Peptic Ulcer Disease
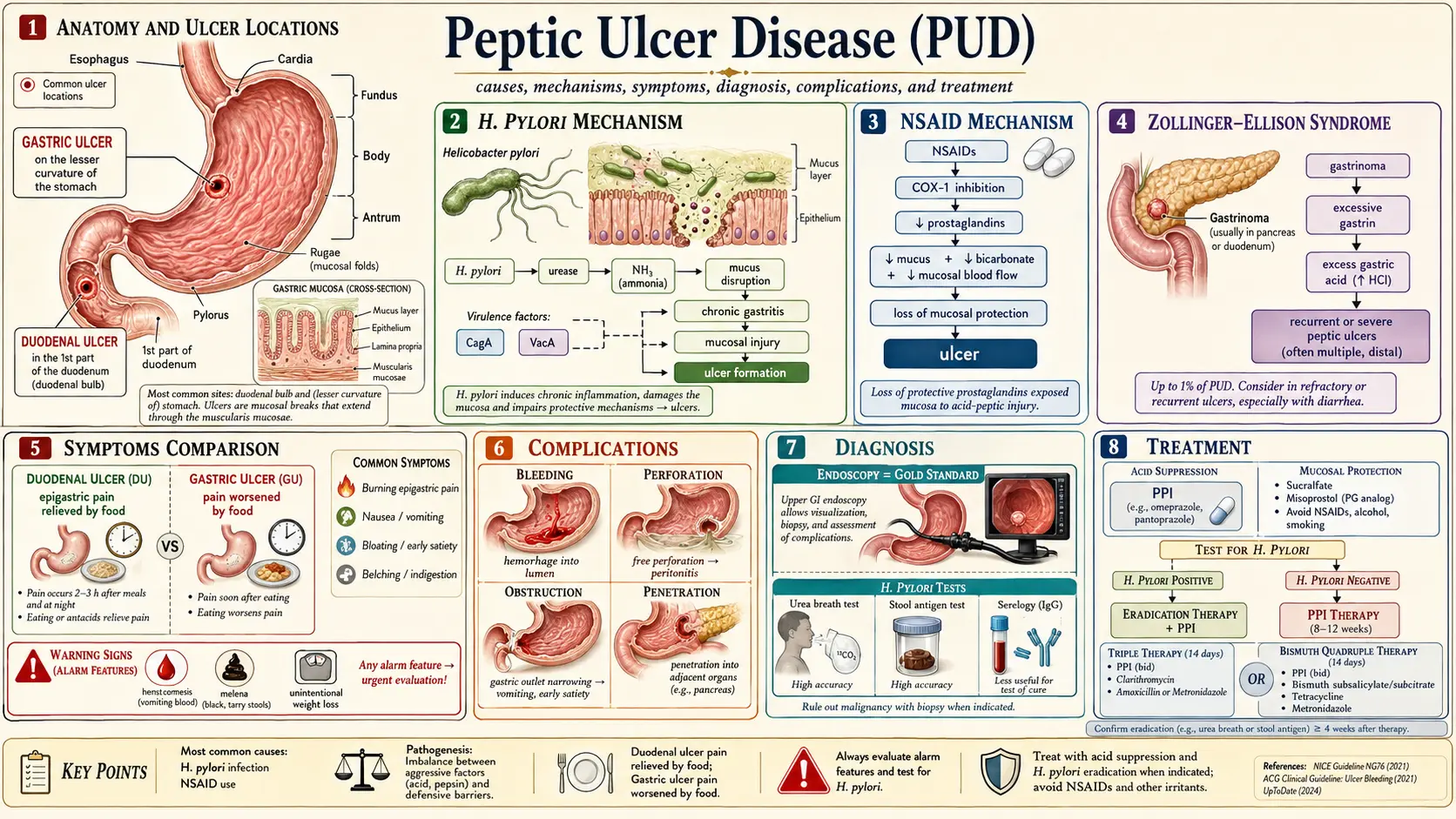
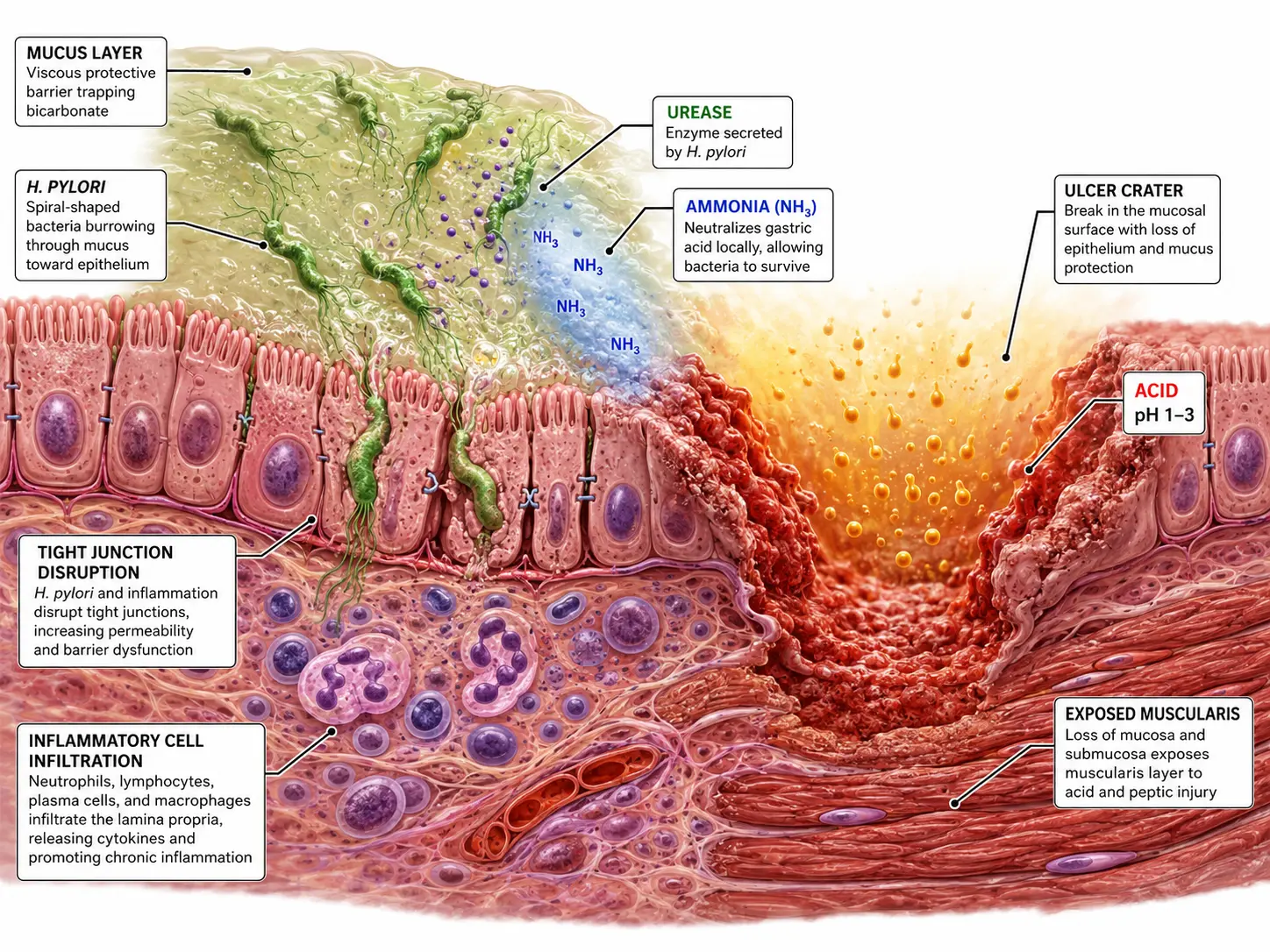
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
Peptic ulcer disease (PUD) refers to mucosal breaks exceeding 5 mm in diameter with appreciable depth, extending through the muscularis mucosae, occurring in regions exposed to acid and pepsin — predominantly the gastric antrum, duodenal bulb, and, less commonly, the distal esophagus, jejunum (in Zollinger-Ellison syndrome), or Meckel's diverticulum. The fundamental pathophysiological principle is an imbalance between aggressive mucosal damaging factors (hydrochloric acid, pepsin, Helicobacter pylori, NSAIDs) and protective mucosal defense mechanisms (mucus-bicarbonate barrier, prostaglandins, mucosal blood flow, epithelial restitution).
