Clostridioides difficile Infection
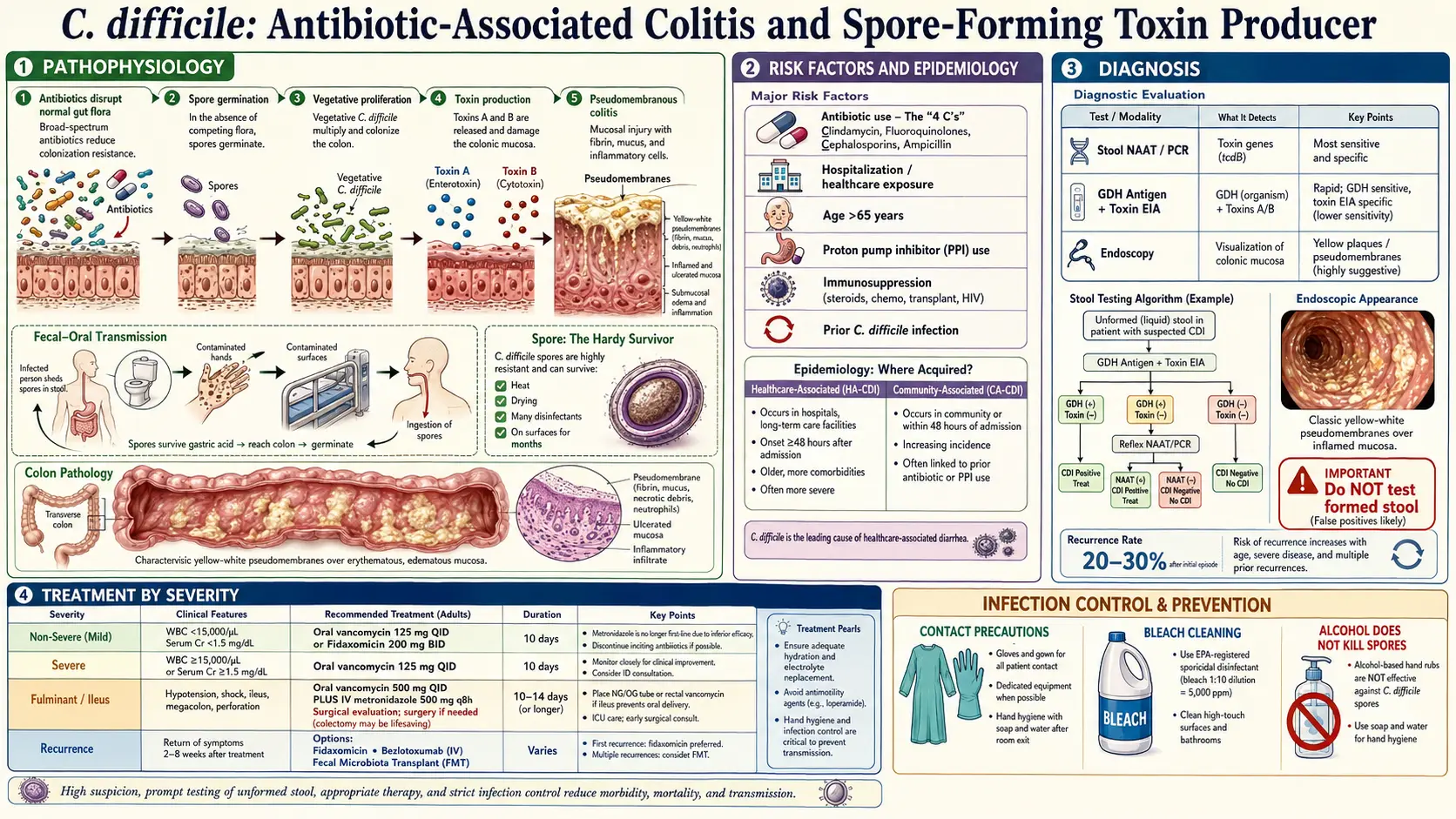
Clostridioides difficile infection (CDI) — formerly classified as Clostridium difficile — is the leading cause of healthcare-associated infectious diarrhea in developed nations and an increasingly prevalent community-acquired pathogen. It represents a major global public health challenge, with approximately 500,000 cases annually in the United States alone, and is associated with substantial morbidity, mortality, and healthcare expenditure.
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research
- References
- Featured Videos
1. Overview
Clostridioides difficile is an obligate anaerobic, spore-forming, gram-positive bacillus that colonizes the human colon and produces potent exotoxins responsible for the clinical syndrome of CDI. The organism was first isolated in 1935 by Hall and O'Toole, who noted it as a commensal in neonatal gut flora and designated it "difficile" for the difficulty encountered in its cultivation. Its pathogenic significance was not fully appreciated until 1978, when it was identified as the causative agent of antibiotic-associated pseudomembranous colitis.
CDI occurs when disruption of the normal gut microbiota — most commonly by antibiotic use — allows colonization by toxigenic C. difficile strains, with subsequent toxin production causing mucosal damage and the characteristic clinical syndrome. The spectrum of disease ranges from mild self-limited diarrhea to fulminant colitis with systemic inflammatory response, toxic megacolon, and death.
