially fluid-filled structures arising from or within the ovary, encountered across all age groups from neonates to post-menopausal women. The majority are benign and resolve spontaneously, but a systematic approach to characterization — using ultrasound morphology, validated risk-stratification systems, and clinical context — is essential to distinguish functional from pathological lesions and to identify the minority requiring surgical evaluation for suspected malignancy.
Table of Contents
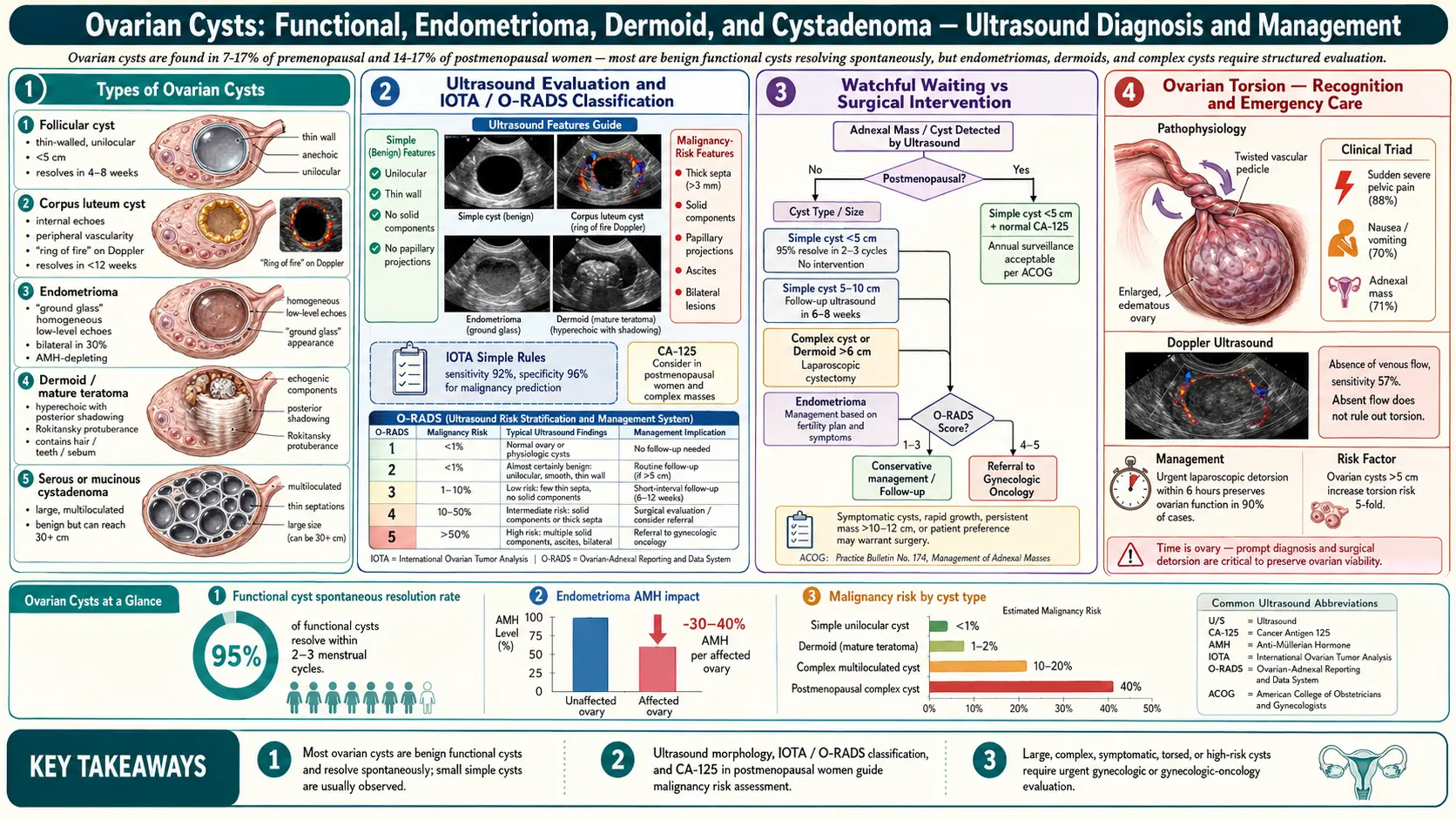
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research
- References
1. Overview
Ovarian cysts encompass a heterogeneous spectrum of lesions that require stratification into functional and pathological categories. Functional cysts (follicular and corpus luteum cysts) are physiological variants arising from normal ovarian cyclicity and represent the vast majority of cysts identified in premenopausal women. Pathological cysts include benign neoplasms (dermoid/mature teratoma, serous and mucinous cystadenoma, endometrioma) and malignant or borderline tumors requiring surgical evaluation and histopathological diagnosis.