Duodenal ulcers (DU) are approximately four times more common than gastric ulcers (GU) in Western populations, though the incidence of NSAID-associated gastric ulcers has increased with widespread NSAID use. Duodenal ulcers are virtually always benign, while gastric ulcers require biopsy to exclude malignancy.
2. Epidemiology
The lifetime prevalence of PUD is approximately 5–10% in Western populations, with an annual incidence of 0.1–0.3%. The overall incidence of PUD has declined significantly over the past 30 years, largely attributable to widespread H. pylori eradication therapy and the introduction of proton pump inhibitors (PPIs). However, NSAID-associated PUD has increased in parallel with aging populations and expanding NSAID use.
The estimated annual incidence in the United States is 500,000 new cases and 4 million recurrences. PUD results in approximately 100,000 hospital admissions and 5,000–6,500 deaths annually in the US, primarily from hemorrhagic or perforated ulcers. The male-to-female ratio for DU is approximately 2:1; for GU, the ratio approaches 1:1 in NSAID-associated disease.
Prevalence of H. pylori infection varies widely — approximately 30–40% in developed countries and 70–90% in developing countries — and is the primary determinant of geographic variation in PUD incidence.
3. Pathophysiology
Protective Mucosal Defense Mechanisms
- Pre-epithelial layer: Mucus gel layer (5–450 µm) secreted by surface mucous cells; unstirred water layer trapping bicarbonate ions; bicarbonate secretion by surface epithelial cells (stimulated by prostaglandins E2 and I2) maintains a pH 6–7 microenvironment at the epithelial surface despite luminal pH of 1–2
- Epithelial layer: Tight junctions preventing back-diffusion of H+ ions; rapid epithelial restitution following superficial injury (migration of adjacent cells within hours); prostaglandin synthesis stimulating mucus and bicarbonate secretion, inhibiting acid secretion, and promoting mucosal blood flow
- Post-epithelial layer: Robust mucosal microvasculature delivering oxygen, bicarbonate, and nutrients; removal of toxic metabolites
Helicobacter pylori Pathogenesis
H. pylori (a gram-negative, spiral, microaerophilic bacillus) disrupts mucosal defenses through multiple mechanisms:
- Urease production: Generates ammonia from urea, neutralizing local pH for bacterial survival and directly damaging epithelial cells
- VacA (vacuolating cytotoxin A): Forms anion channels in epithelial cell membranes; induces vacuolation, apoptosis, and mitochondrial dysfunction; modulates immune responses
- CagA (cytotoxin-associated gene A protein): Translocated via Type IV secretion system into host cells; phosphorylated by Src and Abl kinases; activates CagA-dependent oncogenic signaling (RAS-ERK, SHP2); associated with higher virulence, increased gastric cancer risk
- Hypergastrinemia: H. pylori antral infection suppresses somatostatin (D cell product), thereby removing tonic inhibition of gastrin release from G cells; hypergastrinemia stimulates parietal cell acid hypersecretion (DU pathogenesis)
- Corpus gastritis: H. pylori corpus predominant gastritis damages parietal cells and chief cells, reducing acid output; associated with GU and gastric cancer risk
NSAID Pathogenesis
NSAIDs cause mucosal injury through both topical and systemic mechanisms:
- Topical injury: Acidic NSAIDs (aspirin, ibuprofen, naproxen) are weak acids that remain un-ionized and lipophilic at gastric pH; penetrate the mucosal phospholipid membrane; ionize intracellularly, becoming trapped and causing direct cell injury, mitochondrial uncoupling, and oxidative stress