Nomenclature: The organism was reclassified from Clostridium difficile to Clostridioides difficile in 2016 based on phylogenetic analysis, though the abbreviation CDI and the colloquial term "C. diff" remain in widespread clinical use.
2. Epidemiology
CDI is the most common healthcare-associated infection in US hospitals, surpassing methicillin-resistant Staphylococcus aureus (MRSA) in incidence. The Centers for Disease Control and Prevention (CDC) estimates approximately 500,000 infections and 29,000 attributable deaths annually in the United States. Direct healthcare costs exceed $6 billion per year.
The epidemiology of CDI has undergone a significant transformation since the early 2000s, driven by the emergence of the hypervirulent NAP1/BI/ribotype 027 strain (C. difficile ribotype 027, CDT-027). This strain is characterized by:
- Production of binary toxin (CDT) in addition to toxins A and B
- Deletion in the tcdC gene (negative regulator of toxin production), resulting in markedly elevated toxin output
- A variant form of toxin B with altered receptor binding
- High-level fluoroquinolone resistance (mutations in gyrA and gyrB)
- Increased sporulation efficiency, facilitating environmental persistence and transmission
- Higher rates of severe disease, complications, and mortality compared to non-027 strains
The NAP1/ribotype 027 strain has spread internationally and caused major nosocomial outbreaks in North America and Europe. However, its dominance has waned in some regions, with ribotype 078 and 244 emerging as significant strains, particularly in community-acquired CDI.
Community-acquired CDI (CA-CDI) — defined as CDI occurring in outpatients or within 48 hours of admission with no hospitalization in the preceding 12 weeks — now accounts for approximately 30–40% of all CDI cases. CA-CDI patients tend to be younger, have fewer comorbidities, and demonstrate lower rates of recurrence, though the overall incidence is rising.
CDI predominantly affects older adults; persons aged ≥65 years account for more than 80% of CDI-related deaths. Institutional settings (hospitals, long-term care facilities) remain the highest-risk environments due to high antibiotic use, immunocompromised populations, and environmental contamination with spores.
3. Pathophysiology
Organism Biology and Spore Formation
The pathogenicity of C. difficile is critically dependent on its ability to form highly resistant endospores. Spores are metabolically dormant, resistant to standard disinfectants (including alcohol-based hand gels), heat, and desiccation, and can survive on environmental surfaces for months to years. Spores are ingested via the fecal-oral route, transit the acidic stomach environment unharmed, and germinate in the small intestine under the influence of bile acids — specifically, primary bile acids (taurocholate) trigger germination while secondary bile acids (deoxycholate, lithocholate produced by normal gut flora) inhibit it. This bile acid ecology partly explains why antibiotic-induced reduction of secondary bile acid-producing flora facilitates CDI.
Microbiome Disruption and Colonization Resistance
The healthy adult gut microbiota provides robust colonization resistance against C. difficile through multiple mechanisms: competition for nutrients and mucosal binding sites, production of short-chain fatty acids (SCFAs) and secondary bile acids that inhibit C. difficile growth, stimulation of mucosal IgA secretion, and direct bacteriocin production. Antibiotic disruption — particularly broad-spectrum agents — depletes this protective consortium, allowing C. difficile spores (acquired from the environment or endogenous reservoir) to germinate and proliferate to pathogenic densities.
Toxin A and Toxin B
The cardinal virulence determinants of pathogenic C. difficile strains are two large clostridial toxins encoded on the pathogenicity locus (PaLoc): a 19.6-kb chromosomal element containing tcdA (toxin A), tcdB (toxin B), and three regulatory genes (tcdC, tcdE, tcdR).
Toxin A (TcdA) is a 308-kDa glucosyltransferase enterotoxin. It binds to cell surface receptors (including the carbohydrate moiety on Gal-alpha-1-3-Gal-beta-1-4-GlcNAc), is internalized by endocytosis, and translocates its glucosyltransferase domain into the cytoplasm, where it mono-O-glucosylates and thereby inactivates Rho-family GTPases (RhoA, Rac1, Cdc42). Rho GTPase inactivation disrupts the actin cytoskeleton, breaks tight junctions, increases intestinal permeability, and triggers apoptosis. Toxin A also activates macrophages and mast cells to release proinflammatory cytokines (TNF-alpha, IL-1, IL-6, IL-8) and stimulates substance P release from enteric neurons, amplifying the inflammatory response and causing fluid hypersecretion.
Toxin B (TcdB) is a 270-kDa cytotoxin, approximately 1,000-fold more potent than Toxin A on a molar basis in cell culture assays. It employs a similar glucosyltransferase mechanism targeting Rho GTPases but uses different cell surface receptors: PVRL3 (nectin-3), FZD1, FZD2, and FZD7 (Frizzled receptors). TcdB is now regarded as the primary virulence factor, as strains producing only TcdB (TcdA-/TcdB+) retain full virulence in humans while TcdA+/TcdB- strains cause minimal disease.
The net effect of toxin A and B activity on colonocytes includes: loss of epithelial barrier integrity, inflammatory infiltration (predominantly neutrophilic) of the lamina propria, submucosal edema, vascular congestion, mucosal ulceration, and the formation of pseudomembranes — raised yellow-white plaques composed of fibrin, mucin, neutrophils, and cellular debris that are pathognomonic of pseudomembranous colitis.