Two principal risk-stratification frameworks guide clinical management:
- IOTA (International Ovarian Tumor Analysis) criteria: Evidence-based rules derived from prospective multicenter studies providing sonographic classification into Simple Rules (benign/malignant features), ADNEX model (quantitative malignancy probability), and LR2 logistic regression model
- O-RADS (Ovarian-Adnexal Reporting and Data System): ACR-endorsed standardized lexicon and five-level risk stratification (O-RADS 1–5) for sonographic and MRI assessment, providing management recommendations analogous to BI-RADS for breast lesions
2. Epidemiology
Ovarian cysts are among the most common incidental findings in gynecological practice and emergency imaging:
- Estimated lifetime risk of ovarian cyst diagnosis in women: approximately 8% requiring surgical evaluation
- Incidental adnexal cysts found in approximately 4–18% of post-menopausal women on transvaginal ultrasound
- Simple cysts under 3 cm are identified in approximately 14.8% of post-menopausal women and have near-zero malignancy risk
- Ovarian cancer accounts for approximately 239,000 new cases and 152,000 deaths annually worldwide; most ovarian cancers are diagnosed at advanced stage due to occult early disease
- Functional cysts comprise approximately 90% of adnexal masses in premenopausal women under 40 years
- Endometriomas affect approximately 10% of women in the general population and up to 40–50% of those with infertility
- Dermoid cysts (mature cystic teratomas) represent 10–20% of all ovarian neoplasms and are the most common ovarian tumor in women under 30 years
3. Pathophysiology
Functional Cysts
Follicular cysts arise when a dominant follicle fails to rupture at ovulation, continuing to enlarge beyond the normal 18–22 mm pre-ovulatory size, typically reaching 3–8 cm. Elevated LH levels (as in PCOS or exogenous gonadotropin stimulation) may precipitate multiple follicular cysts. Granulosa cell secretion of estradiol continues until spontaneous regression, which occurs in most cases within 1–3 menstrual cycles via atresia and follicular fluid resorption.
Corpus luteum cysts form when the corpus luteum fails to undergo normal luteolysis, persisting beyond 14 days and often reaching 3–10 cm. The corpus luteum is highly vascular (the most vascularized structure per unit volume in the body) and may hemorrhage internally, producing a hemorrhagic corpus luteum with complex echogenicity — often confused with endometrioma or ectopic pregnancy on ultrasound. Hemorrhagic corpus luteum cysts may rupture, causing hemoperitoneum, particularly in women on anticoagulation.
Endometrioma
Endometriomas ("chocolate cysts") arise from ectopic endometrial glands and stroma implanting on the ovarian cortex and invaginating into the ovarian parenchyma. Cyclical hemorrhage into the enclosed space produces hemosiderin-laden fluid with a characteristic "ground-glass" ultrasound appearance. The cyst wall lacks a true epithelial lining; the interface with the adjacent ovarian cortex is often indistinct, making enucleation technically challenging and predisposing to inadvertent excision of healthy ovarian follicles (reducing ovarian reserve). Endometriomas are estrogen-responsive and recur following surgical excision in up to 50% of cases within 5 years.
Dermoid Cysts (Mature Cystic Teratoma)
Arise from parthenogenetic development of a primordial germ cell, containing mature elements from two or three germ cell layers (ectoderm — skin, hair, sebaceous glands; mesoderm — fat, cartilage, bone; endoderm — thyroid tissue, bronchial epithelium). The sebaceous content appears echogenic with dense shadowing on ultrasound (Rokitansky nodule/dermoid plug). Malignant transformation to squamous cell carcinoma occurs in less than 2% of dermoid cysts, predominantly in post-menopausal women.