- Systemic COX inhibition: NSAIDs inhibit cyclooxygenase-1 (COX-1, constitutively expressed) and COX-2 (inducible); COX-1 inhibition reduces prostaglandin E2 (PGE2) and PGI2 synthesis in the gastric mucosa — impairing mucus and bicarbonate secretion, reducing mucosal blood flow, and increasing epithelial apoptosis; COX-2 selective inhibitors (celecoxib) spare COX-1 and carry significantly lower GI risk
- The combination of NSAID use with H. pylori infection is synergistic — carrying a 3- to 4-fold greater ulcer risk than either factor alone
4. Etiology and Risk Factors
Primary causes:
- H. pylori infection: accounts for 70–80% of DU and 60–70% of GU in non-NSAID users
- NSAID/aspirin use: accounts for most of the remaining cases; dose-dependent risk; naproxen > ibuprofen > diclofenac in GI toxicity
Less common causes:
- Zollinger-Ellison syndrome (ZES): gastrin-secreting pancreatic or duodenal neuroendocrine tumor (gastrinoma); presents with severe, multiple, refractory, or atypically located ulcers; basal acid output typically >15 mEq/h
- Stress ulcers: superficial mucosal erosions in critically ill patients (Cushing's ulcer in CNS injury — high acid; Curling's ulcer in burn patients — ischemia-mediated)
- Idiopathic ulcer: 5–10% of PUD in H. pylori-negative, NSAID-negative patients; poorly understood; possibly related to viral infection, vascular disease, or cocaine use
- Other: CMV or HSV ulcers in immunocompromised patients; Crohn's disease; radiation-induced
Modifying risk factors:
- Cigarette smoking (impairs prostaglandin synthesis, reduces mucosal blood flow)
- Concurrent corticosteroid use (particularly with NSAIDs — synergistic risk)
- Anticoagulant and antiplatelet therapy (increase bleeding risk from pre-existing ulcers)
- Age >65 years
- Prior history of PUD or upper GI bleed
- Alcohol (direct mucosal irritant, delays healing)
- Psychological stress (evidence is indirect and observational)
5. Clinical Presentation
Classic symptoms:
- Epigastric pain: Burning, gnawing, or hunger-like; the most common presenting symptom
- Duodenal ulcer: Pain typically 2–5 hours after meals; relieved by food or antacids; nocturnal pain awakening from sleep (11 PM–2 AM) is characteristic
- Gastric ulcer: Pain often precipitated or worsened by food; less predictable postprandial pattern; weight loss more common due to food aversion
- Nausea and vomiting (particularly with pyloric channel ulcers causing gastric outlet obstruction)
- Anorexia and early satiety
- Heartburn and water brash
Important caveats:
- Up to 40–70% of NSAID-induced ulcers are asymptomatic ("silent ulcers") and present acutely with hemorrhage or perforation
- Symptom-based diagnosis is unreliable — only 25% of patients with typical "ulcer symptoms" have PUD on endoscopy
- Atypical presentations are more common in the elderly and in immunocompromised patients
Alarm features:
- Hematemesis or melena (upper GI hemorrhage)
- Sudden onset severe abdominal pain (perforation)
- Persistent vomiting (gastric outlet obstruction)
- Unintentional weight loss
- Dysphagia
- Iron-deficiency anemia
- Palpable abdominal mass
6. Diagnosis
Upper Endoscopy (EGD)
The gold standard for PUD diagnosis. Advantages over barium radiography include direct visualization, biopsy capability, and therapeutic intervention. Endoscopy is mandatory:
- In all patients with gastric ulcers — to obtain biopsies (4 from rim, 2 from base) to exclude malignancy; follow-up endoscopy at 6–8 weeks to confirm healing is recommended for gastric ulcers
- In any patient with alarm features
- For therapeutic intervention in acute upper GI bleeding