Binary Toxin (CDT)
Approximately 6–10% of clinical C. difficile strains (including ribotype 027 and 078) produce a third toxin, binary toxin CDT, encoded outside the PaLoc on a separate chromosomal element (cdt locus). CDT is an ADP-ribosyltransferase toxin (related to C. perfringens iota toxin) consisting of two components: CDTa (enzymatic) and CDTb (binding/translocation). It ADP-ribosylates G-actin, disrupting actin polymerization and causing unique protrusions (microtubule-based tentacle-like extensions) on the cell surface that dramatically enhance C. difficile adherence. CDT is associated with increased virulence and mortality in epidemiological studies, though its precise contribution to disease relative to TcdA and TcdB is under ongoing investigation.
Host Immune Response
Serum IgG antibody titers against toxins A and B are strongly protective against CDI severity and recurrence. Infants commonly carry C. difficile as asymptotic colonizers in part due to absence of toxin receptors and high levels of maternally transferred IgG. Adults with high anti-toxin A IgG levels are significantly less likely to experience recurrent CDI after initial treatment, a finding that motivated the development of monoclonal antibody therapy (bezlotoxumab).
4. Etiology and Risk Factors
Antibiotic Exposure
Antibiotic use is the single most important modifiable risk factor for CDI. Any antibiotic can predispose to CDI, but the highest-risk agents include:
- Fluoroquinolones (ciprofloxacin, levofloxacin, moxifloxacin): Particularly implicated in the emergence and spread of hypervirulent ribotype 027. The fluoroquinolone resistance of NAP1 provides a selective survival advantage during fluoroquinolone therapy.
- Clindamycin: Historically the most strongly associated antibiotic; classic teaching mnemonic for CDI risk.
- Broad-spectrum cephalosporins (ceftriaxone, cefotaxime, cefepime): Major disruptors of anaerobic gut flora.
- Broad-spectrum penicillins with/without beta-lactamase inhibitors (amoxicillin-clavulanate, piperacillin-tazobactam): High-risk due to broad anaerobic coverage.
- Carbapenems: Among the most disruptive of anaerobic colonization resistance.
Risk is proportional to the number of antibiotic courses, duration of therapy, and spectrum of activity. CDI can occur during antibiotic therapy or up to 8–12 weeks after discontinuation. The 4-week period following antibiotic cessation carries the highest risk.
Patient-Related Risk Factors
- Advanced age (≥65 years): Impaired immune response (reduced IgG production against C. difficile toxins), altered gut flora diversity, high rates of antibiotic use and hospitalization.
- Hospitalization and healthcare exposure: Environmental contamination with spores, density of colonized/infected patients, shared equipment. Duration of hospitalization correlates directly with CDI risk.
- Proton pump inhibitors (PPIs): PPIs reduce gastric acid, potentially allowing more spores to survive transit to the colon. Multiple meta-analyses demonstrate a 1.5–3-fold increased CDI risk with PPI use, though causality remains debated given confounding by indication.
- Inflammatory bowel disease (IBD): Patients with IBD have a 3–5 fold increased CDI risk. CDI in the setting of IBD carries substantially higher rates of colectomy, complications, and mortality. CDI can mimic or trigger IBD flares, complicating diagnosis.
- Immunosuppression: Solid organ and hematopoietic stem cell transplant recipients, patients on systemic corticosteroids or immunomodulatory agents, and those receiving cancer chemotherapy have heightened susceptibility and often atypical presentations.
- Renal failure: Chronic kidney disease is an independent risk factor, possibly through altered bile acid metabolism and immune dysregulation.
- Prior CDI: The strongest predictor of recurrent CDI; risk increases with each subsequent episode (25% after first, 50% after second, >60% with three or more episodes).
- Nasogastric tube use, gastrointestinal surgery: Disrupt GI physiology and promote spore ingestion.
- Malnutrition and low serum albumin: Markers of impaired immunity and associated with more severe disease.
5. Clinical Presentation
Spectrum of Disease
Clinical manifestations of CDI span a broad spectrum from asymptomatic colonization to life-threatening fulminant colitis:
Mild to Moderate CDI
- Diarrhea: The hallmark symptom — typically watery, foul-smelling, 3 or more loose/unformed stools per 24-hour period. Stools may contain mucus but frank blood is uncommon in uncomplicated CDI (gross hematochezia should prompt evaluation for alternative diagnoses).
- Abdominal cramping: Lower abdominal cramping and bloating, often preceding or accompanying diarrhea.
- Low-grade fever: Present in approximately 30–40% of cases.
- Constitutional symptoms: Mild nausea, anorexia, and malaise are common.
- Leukocytosis: WBC of 10,000–20,000 cells/mm³ is typical; a disproportionate leukocytosis (>20,000–30,000 cells/mm³) without other explanation should raise concern for severe or complicated CDI.