Serous and Mucinous Cystadenomas
Serous cystadenomas are lined by fallopian tube-type epithelium, typically unilocular or oligolocular with thin walls and anechoic contents. Serous borderline tumors (serous tumors of low malignant potential) may have micropapillary projections; distinction from low-grade serous carcinoma requires histological assessment. Mucinous cystadenomas may reach large dimensions (greater than 20 cm) and contain thick, mucinous fluid; they are multilocular with variable internal echogenicity. Pseudomyxoma peritonei — peritoneal dissemination of mucin-secreting epithelium — is a rare but serious complication, most commonly arising from appendiceal rather than primary ovarian mucinous tumors.
4. Etiology and Risk Factors
Factors Promoting Functional Cyst Formation
- Ovulation induction with clomiphene citrate or gonadotropins: multiple follicular cysts, ovarian hyperstimulation syndrome (OHSS)
- Polycystic ovary syndrome (PCOS): multiple small follicular cysts (2–9 mm) distributed peripherally — strictly "polycystic ovarian morphology" (PCOM) rather than true cysts
- Hypothyroidism: elevated TSH stimulates FSH-like activity via TSH receptor cross-reactivity, causing large follicular cysts
- Anticoagulant therapy: increases hemorrhagic corpus luteum cyst formation and rupture risk
Risk Factors for Pathological Ovarian Cysts and Malignancy
- Age: Malignancy risk increases substantially after menopause; BRCA1/2 mutation carriers have lifetime risk of 40–60% and 15–25% respectively for ovarian cancer
- Family history: First-degree relative with ovarian or breast cancer; Lynch syndrome (MLH1, MSH2, MSH6, PMS2 mutations) confers 5–10% lifetime ovarian cancer risk
- Nulliparity and infertility: Incessant ovulation theory supports increased surface epithelial trauma as a risk factor for epithelial ovarian carcinoma
- Hormone replacement therapy: Estrogen-only HRT increases borderline tumor and clear cell carcinoma risk
- Endometriosis: Endometriomas are precursors to endometrioid and clear cell carcinoma of the ovary (RR approximately 2.5–4 for malignant transformation)
- Protective factors: Oral contraceptive use reduces ovarian cancer risk by 40–50% per 5 years of use; parity; breastfeeding; tubal ligation; bilateral salpingectomy
5. Clinical Presentation
Asymptomatic Incidental Finding
The majority of ovarian cysts, particularly simple functional cysts under 5 cm, are detected incidentally on pelvic ultrasound performed for other indications (urinary tract symptoms, infertility workup, routine examination). Many resolve before the next menstrual cycle and require only follow-up imaging.
Pelvic Pain and Pressure
Enlarging cysts produce a sense of lower abdominal fullness, pressure, and unilateral pelvic aching. Pain may be cyclical with endometriomas (dysmenorrhea, deep dyspareunia) or constant with large neoplastic cysts compressing adjacent pelvic organs. Acute, severe unilateral pain should prompt immediate evaluation for torsion or rupture.
Menstrual Irregularity
Functional cysts may produce hormonal disturbance — follicular cysts secrete estrogen causing endometrial proliferation and delayed menstruation followed by irregular or heavy withdrawal bleeding; corpus luteum cysts maintain progesterone levels mimicking early pregnancy with amenorrhea. PCOM associated with PCOS produces oligomenorrhea and anovulatory cycles.
Ovarian Torsion
A gynecological emergency caused by partial or complete rotation of the ovary (with or without the fallopian tube) around the infundibulopelvic and utero-ovarian ligament axes, compromising vascular supply. Torsion occurs most commonly with cysts of 5–10 cm — large enough to tip the center of gravity but not so heavy as to prevent rotation. Classic presentation: acute onset severe unilateral lower abdominal pain (colicky then constant), nausea, vomiting, and low-grade fever. Physical examination reveals exquisite adnexal tenderness; an adnexal mass may be palpable. Torsion may be intermittent (partial torsion with spontaneous resolution and recurrence).
Cyst Rupture
Spontaneous or traumatic rupture (post-coital, post-exercise) results in intraperitoneal spillage. Simple cyst rupture typically produces transient pelvic pain that resolves within hours. Hemorrhagic corpus luteum rupture may cause hemoperitoneum with peritoneal signs, hypotension, and significant blood loss requiring surgical intervention, particularly in women on anticoagulants.