- When barium swallow is non-diagnostic
Forrest Classification (Endoscopic Staging of Bleeding Peptic Ulcers)
- Ia: Active spurting hemorrhage — 90% rebleeding risk without intervention
- Ib: Active oozing hemorrhage — 30–50% rebleeding risk
- IIa: Non-bleeding visible vessel — 43–55% rebleeding risk; endoscopic therapy indicated
- IIb: Adherent clot — 22–30% rebleeding risk; consider clot removal and treatment
- IIc: Flat pigmented spot — 7–10% rebleeding risk; endoscopic therapy not routinely required
- III: Clean ulcer base — 3–5% rebleeding risk; endoscopic therapy not indicated
H. pylori Testing
All patients with PUD should be tested for H. pylori. Testing strategies:
- Non-invasive tests:
- Urea breath test (UBT): 13C or 14C-labeled urea; sensitivity and specificity >95%; preferred for test-and-treat strategy and confirmation of eradication
- Stool antigen test (HpSA): Monoclonal antibody-based; sensitivity and specificity >94%; equivalent to UBT; less expensive
- Serology (IgG anti-H. pylori): Cannot distinguish active from past infection; not recommended for post-treatment testing
- Invasive tests (endoscopy-based):
- Rapid urease test (RUT): Antral biopsy in urease-containing medium; result in 1 hour; sensitivity 90–95%, specificity >95%
- Histology: H&E + Giemsa or Warthin-Starry staining; allows simultaneous assessment of gastritis pattern, atrophy, and intestinal metaplasia
- Culture: Gold standard for antibiotic susceptibility testing; indicated when eradication fails
- Note: PPIs, bismuth, and antibiotics reduce H. pylori test sensitivity; UBT and stool antigen should be performed ≥4 weeks after antibiotics and ≥2 weeks after PPI cessation
Serum Gastrin Level
Indicated in patients with multiple ulcers, refractory ulcers, ulcers in atypical locations (jejunum, post-bulbar), or prominent gastric rugal folds to exclude ZES. Fasting serum gastrin >1000 pg/mL with gastric pH <2.0 is diagnostic; secretin stimulation test (rise >200 pg/mL above baseline) confirms ZES if gastrin is 100–1000 pg/mL.
7. Treatment
Acid Suppression — Proton Pump Inhibitors (PPIs)
PPIs are the cornerstone of PUD treatment. They irreversibly inhibit the H+/K+-ATPase (proton pump) on the apical surface of gastric parietal cells, reducing intragastric acidity by 80–95%. PPIs should be taken 30–60 minutes before the first meal to maximize efficacy:
- Standard dose for uncomplicated PUD: omeprazole 20 mg, lansoprazole 30 mg, pantoprazole 40 mg, rabeprazole 20 mg, or esomeprazole 40 mg — once daily for 4–8 weeks
- DU: 4 weeks of PPI therapy achieves >90% healing rates
- GU: 8 weeks required due to larger ulcer size and thicker stomach wall; confirm healing endoscopically
- Maintenance PPI therapy is recommended for patients on chronic NSAIDs, aspirin, or anticoagulants, and for patients with prior complicated ulcers
H. pylori Eradication Regimens
Eradication reduces the 1-year DU recurrence rate from 80% to <10%. Selection of regimen should be guided by local clarithromycin resistance rates (<15% threshold for use):
- Standard triple therapy (14 days): PPI (standard dose BID) + clarithromycin 500 mg BID + amoxicillin 1 g BID; eradication rates 70–85%; no longer first-line where clarithromycin resistance >15%
- Bismuth quadruple therapy (14 days): PPI BID + bismuth subcitrate 120–300 mg QID + tetracycline 500 mg QID + metronidazole 500 mg TID; eradication rates 85–95%; preferred first-line in areas with high clarithromycin resistance; also used as salvage therapy
- Concomitant therapy (14 days): PPI BID + clarithromycin 500 mg BID + amoxicillin 1 g BID + metronidazole 500 mg BID; overcomes clarithromycin resistance; eradication rates >90%