Severe CDI
The IDSA/SHEA 2017 Clinical Practice Guidelines define severe CDI by either of the following criteria:
- Serum creatinine ≥1.5 mg/dL (new elevation or worsening from baseline)
- WBC ≥15,000 cells/mm³
Additional features suggesting severity include: serum albumin <3.0 g/dL, age >65 years, temperature >38.5°C, intensive care unit admission, and immunocompromised state. Severe CDI carries substantially higher rates of complications and 30-day mortality.
Fulminant CDI
Defined by the IDSA/SHEA guidelines as CDI with any of the following: hypotension or shock, ileus, or toxic megacolon. Formerly designated "severe complicated CDI," fulminant CDI represents the most life-threatening presentation:
- Ileus: Colonic dysmotility may paradoxically reduce or eliminate diarrhea despite severe underlying disease; absence of diarrhea does not exclude CDI in the appropriate clinical context.
- Toxic megacolon: Colonic dilatation (>6 cm on plain radiograph, or >9 cm for cecum) with systemic toxicity (fever, tachycardia, leukocytosis, hypovolemia). Risk of perforation and death is high without urgent intervention.
- Septic shock: Refractory hypotension despite adequate resuscitation, multi-organ dysfunction.
- Markedly elevated WBC: Often >30,000–50,000 cells/mm³; extreme leukemoid reaction (>50,000 cells/mm³) has been reported with ribotype 027 strains.
Recurrent CDI
Recurrent CDI (rCDI) is defined as recurrence of symptoms within 8 weeks of completing treatment for a prior episode with documented microbiological cure (negative test). It is essential to distinguish recurrence (re-infection with the same or different strain, or relapse from persistent spores) from treatment failure (persistent symptoms without symptomatic resolution). First recurrence occurs in approximately 20–25% of patients; subsequent recurrences become progressively more frequent, creating a vicious cycle of dysbiosis and re-infection.
6. Diagnosis
Accurate diagnosis of CDI requires both clinical assessment and laboratory confirmation. Testing should be performed only in patients with clinically significant diarrhea (≥3 unformed stools in 24 hours) who have no other identifiable cause. Testing asymptomatic patients or formed stool specimens is strongly discouraged as it leads to overdiagnosis and inappropriate treatment of colonized individuals.
Laboratory Testing
The optimal diagnostic strategy is an area of ongoing debate and institutional variation. The key assays are:
- Nucleic acid amplification test (NAAT) / PCR: Detects tcdB (and other PaLoc genes) with sensitivity >95% and specificity >97%. Highly sensitive but cannot distinguish active infection from asymptomatic colonization in patients with positive toxin-encoding genes but absent toxin production. The high sensitivity of PCR contributes to overdiagnosis and overtreatment when used as a standalone test.
- Enzyme immunoassay (EIA) for glutamate dehydrogenase (GDH): GDH is a metabolic enzyme produced by all C. difficile strains (toxigenic and non-toxigenic). High sensitivity (~90%) but low specificity; used as a screening test in multistep algorithms. A negative GDH reliably excludes CDI.
- Enzyme immunoassay for toxins A and B (EIA): Direct detection of free toxin in stool, with specificity ~95–98% but limited sensitivity (~75–85%). Positive toxin EIA in the appropriate clinical context has high positive predictive value. Importantly, a positive toxin EIA correlates better with clinically significant disease than PCR alone.
- Cell cytotoxicity neutralization assay (CCNA): The historical gold standard — detects the cytopathic effect of toxin B on cell monolayers, neutralized by antitoxin serum. Sensitivity ~90%, specificity ~98–99%, but requires 24–48 hours and specialized laboratory infrastructure. Largely replaced by NAAT in clinical practice.
- Toxigenic culture: Anaerobic culture of stool followed by toxin testing of isolated colonies. The absolute gold standard for sensitivity but requires 5–7 days; primarily a research and epidemiological tool.
The IDSA/SHEA 2017 guidelines and Infectious Diseases Society of America endorse either of two preferred diagnostic approaches:
- Multistep algorithm: GDH EIA + toxin EIA as initial screen; discordant results reflexed to NAAT. This approach maximizes specificity and avoids overdiagnosis.
- NAAT alone (or NAAT + toxin EIA): Acceptable if only patients meeting strict clinical criteria for testing are tested. NAAT alone as a population-wide screening test is discouraged.
Endoscopy
Colonoscopy or flexible sigmoidoscopy is not routinely required for CDI diagnosis but may be necessary in select circumstances:
- Clinical suspicion is high but stool tests are repeatedly negative (ileus preventing diarrhea)
- Urgent diagnosis required when tests cannot be completed rapidly
- Evaluation for alternative diagnoses (IBD, ischemic colitis, CMV colitis)
Pseudomembranes — raised, confluent, yellow-white plaques 2–10 mm in diameter overlying hyperemic and edematous mucosa — are pathognomonic for pseudomembranous colitis on endoscopy. They are present in approximately 50% of CDI cases; their absence does not exclude CDI. Biopsies demonstrate the characteristic "summit" or "volcanic" lesion on histology.