Features Suggesting Malignancy
Post-menopausal presentation, bilateral lesions, rapid growth, solid or complex ultrasound morphology, ascites, and elevated CA-125 are features warranting urgent specialist assessment. Paraneoplastic syndromes (hypercalcemia from small cell carcinoma; androgenic excess from Sertoli-Leydig cell tumors causing virilization) may present as systemic rather than pelvic symptoms.
6. Diagnosis
Transvaginal Ultrasound (TVUS)
The primary imaging modality for ovarian cyst assessment. Key descriptors include: cyst size (maximum diameter in three planes), morphology (unilocular, multilocular, solid, mixed), wall characteristics (thin/regular vs. irregular/thick), internal echogenicity (anechoic, ground-glass, mixed, hyperechoic), presence and characteristics of papillary projections or solid components, septation (number, thickness), and Doppler flow within solid components or septa.
IOTA Simple Rules
Validated ultrasound classification system using five benign features (B-rules) and five malignant features (M-rules):
Benign (B) features:
- B1: Unilocular cyst
- B2: Solid components where the largest solid component is less than 7 mm
- B3: Acoustic shadowing
- B4: Multilocular tumor with diameter less than 10 cm and smooth surface
- B5: No blood flow (color score 1)
Malignant (M) features:
- M1: Irregular solid tumor
- M2: Ascites
- M3: At least four papillary projections
- M4: Irregular multilocular solid tumor with diameter 10 cm or more
- M5: Very strong blood flow (color score 4)
If only B features are present: classify as benign (sensitivity 95%, specificity 74%). If only M features: classify as malignant (sensitivity 92%, specificity 96%). If both or neither: inconclusive — apply further assessment (ADNEX model, MRI, specialist referral).
IOTA ADNEX Model
A multivariate logistic regression model incorporating five ultrasound variables (maximum lesion diameter, proportion of solid tissue, number of papillary projections, more than 10 locules, acoustic shadows) and two clinical variables (patient age, CA-125, and clinical setting — oncology center vs. non-oncology). Outputs continuous probability estimates for five diagnostic categories: benign, borderline, stage I invasive, stage II–IV invasive, and secondary metastatic. An ADNEX risk above 10% for malignancy is generally used as a threshold for specialist referral.
O-RADS Ultrasound Classification
The ACR O-RADS ultrasound lexicon assigns risk categories:
- O-RADS 0: Incomplete evaluation — further imaging required
- O-RADS 1: Normal ovary — no cyst identified (almost zero malignancy risk)
- O-RADS 2: Almost certainly benign — physiological or almost certainly benign lesion (malignancy risk less than 1%); examples: simple unilocular cyst, typical hemorrhagic cyst, typical endometrioma less than 10 cm, dermoid
- O-RADS 3: Low malignancy risk (1–less than 10%); examples: simple cyst 10 cm or more, multilocular cyst without solid components, atypical hemorrhagic cyst
- O-RADS 4: Intermediate malignancy risk (10–less than 50%); examples: any cyst with papillary projections with or without flow, multilocular cyst with smooth solid component
- O-RADS 5: High malignancy risk (50% or greater); examples: irregular solid tumor, ascites with peritoneal nodules, strong Doppler flow in solid component
O-RADS MRI
MRI is the second-line modality for indeterminate lesions on ultrasound (O-RADS 3–4 or inconclusive IOTA Simple Rules). MRI O-RADS assigns categories 1–5 based on T1/T2 signal characteristics, enhancement patterns, restricted diffusion, and associated findings. Key MRI features: T1 hyperintensity (dermoid fat, endometrioma blood products), T2 "shading" (endometrioma), T2 hypointensity of fibrous solid components (fibroma, Brenner tumor), restricted diffusion in malignant solid components (ADC below 0.9 × 10⁻³ mm²/s), and early arterial enhancement.
