Cholesterol Management
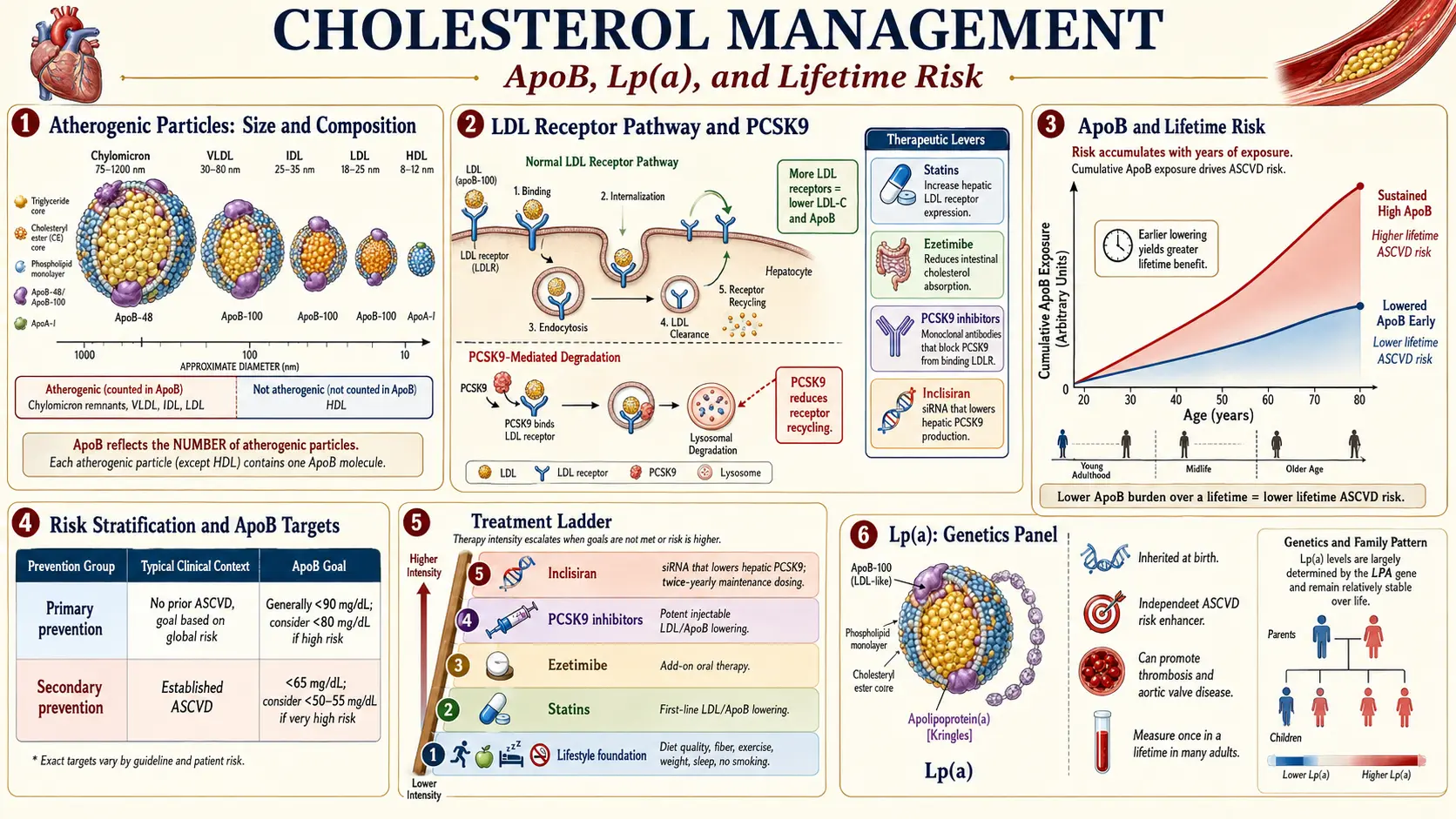
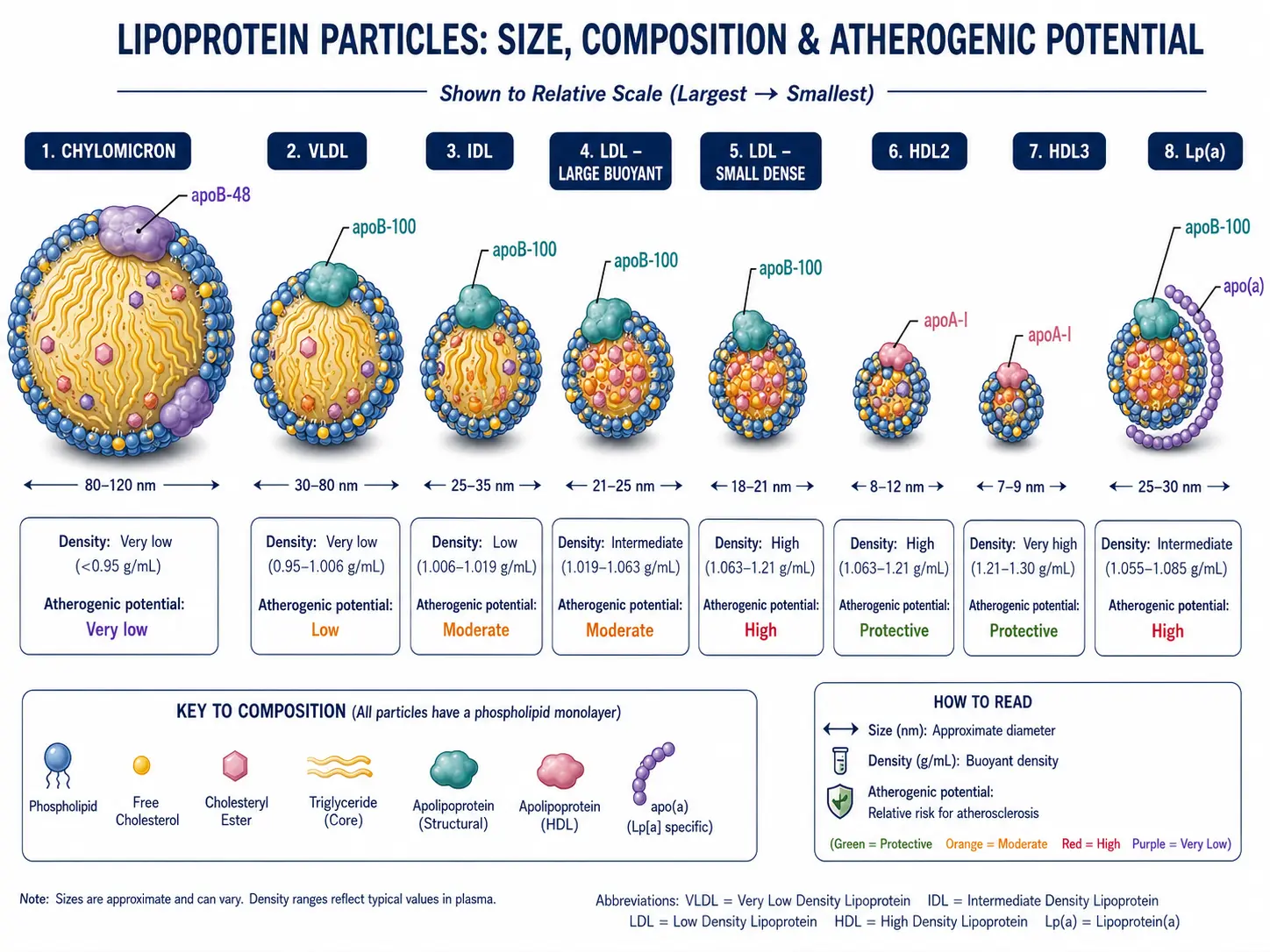
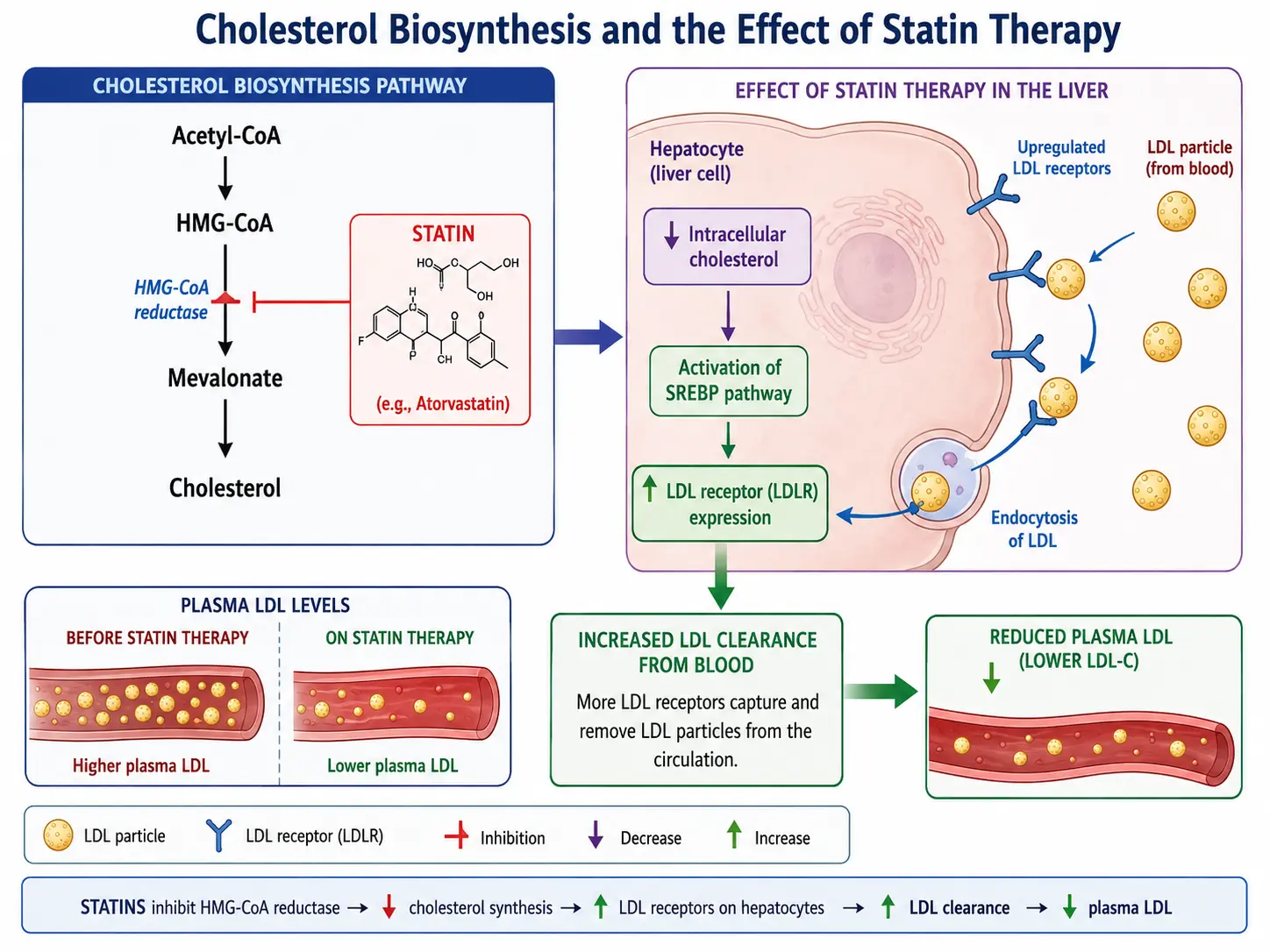
Table of Contents
- What is Cholesterol Management?
- Lipid Particles Explained
- Lab Targets and Goals
- Statins and How They Work
- Beyond Statins
- Lifestyle Cornerstones
- Special Populations
- Research Papers
- Connections
- Featured Videos
What is Cholesterol Management?
Cholesterol management is the long-term, risk-based process of lowering atherogenic (artery-clogging) lipoproteins in the blood to prevent heart attacks, strokes, and peripheral artery disease. The goal is not to chase a "normal cholesterol" number on a generic chart — it is to drive the particles that drive plaque as low as your personal risk warrants, and to keep them there for life.
Most people are still told their "total cholesterol" or even just their "LDL cholesterol" (LDL-C). Both numbers matter, but both can mislead. Total cholesterol