- Levofloxacin-based triple therapy: PPI BID + levofloxacin 500 mg daily + amoxicillin 1 g BID × 14 days; recommended as second-line after failure of clarithromycin-based regimen
- Eradication confirmation: UBT or stool antigen test ≥4 weeks after completing antibiotic course and ≥2 weeks after PPI cessation
NSAID-Associated PUD Management
- Discontinue NSAID if possible; switch to acetaminophen, topical NSAIDs, or selective COX-2 inhibitors if pain control is required
- PPI co-therapy with NSAID or aspirin: omeprazole 20 mg daily reduces NSAID-associated ulcer risk by 80%; essential for high-risk patients (prior PUD, age >65, concurrent corticosteroids or anticoagulants)
- Misoprostol (prostaglandin E1 analogue, 200 µg QID) is an alternative gastroprotective agent but is limited by diarrhea and need for four-times-daily dosing
Endoscopic Hemostasis for Bleeding Ulcers
For Forrest Ia, Ib, and IIa ulcers, dual endoscopic therapy is recommended:
- Injection therapy: epinephrine (1:10,000) injection — reduces blood flow locally; should not be used as monotherapy
- Thermal coagulation: heater probe, bipolar (BICAP) electrocoagulation, or argon plasma coagulation (APC)
- Mechanical clips (hemoclips): equally effective as thermal therapy; particularly useful for visible vessels
- High-dose PPI infusion post-endoscopy: pantoprazole or omeprazole 80 mg IV bolus then 8 mg/h continuous infusion × 72 hours reduces rebleeding risk by maintaining intragastric pH >6 and stabilizing clot
Surgical Treatment
Surgery for PUD has dramatically declined in the PPI era but remains necessary for:
- Perforation: primary closure with Graham patch (omental patch); laparoscopic approach preferred when feasible
- Refractory bleeding uncontrolled by endoscopy or interventional radiology
- Gastric outlet obstruction unresponsive to endoscopic dilation
- Suspicion of malignancy on gastric ulcer
- Zollinger-Ellison syndrome: surgical resection of gastrinoma when localized
8. Complications
- Upper gastrointestinal hemorrhage (UGIH): Most common complication; occurs in 15–20% of PUD patients; peptic ulcer is the cause of 50% of all UGIH; presentations range from occult blood loss to massive hematemesis; mortality 5–10%
- Perforation: Occurs in 2–10% of PUD; typically presents as sudden onset severe periumbilical or generalized abdominal pain with peritoneal signs; free air under the diaphragm on erect chest X-ray or CT; mortality 10–40% depending on time to surgery and patient comorbidity
- Penetration: Posterior DU penetrates into the pancreas (presenting as pancreatitis or back pain); posterior GU may penetrate into the liver or lesser sac
- Gastric outlet obstruction: Results from edema and/or fibrotic scarring of the pyloric channel or duodenal bulb; presents with recurrent vomiting of undigested food, early satiety, and weight loss; Succussion splash positive; metabolic alkalosis with hypokalemia and hypochloremia (from repeated vomiting of HCl)
- Malignant transformation: Not applicable to DU; gastric ulcers may represent malignancy — all gastric ulcers require endoscopic biopsy and histological confirmation of healing
9. Prognosis
The prognosis of uncomplicated PUD is excellent with appropriate treatment. H. pylori eradication leads to ulcer healing and markedly reduces recurrence: 1-year recurrence rates fall from 70–80% (without eradication) to <5–10% after successful eradication. Patients whose ulcers are not attributable to H. pylori or NSAIDs (idiopathic) have the highest recurrence rates and may require indefinite PPI maintenance.