Serum Biomarkers
- CA-125: Elevated (above 35 U/mL) in approximately 80% of advanced epithelial ovarian cancers but only 50% of stage I disease; elevated in numerous benign conditions (endometriosis, fibroids, PID, pregnancy, liver disease, pleural effusion). Menopausal status is critical — post-menopausal women with elevated CA-125 and complex adnexal mass have a high positive predictive value for malignancy
- HE4 (Human epididymis protein 4): More specific than CA-125 for epithelial ovarian cancer, not elevated in endometriosis; combined with CA-125 in the ROMA (Risk of Ovarian Malignancy Algorithm) or OVA1 panel improves sensitivity for malignancy
- AFP and beta-hCG: Elevated in germ cell tumors (yolk sac tumor, embryonal carcinoma, choriocarcinoma) — essential markers in women under 40 with complex solid ovarian masses
- LDH: Elevated in dysgerminoma
- Inhibin A/B: Elevated in granulosa cell tumors (sex cord-stromal tumors); associated with menstrual irregularity and post-menopausal bleeding due to estrogen secretion
- Testosterone and DHEAS: Elevated with androgen-secreting tumors (Sertoli-Leydig cell tumor, Leydig cell tumor)
Additional Investigations
- Pregnancy test: mandatory to exclude ectopic pregnancy in any reproductive-age woman with pelvic pain and adnexal mass
- Full blood count: leukocytosis with torsion, anemia with hemorrhagic rupture
- CT abdomen/pelvis with contrast: staging when malignancy suspected (peritoneal disease, lymphadenopathy, hepatic metastases)
- Genetic counseling and BRCA1/2 testing: indicated for women with strong personal/family history of ovarian or breast cancer
7. Treatment
Expectant Management — Functional Cysts
Simple unilocular cysts under 5 cm in pre-menopausal women (O-RADS 2) require no intervention — approximately 70% resolve spontaneously within 2–3 cycles. Reassurance and repeat TVUS in 6–12 weeks is appropriate. The role of oral contraceptives to promote cyst regression has not been supported in randomized trials and is not routinely recommended for established cysts; however, OCP use does reduce new functional cyst formation.
RCOG (Royal College of Obstetricians and Gynaecologists) guidelines recommend observation without surgery for simple unilocular ovarian cysts under 5 cm in both pre- and post-menopausal women, with annual ultrasound follow-up. ACOG (American College of Obstetricians and Gynecologists) and SRU (Society of Radiologists in Ultrasound) provide size-stratified follow-up intervals based on O-RADS category and menopausal status.
Management by Cyst Type
Endometrioma
Expectant management is appropriate for small endometriomas (under 3–4 cm) in asymptomatic women not planning IVF. Surgical management (laparoscopic cystectomy — stripping technique) is recommended for: symptomatic endometriomas causing intractable pain or dyspareunia, endometriomas above 4 cm in women planning IVF (to reduce contamination risk during oocyte retrieval and improve follicular access), and when malignancy cannot be excluded. The stripping technique (Bruhat method) — developing the cleavage plane between the cyst wall and ovarian cortex — is superior to fenestration/coagulation in reducing recurrence rates. However, follicle-containing ovarian cortex is inevitably removed, permanently reducing ovarian reserve (AMH decrease of 30–40% per operated ovary). Medical therapy (continuous OCP, progestins, GnRH agonists with add-back, dienogest) is used for symptom control and recurrence prevention but does not eliminate endometriomas.
Dermoid Cyst (Mature Cystic Teratoma)
Surgical removal is indicated for dermoids above 5–6 cm due to torsion risk, symptomatic lesions, and diagnostic uncertainty. Laparoscopic cystectomy with spillage prevention is the preferred approach — copious irrigation is mandatory if spillage occurs to prevent chemical peritonitis from sebaceous material. Oophorectomy is indicated for very large dermoids occupying the entire ovary, suspected malignant transformation (solid component, rapid growth in post-menopausal women), or surgical inaccessibility of the normal ovarian parenchyma. Bilateral involvement occurs in 10–15% of cases; careful inspection of the contralateral ovary is mandatory.
Cystadenoma
Serous and mucinous cystadenomas require laparoscopic or open cystectomy or oophorectomy depending on lesion size, patient age, and desire for fertility preservation. Spillage of mucinous contents should be minimized to reduce pseudomyxoma peritonei risk (though primary ovarian mucinous carcinoma with pseudomyxoma is far less common than appendiceal primary). Frozen section analysis is recommended intraoperatively for large or complex lesions to guide extent of surgery. Borderline tumors (low malignant potential) typically require only salpingo-oophorectomy with comprehensive surgical staging in post-menopausal women; fertility-preserving cystectomy may be considered in young women with stage I borderline tumors after thorough counseling.