lumps the bad (LDL, VLDL, Lp(a)) with the good (HDL). LDL-C measures the weight of cholesterol inside LDL particles — not the number of particles. Two people can have an identical LDL-C of 100 mg/dL, yet one has half as many LDL particles as the other. The person with more particles has the higher cardiovascular risk. That is why modern lipidology has shifted toward two better markers:
- ApoB — one molecule of apolipoprotein B sits on every atherogenic particle (LDL, VLDL, IDL, Lp(a), chylomicron remnant). One ApoB equals one particle. ApoB is the most direct measure of how many bullets are in the gun.
- Lp(a) — lipoprotein(a) is a genetic variant of LDL that is roughly six times more atherogenic, gram-for-gram, than ordinary LDL. About one in five people has elevated Lp(a) and most have never been tested. Lp(a) is the single biggest source of residual risk — the heart attacks that still happen in people whose LDL-C looks "fine."
Residual risk is the central problem. The major statin trials cut events by roughly 25-35 percent on top of background care. That is excellent — and it means 65-75 percent of events still happen. Closing that gap is what cholesterol management in 2026 is actually about: lower LDL further, find and treat Lp(a), reduce inflammation, and pick up subclinical plaque early with a coronary calcium score.
The framing principle: cholesterol is a lifetime cumulative exposure problem, not a single-decade problem. Plaque builds slowly over decades. Lower numbers earlier — even modestly — bend the lifetime curve dramatically.
Lipid Particles Explained
Cholesterol and triglycerides do not float freely in blood — they are wrapped in protein-coated spheres called lipoproteins. Each kind of particle has a different size, density, cargo, and atherogenic potential. The image above shows them roughly to scale.
Chylomicron
- Largest particle (75-1200 nm). Made by the small intestine after a fatty meal.