Imaging
CT of the abdomen and pelvis with intravenous contrast is the preferred imaging modality for suspected complicated CDI. Characteristic findings include:
- Pancolonic or segmental colonic wall thickening (>4 mm)
- "Accordion sign" — alternating bands of oral contrast trapped between thickened folds on CT
- Pericolonic fat stranding
- Ascites (in severe cases)
- Colonic dilatation (>6 cm) suggesting toxic megacolon
- Free air indicating perforation
7. Treatment
General Principles
First, any inciting antibiotic(s) should be discontinued if clinically feasible — this alone results in resolution of diarrhea in approximately 15–23% of CDI patients. Fluid and electrolyte replacement is critical, particularly in elderly patients. Antiperistaltic agents (loperamide, opioids) are contraindicated in CDI as they can precipitate toxic megacolon. Patients should be placed on contact precautions.
Antibiotic Treatment — IDSA/SHEA 2017 Guidelines
The 2017 IDSA/SHEA Clinical Practice Guidelines (Cohen et al.) represent the current standard of care for CDI treatment in the United States, with updates anticipated based on emerging fecal microbiota transplant (FMT) and microbiome-based therapies:
- Non-severe CDI (initial episode): Fidaxomicin 200 mg orally twice daily for 10 days is preferred over vancomycin (IDSA 2017 strong recommendation, high quality evidence) due to significantly lower recurrence rates (approximately 15% vs. 25–27% with vancomycin). Vancomycin 125 mg orally four times daily for 10 days remains an acceptable alternative (strong recommendation, high quality evidence). Metronidazole 500 mg orally three times daily for 10 days is now considered an alternative only when fidaxomicin and vancomycin are unavailable (weak recommendation), reflecting inferior efficacy in recent trials.
- Severe CDI (initial episode): Vancomycin 125 mg orally four times daily for 10 days (strong recommendation, high quality evidence). Fidaxomicin 200 mg orally twice daily for 10 days is an equally acceptable alternative (strong recommendation, moderate quality evidence). Metronidazole is not recommended for severe CDI.
- Fulminant CDI: Vancomycin 500 mg orally or via nasogastric tube four times daily, plus metronidazole 500 mg intravenously every 8 hours (strong recommendation, moderate quality evidence). In the setting of ileus or colonic dilatation, vancomycin enemas (500 mg in 100 mL saline every 6 hours via retention enema) should be added to ensure adequate colonic drug levels. Surgical consultation should be obtained early for patients with fulminant CDI.
Fidaxomicin
Fidaxomicin is a macrocyclic antibiotic with a narrow spectrum of activity highly specific for C. difficile. Its key pharmacological advantages over vancomycin include: minimal systemic absorption (99.5% of the dose remains in the colonic lumen), negligible impact on the intestinal microbiota beyond C. difficile, and bactericidal activity that persists after the dosing interval (post-antibiotic effect). Two pivotal phase III randomized controlled trials (OPCDT-3 and OPCDT-4) demonstrated clinical cure rates equivalent to vancomycin but significantly lower recurrence rates, particularly for non-NAP1 strains. For NAP1/ribotype 027 strains, fidaxomicin's recurrence advantage is attenuated but not eliminated. The primary limitation of fidaxomicin is cost.
Recurrent CDI
- First recurrence: If metronidazole was used for the initial episode, treat with vancomycin standard course. If vancomycin standard course was used, treat with fidaxomicin (preferred) or a vancomycin tapered/pulsed regimen (e.g., 125 mg four times daily for 10–14 days, then twice daily for 7 days, then once daily for 7 days, then every 2–3 days for 2–8 weeks) (IDSA 2017 weak recommendation, low quality evidence).
- Second or subsequent recurrences: Vancomycin tapered/pulsed regimen OR vancomycin followed by rifaximin 400 mg three times daily for 20 days (weak recommendation). Fecal microbiota transplantation (FMT) should be strongly considered for patients with three or more recurrences (strong recommendation, moderate quality evidence) given cure rates exceeding 85–90%.
Bezlotoxumab
Bezlotoxumab (Zinplava, Merck) is a fully human IgG1 monoclonal antibody targeting C. difficile toxin B, approved by the FDA in 2016 as an adjunct to standard antibiotic therapy to reduce CDI recurrence in high-risk adult patients. In the pivotal MODIFY I and MODIFY II trials, bezlotoxumab (10 mg/kg IV single dose) reduced global CDI recurrence rates from approximately 26–28% (placebo + antibiotic) to 17% (bezlotoxumab + antibiotic) — a relative risk reduction of approximately 40%. Benefit was concentrated in high-risk subgroups: age ≥65 years, immunocompromised status, severe CDI, and CDI due to ribotype 027 strains.
Bezlotoxumab acts by direct high-affinity neutralization of toxin B, preventing toxin B-mediated mucosal damage and dampening the inflammatory cascade that perpetuates dysbiosis. Actoxumab (anti-toxin A monoclonal) did not add benefit when combined with bezlotoxumab. A caution note: bezlotoxumab was associated with a higher rate of heart failure exacerbations in patients with underlying cardiac disease in the MODIFY trials (heart failure AE: 12.7% bezlotoxumab vs. 4.8% placebo); its use should be carefully considered in patients with a history of congestive heart failure.