Ovarian Torsion — Surgical Emergency
Torsion requires urgent surgical intervention, ideally within 6–8 hours of symptom onset to maximize ovarian salvage. Laparoscopic detorsion (untwisting the pedicle) is the preferred approach regardless of ovarian appearance — even a necrotic-appearing ovary should be detorsed before oophorectomy decision, as studies demonstrate that up to 93% of clinically black/necrotic ovaries recover function after detorsion. The ovary should be reassessed laparoscopically 24–48 hours post-detorsion via a second-look procedure if viability remains questionable. Concurrent cystectomy at the time of detorsion is controversial — cystectomy may be performed if the cyst is clearly benign (dermoid, simple cyst), but delayed elective cystectomy 6–8 weeks post-detorsion is preferred to minimize the risk of reoperation in an acutely inflamed pelvis. Oophoropexy (shortening the utero-ovarian ligament or fixing the ovary to the posterior leaf of the broad ligament) is recommended to prevent recurrent torsion, particularly in young women after detorsion and in the contralateral ovary in cases of congenitally long utero-ovarian ligament.
Hemorrhagic Cyst Rupture
Management depends on hemodynamic stability. Hemodynamically stable patients with small-to-moderate hemoperitoneum (identified on ultrasound or CT) and resolving pain are managed conservatively with close observation, analgesia, CBC monitoring, and serial abdominal examination. Hemodynamically unstable patients or those with refractory pain and significant hemoperitoneum require laparoscopic surgical exploration with hemostasis (electrocautery, suture, topical hemostatic agents) and evacuation of hemoperitoneum. Blood transfusion as required. Women on anticoagulation are at highest risk and require low threshold for surgical intervention.
Suspected Ovarian Malignancy — Specialist Referral
Women with O-RADS 4–5 lesions, ADNEX malignancy probability above 10–15%, or elevated CA-125 and post-menopausal status should be referred to a gynecologic oncologist. Comprehensive surgical staging (peritoneal washings, total abdominal hysterectomy, bilateral salpingo-oophorectomy, omentectomy, pelvic and para-aortic lymph node sampling, biopsy of any suspicious lesions) is the standard for suspected invasive ovarian carcinoma. Fertility-preserving staging (unilateral salpingo-oophorectomy) may be considered for apparent stage IA disease in young women after thorough staging and counseling.
8. Complications
- Ovarian torsion: Vascular compromise leading to ovarian infarction, necrosis, and permanent loss of ovarian function if not promptly treated; bilateral torsion, though rare, causes premature ovarian insufficiency
- Cyst rupture with hemoperitoneum: Potentially life-threatening blood loss from hemorrhagic corpus luteum rupture, particularly in anticoagulated patients
- Chemical peritonitis: Dermoid cyst spillage releasing sebaceous material causes severe peritoneal inflammation, adhesion formation, and bowel obstruction
- Malignant transformation: Endometriomas (endometrioid/clear cell carcinoma, RR 2.5–4), dermoids (squamous cell carcinoma, less than 2%), serous borderline tumors progressing to low-grade serous carcinoma
- Ovarian reserve depletion: Endometrioma cystectomy permanently reduces ovarian reserve; repeat surgeries compound follicle loss and accelerate ovarian aging
- Pseudomyxoma peritonei: Rare but devastating complication of mucinous tumor rupture with peritoneal dissemination
- Infertility: Endometriomas impair IVF outcomes through follicle destruction, toxic effects of hemosiderin on adjacent oocytes, and reduced response to ovarian stimulation
- Surgical complications: Oophorectomy-induced premature menopause in pre-menopausal women (cardiovascular, skeletal, and cognitive consequences); adhesion formation; bowel and ureteral injury
- Featured Videos
9. Prognosis
The prognosis of ovarian cysts is overwhelmingly favorable for functional and benign neoplastic lesions. Simple functional cysts resolve in 70–80% of pre-menopausal women within 2–3 cycles without intervention. Endometriomas recur in approximately 30–50% within 5 years of cystectomy; recurrence rates are reduced by post-operative hormonal suppression (dienogest, OCP). Dermoid cysts do not recur after complete excision; bilateral tumors are present in 10–15% and require vigilant follow-up of the contralateral ovary.
Ovarian torsion with prompt detorsion within 6–8 hours preserves ovarian function in approximately 90–95% of cases. When ovarian ischemia is prolonged (greater than 36 hours), oophorectomy is frequently required, with permanent loss of ovarian function on the affected side.