Complicated PUD (bleeding, perforation) carries significantly higher morbidity and mortality. Risk stratification tools such as the Blatchford score (pre-endoscopy) and Rockall score (post-endoscopy) guide triage decisions. Mortality from UGIH in PUD is approximately 5–10%, increasing to 30–40% in perforated ulcers, particularly in the elderly. Long-term outcomes after surgery for PUD complications include post-vagotomy syndromes (dumping syndrome, diarrhea, afferent/efferent loop syndrome) in patients who underwent partial gastrectomy.
10. Prevention
- H. pylori eradication in infected individuals: Prevents ulcer recurrence; may reduce gastric cancer risk particularly when performed before the development of atrophic gastritis or intestinal metaplasia
- PPI gastroprotection with NSAIDs: Strongly recommended for high-risk patients on chronic NSAIDs (prior ulcer, age >65, concurrent anticoagulants or corticosteroids); reduces NSAID-associated ulcer risk by 80%
- COX-2 selective inhibitors: 3–5-fold lower incidence of symptomatic ulcers compared to non-selective NSAIDs; cardiovascular risk should be assessed before prescribing
- Tobacco cessation: Smoking impairs ulcer healing, reduces PPI efficacy, and promotes recurrence
- Stress ulcer prophylaxis: IV PPI or H2RA in mechanically ventilated ICU patients (high-risk) reduces the incidence of clinically significant stress ulcer bleeding; not recommended for low-risk hospitalized patients due to risks of Clostridioides difficile infection and pneumonia
- Alcohol moderation: Reduces direct mucosal irritation and improves adherence to treatment
11. Recent Research and Advances
- Potassium-competitive acid blockers (P-CABs): Vonoprazan (TAK-438) is a novel acid suppressor that competitively inhibits H+/K+-ATPase by binding to the K+ site; achieves faster, more consistent, and more potent acid suppression than PPIs; approved in Japan and Southeast Asia; FDA-approved in the US in 2022 (Voquezna) for H. pylori eradication and erosive GERD; Phase 3 trials demonstrate superior eradication rates compared to omeprazole-based triple therapy, particularly with clarithromycin-resistant strains.
- Antibiotic resistance surveillance: Global H. pylori clarithromycin resistance rates now exceed 15–20% in most Western countries; sequential resistance testing and culture-guided therapy are increasingly recommended as first-line approaches in academic centers.
- Rifabutin-based rescue therapy: Vonoprazan + amoxicillin + rifabutin (Talicia) — FDA-approved in 2019; effective against metronidazole and clarithromycin-resistant strains; restricted use to preserve rifabutin activity against mycobacterial infections.
- Endoscopic over-the-scope clip (OTSC): Superior to standard hemoclips for refractory or recurrent high-risk bleeding ulcers; STING trial (2020) demonstrated significantly lower 30-day rebleeding rates with OTSC as first-line therapy for Forrest Ia/IIa ulcers compared with standard therapy.
- Hemostatic powder sprays: Hemospray (TC-325) — inorganic mineral powder applied endoscopically; acts as a mechanical barrier and activates coagulation cascade; effective as bridge therapy for massive hemorrhage pending definitive therapy.
- Microbiome and PUD: Emerging data suggest that gastric and duodenal microbiome dysbiosis beyond H. pylori contributes to PUD susceptibility; multi-omic studies are characterizing the gastric microbiome in ulcer-prone versus cancer-prone H. pylori-positive individuals.
12. References
- Lanas A, Chan FKL. Peptic ulcer disease. Lancet. 2017;390(10094):613–624.
- Malfertheiner P, Megraud F, Rokkas T, et al. Management of Helicobacter pylori infection: the Maastricht VI/Florence consensus report. Gut. 2022;71(9):1724–1762.
- Chey WD, Leontiadis GI, Howden CW, Moss SF. ACG Clinical Guideline: Treatment of Helicobacter pylori Infection. Am J Gastroenterol. 2017;112(2):212–239.