Fecal Microbiota Transplantation (FMT)
FMT involves transfer of processed stool from a healthy screened donor into the gastrointestinal tract of the recipient, with the goal of restoring a diverse and protective microbiome. It is the most effective treatment for recurrent CDI, with clinical cure rates of 85–92% in randomized controlled trials and up to 90–95% in large case series, far exceeding antibiotic therapy alone. FMT is recommended by IDSA/SHEA for patients with ≥2 recurrences of CDI (three or more CDI episodes total).
Delivery routes include: colonoscopy (most widely used in the US, allows direct visualization of mucosa), nasojejunal tube or enema (for patients unable to tolerate colonoscopy), and upper GI routes (nasoduodenal or esophagogastroduodenoscopy via pylorus). Capsulized frozen FMT preparations have demonstrated non-inferiority to colonoscopic delivery in randomized trials, substantially simplifying administration.
Donor screening protocols are rigorous (stool culture, ova and parasites, C. difficile testing, and serological testing for bloodborne pathogens) to minimize transmission risk. In 2019, two immunocompromised FMT recipients at Massachusetts General Hospital died following transmission of extended-spectrum beta-lactamase (ESBL)-producing E. coli from an inadequately screened donor, prompting FDA to strengthen donor screening requirements, including expanded multi-drug-resistant organism testing and updated exclusion criteria for recent gastrointestinal symptoms.
The FDA approved the first standardized FMT product, REBYOTA (fecal microbiota, live-jslm; RBX2660, Ferring Pharmaceuticals) in November 2022 for prevention of CDI recurrence following antibiotic treatment in adults ≥18 years, followed by VOWST (fecal microbiota spores, live-brpk; SER-109, Seres Therapeutics) — the first oral FMT capsule product — approved in April 2023. These regulatory approvals mark a pivotal transition from investigational to standardized microbiome-based therapeutics.
Surgical Management
Surgery is reserved for fulminant CDI refractory to medical management, or with complications (perforation, toxic megacolon failing medical therapy). Subtotal colectomy with end ileostomy has traditionally been the procedure of choice and carries 30-day mortality of 35–80% when performed for fulminant CDI in extremis. Loop ileostomy with colonic lavage (Neal procedure) is a less morbid alternative that preserves the colon: an ileostomy is created, the colon is lavaged with polyethylene glycol via the ileostomy, and anterograde vancomycin flushes are administered through the ileostomy postoperatively. A randomized trial demonstrated significantly lower mortality with loop ileostomy versus colectomy (19% vs. 50%) in carefully selected patients, though patient selection criteria for this approach remain an area of ongoing refinement.
Early surgical consultation — ideally when serum lactate >5 mmol/L, WBC >50,000/mm³, or clinical deterioration is evident — is associated with improved outcomes. Delay in surgical referral until hemodynamic collapse significantly worsens prognosis.
8. Complications
Toxic Megacolon
Transmural colonic inflammation leads to functional obstruction, massive dilatation (>6 cm), and systemic toxicity. Plain abdominal radiograph demonstrates markedly dilated, often featureless colon. Management includes bowel rest, nasogastric decompression, urgent colonoscopic decompression (with extreme caution given perforation risk), maximal medical CDI therapy, and urgent surgical consultation. Perforation risk with toxic megacolon is approximately 20–30%, carrying mortality up to 60%.
Colonic Perforation
Transmural necrosis or toxic megacolon can lead to free perforation with fecal peritonitis, septic shock, and death. Requires emergency laparotomy. In-hospital mortality exceeds 50% in most case series.
Dehydration, Electrolyte Imbalance, and Acute Kidney Injury
Profuse diarrhea leads to hypovolemia, hyponatremia, hypokalemia, and prerenal or intrinsic acute kidney injury (AKI). AKI is both a criterion for severe CDI and an independent predictor of mortality. Aggressive IV fluid resuscitation and electrolyte repletion are essential, while avoiding nephrotoxic agents.
Reactive Arthritis
A rare post-infectious complication (<1% of CDI cases) characterized by asymmetric oligoarthritis involving large joints, typically occurring 2–6 weeks after CDI. It is thought to be immune-mediated rather than a direct joint infection. Usually self-limited but may require NSAIDs or short-course corticosteroids.
Recurrent CDI
Recurrence is the most clinically significant complication of CDI. Each recurrent episode further disrupts the gut microbiome, perpetuating a cycle of dysbiosis and susceptibility. Recurrent CDI is associated with progressive decline in quality of life, healthcare utilization, functional status, and mortality, particularly in elderly patients.
CDI in Special Populations
IBD patients with CDI have threefold higher rates of colectomy and fivefold higher in-hospital mortality compared to CDI patients without IBD. Distinguishing CDI from IBD flare is imperative, and concurrent CDI should be excluded in all IBD patients presenting with a disease flare.
Immunocompromised patients (transplant recipients, HIV/AIDS, chemotherapy) may present with atypical features, prolonged course, and reduced inflammatory markers despite severe disease. A high index of suspicion is warranted.
Pregnancy: CDI during pregnancy is rare but associated with adverse maternal and fetal outcomes. Oral vancomycin (minimally absorbed) is preferred; metronidazole is avoided in the first trimester due to teratogenicity concerns, though data are limited.