Ovarian cancer prognosis is stage-dependent: 5-year survival for stage I disease is approximately 90%; stage II 70%; stage III 25–45%; stage IV 5–20%. The poor overall prognosis for ovarian cancer (combined 5-year survival approximately 47%) reflects the predominance of advanced-stage diagnoses due to late symptom onset and lack of effective screening.
10. Prevention
- Oral contraceptives: Reduce functional ovarian cyst formation and provide approximately 40–50% risk reduction for epithelial ovarian cancer per 5 years of use; protection persists for 10–30 years after cessation
- Risk-reducing bilateral salpingo-oophorectomy (RRBSO): Recommended at age 35–40 (BRCA1) or 40–45 (BRCA2) after completion of childbearing in high-risk mutation carriers; reduces ovarian cancer risk by approximately 80% and breast cancer risk by 50% when performed before natural menopause
- Bilateral salpingectomy at time of hysterectomy or sterilization: Emerging evidence that tubal ligation and salpingectomy substantially reduce subsequent ovarian cancer risk, supporting the fimbrial/tubal origin theory for high-grade serous carcinoma
- Endometriosis management: Continuous progestogen therapy (dienogest, LNG-IUS) suppresses endometrioma recurrence and slows disease progression
- Genetic counseling: BRCA1/2 and Lynch syndrome testing and cascade testing of first-degree relatives enables targeted surveillance and risk reduction
- Parity and breastfeeding: Each pregnancy reduces ovarian cancer risk by approximately 13–19%; breastfeeding provides additional protection through anovulation
11. Recent Research
- IOTA ADNEX external validation: Multiple international prospective validation studies (UKCTOCS, UKOSSS) confirm ADNEX model superiority over CA-125 alone and Risk of Malignancy Index (RMI) in pre-operative risk stratification, with AUC 0.93 for distinguishing benign from malignant lesions when CA-125 is included.
- O-RADS MRI multicenter prospective validation: The ACR-sponsored O-RADS MRI prospective study across 13 international sites (published 2022–2023) demonstrated AUC 0.96 for malignancy, with O-RADS MRI 2 carrying less than 2% malignancy risk, supporting safe avoidance of surgery for these lesions.
- Endometrioma and ovarian reserve — laparoscopic vs. ablation: RCT and meta-analysis data continue to confirm that ablative techniques (CO2 laser, bipolar coagulation) cause less AMH reduction than cystectomy at 3 months post-operatively, though recurrence rates are higher; combined approaches (partial ablation of dense adherent wall) may optimize the balance.
- Dietary factors and endometrioma: Omega-3 fatty acid supplementation and Mediterranean diet adherence have been associated in prospective cohort studies with reduced endometriosis risk and severity, potentially via anti-inflammatory prostaglandin modulation.
- ctDNA and liquid biopsy: Early-phase studies identify circulating tumor DNA methylation patterns that distinguish high-grade serous ovarian carcinoma from benign adnexal masses with high specificity, offering a potential non-invasive complement to imaging in the pre-operative workup.
- Torsion detorsion and ovarian blood flow: Doppler studies confirm that absence of ovarian blood flow on preoperative ultrasound does not reliably predict non-viability; detorsion should be attempted in all cases regardless of Doppler findings, with subsequent assessment of perfusion restoration.
- Dienogest for endometrioma recurrence prevention: Phase III data from VISIBLE trials confirm that dienogest 2 mg/day after laparoscopic cystectomy significantly reduces 24-month recurrence rates (approximately 15% vs. 37% placebo) and improves dysmenorrhea scores without impairing subsequent fertility when discontinued prior to attempting conception.
12. References
- Timmerman D, Ameye L, Fischerova D, et al. Simple ultrasound rules to distinguish between benign and malignant adnexal masses before surgery: prospective validation by IOTA group. BMJ. 2010;341:c6839.
- Van Calster B, Valentin L, Savelli L, et al. Triaging women with ovarian masses for surgery: observational diagnostic study to compare IOTA ADNEX model, RMI and CA125. BJOG. 2017;124(1):132–141.
- Andreotti RF, Timmerman D, Strachowski LM, et al. O-RADS US Risk Stratification and Management System: A Consensus Guideline from the ACR Ovarian-Adnexal Reporting and Data System Committee. Radiology. 2020;294(1):168–185.