- Lau JY, Barkun A, Fan DM, et al. Challenges in the management of acute peptic ulcer bleeding. Lancet. 2013;381(9882):2033–2043.
- Sung JJ, Chiu PW, Chan FKL, et al. Asia-Pacific working group consensus on non-variceal upper gastrointestinal bleeding: an update 2018. Gut. 2018;67(10):1757–1768.
- Gisbert JP, Calvet X. Review article: the effectiveness of standard triple therapy for Helicobacter pylori has not changed over the last decade, but it is not good enough. Aliment Pharmacol Ther. 2011;34(11–12):1255–1268.
- Chan FK, Hung LC, Suen BY, et al. Celecoxib versus diclofenac and omeprazole in reducing the risk of recurrent ulcer bleeding in patients with arthritis. N Engl J Med. 2002;347(26):2104–2110.
- Sreedharan A, Martin J, Leontiadis GI, et al. Proton pump inhibitor treatment initiated prior to endoscopic diagnosis in upper gastrointestinal bleeding. Cochrane Database Syst Rev. 2010;(7):CD005415.
- Graham DY, Tansel A. Interchangeable Use of Proton Pump Inhibitors Based on Relative Potency. Clin Gastroenterol Hepatol. 2018;16(6):800–808.
- Miwa H, Uedo N, Watari J, et al. Randomised clinical trial: efficacy and safety of vonoprazan vs. lansoprazole in patients with gastric or duodenal ulcers. Aliment Pharmacol Ther. 2017;45(2):240–252.
- Blatchford O, Murray WR, Blatchford M. A risk score to predict need for treatment for upper-gastrointestinal haemorrhage. Lancet. 2000;356(9238):1318–1321.
- Forrest JA, Finlayson ND, Shearman DJ. Endoscopy in gastrointestinal bleeding. Lancet. 1974;2(7877):394–397.
- Gisbert JP, Abraira V. Accuracy of Helicobacter pylori diagnostic tests in patients with bleeding peptic ulcer: a systematic review and meta-analysis. Am J Gastroenterol. 2006;101(4):848–863.
- Schmulson MJ, Drossman DA. What Is New in Rome IV. J Neurogastroenterol Motil. 2017;23(2):151–163.
- Kavitt RT, Lipowska AM, Anyane-Yeboa A, Gralnek IM. Diagnosis and Treatment of Peptic Ulcer Disease. Am J Med. 2019;132(4):447–456.
- Barkun AN, Almadi M, Kuipers EJ, et al. Management of Nonvariceal Upper Gastrointestinal Bleeding: Guideline Recommendations From the International Consensus Group. Ann Intern Med. 2019;171(11):805–822.
Research Papers
Curated PubMed topic searches of peer-reviewed literature on Peptic Ulcer Disease. Each link opens a live PubMed query so you always see the most current studies.
- PubMed: H. pylori and peptic ulcer
- PubMed: NSAID-induced peptic ulcer
- PubMed: H. pylori eradication
- PubMed: Bleeding peptic ulcer
- PubMed: PPI and ulcer healing
- PubMed: Perforated peptic ulcer
- PubMed: Zollinger-Ellison syndrome
- PubMed: Endoscopic diagnosis
- PubMed: Antibiotic resistance in H. pylori
- PubMed: Duodenal ulcer pathophysiology
- PubMed: Stress ulcer prophylaxis
- PubMed: Epidemiology of peptic ulcer
Connections
- Gastroesophageal Reflux Disease
- SIBO
- Irritable Bowel Syndrome
- Gut Healing
- Licorice (DGL)
- Ginger
- Turmeric
- Zinc
- Vitamin C
- Pancreatitis
- Crohn's Disease
- Gallbladder Disease
- Inflammatory Bowel Disease
- Abdominal Pain
- Anemia
- Nausea and Vomiting
- Aspirin
- Diverticulitis