9. Prognosis
Prognosis in CDI is highly variable and depends on disease severity, patient characteristics, and adequacy of treatment:
- Mortality: Overall 30-day all-cause mortality attributable to CDI is approximately 5–10% in hospitalized patients. In fulminant CDI requiring surgery, 30-day mortality ranges from 35–80%. Mortality in elderly ICU patients with severe CDI may exceed 30%.
- Recurrence: First recurrence rate is 20–25%, rising to 40–65% with two or more prior episodes. Recurrence is not predicted by antibiotic choice in most studies but by host factors: advanced age, ongoing antibiotic use, immunosuppression, and prior CDI episodes.
- Long-term sequelae: Survivors of severe CDI may have persistent gut dysbiosis, increased susceptibility to other infections, reduced quality of life, irritable bowel syndrome-like symptoms, and functional decline.
Validated severity scoring tools include the ATLAS score (age, treatment with systemic antibiotics, leukocyte count, albumin, and serum creatinine) and the IDSA/SHEA severity classification. The PSEP (Predictors of Severe and Severe Complicated CDI) model incorporates admission WBC, serum creatinine, albumin, age, and ICU status. These tools aid in stratifying patients who require closer monitoring and escalated therapy.
10. Prevention
Antibiotic Stewardship
Antibiotic stewardship programs (ASPs) are the most effective institutional intervention for reducing CDI incidence. Core ASP strategies include: restriction of high-risk antibiotics (fluoroquinolones, clindamycin, broad-spectrum cephalosporins and carbapenems) without appropriate indications; de-escalation to narrower-spectrum agents when culture and sensitivity data are available; shortened antibiotic courses; and minimizing prophylactic antibiotic use. Robust evidence supports that ASP implementation reduces CDI rates by 20–50% in hospital settings.
Infection Control and Environmental Decontamination
C. difficile spores are resistant to standard alcohol-based hand sanitizers and quaternary ammonium-based disinfectants. Key infection control measures include:
- Contact precautions: Gown and gloves for all contacts with CDI patients; private room or cohorting of CDI patients.
- Hand hygiene with soap and water: Mechanical removal of spores by friction; alcohol hand gel alone is inadequate for CDI prevention. Handwashing with soap and water is mandatory after contact with CDI patients or their environment.
- Sodium hypochlorite (bleach) disinfection: Sporicidal agents such as 1:10 dilution bleach solution or commercially available hypochlorite-based products are required for environmental decontamination of rooms occupied by CDI patients. Standard quaternary ammonium compounds are not sporicidal and are inadequate.
- Enhanced terminal cleaning: Thorough terminal cleaning of rooms after CDI patient discharge, including high-touch surfaces (bed rails, call buttons, infusion pumps), using sporicidal agents.
- Duration of precautions: SHEA guidelines recommend maintaining contact precautions for at least 48 hours after diarrhea resolution; some institutions extend precautions for the duration of hospitalization given environmental spore persistence.
- UV-C light disinfection: Pulsed xenon UV-C technology is increasingly used as an adjunct to manual cleaning; shown to reduce environmental C. difficile contamination when implemented alongside standard precautions.
Prophylactic Measures
- Probiotics: Meta-analyses suggest Lactobacillus and Saccharomyces boulardii-containing preparations may reduce CDI incidence when administered concurrently with antibiotics, but evidence quality is moderate and recommendations are not universal. The PLACIDE trial (a large UK RCT) did not demonstrate benefit in elderly patients. Routine probiotic prophylaxis is not currently recommended by IDSA/SHEA.
- Vancomycin prophylaxis: Oral vancomycin 125 mg twice daily during and for 5 days after subsequent antibiotic courses is increasingly used in patients with multiple prior CDI episodes, based on observational data and small RCTs demonstrating reduced recurrence rates. This practice is gaining acceptance in expert guidelines though formal recommendation strength remains moderate.
- PPI deprescribing: Reviewing and discontinuing unnecessary PPIs in hospitalized patients reduces CDI risk and is a component of antimicrobial stewardship bundles.
11. Recent Research
- FDA-approved microbiome therapeutics: The approval of REBYOTA (RBX2660) in 2022 and VOWST (SER-109 oral capsules) in 2023 represents a landmark shift toward standardized microbiome-based therapies, replacing largely unregulated donor-specific FMT with quality-controlled pharmaceutical products. Ongoing studies are evaluating optimal indications, dosing, and long-term durability.
- Live biotherapeutic products (LBPs): Beyond traditional FMT, defined microbial consortia comprising specific bacterial species (e.g., VE303, a consortium of eight Clostridia species) are being investigated. Phase II data for VE303 demonstrated significant reduction in rCDI compared to placebo.
- Ribotype ecology and evolution: Genomic surveillance studies using whole-genome sequencing (WGS) have demonstrated that most CDI cases arise from distinct environmental acquisitions rather than patient-to-patient transmission in modern hospital settings, challenging traditional nosocomial transmission paradigms and suggesting community reservoirs (food supply, domestic animals) warrant greater attention.
- Bezlotoxumab real-world effectiveness: Post-approval registries and cohort studies have confirmed the real-world benefit of bezlotoxumab in reducing rCDI among high-risk patients, with greatest benefit in those with ≥1 prior CDI episode, age ≥65, and ribotype 027 infection.