- Thomassin-Naggara I, Poncelet E, Jalaguier-Coudray A, et al. Ovarian-Adnexal Reporting Data System Magnetic Resonance Imaging (O-RADS MRI) Score for Risk Stratification of Sonographically Indeterminate Adnexal Masses. JAMA Netw Open. 2020;3(1):e1919896.
- Guerriero S, Condous G, van den Bosch T, et al. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: a consensus opinion from the International Deep Endometriosis Analysis (IDEA) group. Ultrasound Obstet Gynecol. 2016;48(3):318–332.
- RCOG. Management of Suspected Ovarian Masses in Premenopausal Women. Green-top Guideline No. 62. Royal College of Obstetricians and Gynaecologists; 2011 (revised 2016). Available at: rcog.org.uk
- Practice Bulletin No. 174: Evaluation and Management of Adnexal Masses. Obstet Gynecol. 2016;128(5):e210–e226.
- Somigliana E, Berlanda N, Benaglia L, et al. Surgical excision of endometriomas versus ovarian cystectomy with reconsideration of the ovarian reserve. Fertil Steril. 2012;98(6):1531–1537.
- Muzii L, Di Tucci C, Di Feliciantonio M, et al. The effect of surgery for endometrioma on ovarian reserve evaluated by antral follicle count: a systematic review and meta-analysis. Hum Reprod. 2014;29(10):2190–2198.
- Pereira N, Petrini AC, Lekovich JP, Elias RT, Spandorfer SD. Surgical management of endometrioma prior to in vitro fertilization: a systematic review. J Endometr Pelvic Pain Disord. 2016;8(2):53–60.
- McGrath S, Reid F. Ovarian torsion: evidence base for current and future surgical management. Best Pract Res Clin Obstet Gynaecol. 2021;74:25–36.
- Oelsner G, Shashar D. Adnexal torsion. Clin Obstet Gynecol. 2006;49(3):459–463.
- Nezhat F, Datta MS, Hanson V, Pejovic T, Nezhat C, Nezhat C. The relationship of endometriosis and ovarian malignancy: a review. Fertil Steril. 2008;90(5):1559–1570.
- Colombo N, Sessa C, du Bois A, et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Ann Oncol. 2019;30(5):672–705.
- Rebbeck TR, Kauff ND, Domchek SM. Meta-analysis of risk reduction estimates associated with risk-reducing salpingo-oophorectomy in BRCA1 or BRCA2 mutation carriers. J Natl Cancer Inst. 2009;101(2):80–87.
- Kho RM, Magrina JF, Magtibay PM. Pathologic findings and outcomes of a minimally invasive approach to ovarian dermoid cyst. Fertil Steril. 2007;87(5):1210–1213.
- Stukan M. Drainage of malignant ascites: patient selection and perspectives. Cancer Manag Res. 2017;9:115–130.
- Worley MJ, Welch WR, Berkowitz RS, Ng SW. Endometriosis-associated ovarian cancer: a review of pathogenesis. Int J Mol Sci. 2013;14(3):5367–5379.
Research Papers
The following PubMed topic searches aggregate the current peer-reviewed literature. Each link opens a live PubMed query — results update as new studies are indexed.
- PubMed — ovarian cyst
- PubMed — functional ovarian cyst
- PubMed — ovarian endometrioma
- PubMed — dermoid cyst ovary
- PubMed — ovarian cystadenoma
- PubMed — transvaginal ultrasound ovarian cyst
- PubMed — ovarian torsion
- PubMed — ruptured ovarian cyst
- PubMed — laparoscopic cystectomy
- PubMed — CA-125 ovarian mass
- PubMed — IOTA simple rules ovarian
- PubMed — polycystic ovary syndrome
Connections
- Endometriosis
- Infertility
- Polycystic Ovary Syndrome
- Perimenopause
- Uterine Fibroids
- Menopause and HRT
- Laparoscopy: Excision vs Ablation
- Vitamin D3
- Magnesium
- Diagnosis Delay and Imaging
- Hormonal Therapy Options
- Fertility and Endometriosis
- Adenomyosis Overlap
- Full Body MRI
- Testosterone
- Anemia
- Gestational Diabetes
- Preeclampsia