- Extended-pulsed fidaxomicin (EXTEND regimen): The EXTEND trial demonstrated that fidaxomicin 200 mg twice daily for 5 days followed by once-every-other-day for 20 days significantly reduced CDI recurrence compared to standard fidaxomicin, suggesting post-treatment microbiome colonization resistance can be restored with prolonged low-dose exposure. This regimen is not yet incorporated into guidelines but is an option in clinical practice.
- Gut microbiome-based biomarkers: Research is identifying specific microbiome signatures (loss of Lachnospiraceae, Ruminococcaceae; expansion of Proteobacteria) that predict CDI recurrence, paving the way for microbiome-guided therapeutic decision-making.
12. References
- McDonald LC, Gerding DN, Johnson S, et al. Clinical Practice Guidelines for Clostridium difficile Infection in Adults and Children: 2017 Update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis. 2018;66(7):e1-e48.
- Lessa FC, Mu Y, Bamberg WM, et al. Burden of Clostridium difficile infection in the United States. N Engl J Med. 2015;372(9):825-834.
- Gerding DN, Johnson S, Rupnik M, Aktories K. Clostridium difficile binary toxin CDT: mechanism, epidemiology, and potential clinical importance. Gut Microbes. 2014;5(1):15-27.
- Janoir C. Virulence factors of Clostridium difficile and their role during infection. Anaerobe. 2016;37:13-24.
- Wilcox MH, Gerding DN, Poxton IR, et al. Bezlotoxumab for prevention of recurrent Clostridium difficile infection. N Engl J Med. 2017;376(4):305-317.
- Cornely OA, Crook DW, Esposito R, et al. Fidaxomicin versus vancomycin for infection with Clostridium difficile in Europe, Canada, and the USA: a double-blind, non-inferiority, randomised controlled trial. Lancet Infect Dis. 2012;12(4):281-289.
- van Nood E, Vrieze A, Nieuwdorp M, et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368(5):407-415.
- Feuerstadt P, Louie TJ, Lashner B, et al. SER-109, an oral microbiome therapy for recurrent Clostridioides difficile infection. N Engl J Med. 2022;386(3):220-229.
- Khanna S, Gerding DN. Current and future trends in Clostridioides (Clostridium) difficile infection management. Anaerobe. 2019;58:10-15.
- Neal MD, Alverdy JC, Hall DE, Simmons RL, Zuckerbraun BS. Diverting loop ileostomy and colonic lavage: an alternative to total abdominal colectomy for the treatment of severe, complicated Clostridium difficile associated disease. Ann Surg. 2011;254(3):423-429.
- Surawicz CM, Brandt LJ, Binion DG, et al. Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am J Gastroenterol. 2013;108(4):478-498.
- Rao K, Malani PN. Diagnosis and treatment of Clostridioides (Clostridium) difficile infection in adults in 2020. JAMA. 2020;323(14):1403-1404.
- Crobach MJT, Vernon JJ, Loo VG, et al. Understanding Clostridium difficile colonization. Clin Microbiol Rev. 2018;31(2):e00021-17.
- Warny M, Pepin J, Fang A, et al. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 2005;366(9491):1079-1084.
- Kyne L, Warny M, Qamar A, Kelly CP. Association between antibody response to toxin A and protection against recurrent Clostridium difficile diarrhoea. Lancet. 2001;357(9251):189-193.
- Debast SB, Bauer MP, Kuijper EJ; European Society of Clinical Microbiology and Infectious Diseases. European Society of Clinical Microbiology and Infectious Diseases: update of the treatment guidance document for Clostridium difficile infection. Clin Microbiol Infect. 2014;20 Suppl 2:1-26.
- Tixier EN, Verheyen E, Ungaro RC, Grinspan AM. Faecal microbiota transplant decreases mortality in severe and fulminant Clostridioides difficile infection in critically ill patients. Aliment Pharmacol Ther. 2019;50(10):1094-1099.
- Gerding DN, Muto CA, Owens RC. Treatment of Clostridium difficile infection. Clin Infect Dis. 2008;46 Suppl 1:S32-42.
Research Papers
The following PubMed topic searches retrieve current peer-reviewed literature on Clostridium difficile. Each link opens a live PubMed query so you always see the most recent publications.
- Clostridium difficile infection review
- Clostridioides difficile fidaxomicin vs vancomycin
- Fecal microbiota transplantation C difficile
- C difficile recurrence risk factors
- Proton pump inhibitor C difficile risk
- C difficile epidemiology hospital acquired
- Bezlotoxumab C difficile recurrence
- C difficile toxin A toxin B pathogenesis
- IDSA SHEA C difficile guideline
- Saccharomyces boulardii C difficile probiotic
- C difficile antibiotic stewardship
- Ribotype 027 Clostridium difficile
Connections
- Inflammatory Bowel Disease
- Probiotics
- Oral Microbiome
- Gut Healing
- Oregano
- Goldenseal
- Bone Broth
- Silver Nanoparticles
- Irritable Bowel Syndrome
- Gut-Brain Axis
- Staphylococcus Aureus
- Inflammatory Markers
- Bloating
- Gut Microbiome
- Parasites
- Kidney Disease
- Creatinine
- Sepsis