- Carries dietary triglycerides from the gut to the liver and peripheral tissues.
- Cleared rapidly — usually invisible after a 12-hour fast. Their remnants (after triglyceride drop-off) are atherogenic.
VLDL (Very-Low-Density Lipoprotein)
- Made by the liver to export triglycerides to the body. Roughly 30-80 nm.
- Each VLDL carries one ApoB-100. As triglycerides are stripped out, VLDL shrinks into IDL, then into LDL.
- VLDL remnants are atherogenic. High triglycerides means high VLDL means high non-HDL cholesterol — one reason non-HDL outperforms LDL-C alone.
IDL (Intermediate-Density Lipoprotein)
- Short-lived bridge between VLDL and LDL. Atherogenic when it accumulates (genetic dysbetalipoproteinemia, type 2 diabetes, kidney disease).
LDL (Low-Density Lipoprotein) — the main villain
- Roughly 18-25 nm. Carries about 70 percent of the cholesterol in the blood. One ApoB-100 per particle.
- Subtypes by size and density:
- Pattern A — large, buoyant LDL: bigger, lighter, less atherogenic. Common in people with low triglycerides and high HDL.
- Pattern B — small, dense LDL: smaller, heavier, oxidizes more easily, slips through the artery wall more readily, and stays trapped longer. Strongly linked to insulin resistance, high triglycerides, and metabolic syndrome.
- Same LDL-C number can hide very different particle counts. A high triglyceride / low HDL pattern almost always means more, smaller, denser, more atherogenic LDL particles. Hence ApoB.
HDL (High-Density Lipoprotein)
- Roughly 5-12 nm — the smallest. Reverse-cholesterol transport: pulls cholesterol out of the artery wall back to the liver.
- HDL2 (larger, more buoyant) is generally considered more cardioprotective than HDL3 (smaller, denser).
- HDL is associated with lower risk — but raising HDL pharmacologically (CETP inhibitors, niacin) has not reliably reduced events. HDL function (efflux capacity) matters more than HDL number.
Lp(a) — lipoprotein(a)
- An LDL particle with a long, kinky apolipoprotein(a) tail covalently bound to ApoB-100. Genetically determined — your level is set at birth and barely moves with diet, exercise, or statins.
- Roughly six times more atherogenic per particle than LDL. Promotes plaque, calcification, thrombosis, and aortic stenosis.
- Affects an estimated 20 percent of the population. Test once in a lifetime. Currently no approved therapy specifically lowers Lp(a) to a target, though several are in late-stage trials (pelacarsen, olpasiran, lepodisiran). PCSK9 inhibitors lower Lp(a) by 20-30 percent as a side benefit.
Lab Targets and Goals
"Optimal" lipid numbers depend on your overall cardiovascular risk. The same LDL-C of 110 mg/dL is reasonable for a 25-year-old with no risk factors and unacceptable for a 60-year-old with diabetes and a prior heart attack. Modern guidelines (ACC/AHA, ESC, NLA) stratify patients into risk tiers, then set lower-is-better targets within each tier.
The Five Numbers That Matter Most
- LDL-C (LDL cholesterol) — still the workhorse marker. Calculated or directly measured.
- ApoB — the particle count. Increasingly preferred when LDL-C and ApoB disagree.
- Lp(a) — one-time genetic test. Reported as nmol/L (preferred) or mg/dL.
- Non-HDL cholesterol — total minus HDL. Captures all atherogenic particles in one number. Free on every standard lipid panel.
- Triglycerides — high triglycerides drive small-dense LDL and reflect insulin resistance.
Goals by Risk Tier (ACC/AHA framework, simplified)
Primary Prevention — Low Risk
- 10-year ASCVD risk under 5 percent, no diabetes, no family history of premature CAD.
- LDL-C goal: < 130 mg/dL (ideally < 100).
- ApoB goal: < 90 mg/dL.
- Non-HDL: < 160 mg/dL.
Primary Prevention — Borderline / Intermediate Risk
- 10-year risk 5-20 percent, or one major risk factor (hypertension, diabetes, smoking, family history, elevated Lp(a), CAC > 0).
- LDL-C goal: < 100 mg/dL, ideally < 70.
- ApoB goal: < 80 mg/dL.
- Strong indication for statin if CAC > 100 Agatston units or any plaque visible.
Secondary Prevention — Established ASCVD or "Very High Risk"
- Prior heart attack, stroke, peripheral artery disease, coronary stent or bypass, diabetes with target organ damage, familial hypercholesterolemia, CAC > 300.
- LDL-C goal: < 70 mg/dL at minimum; current ESC guidelines push < 55 mg/dL, and very-high-risk recurrent-event patients are often targeted to < 40.
- ApoB goal: < 65 mg/dL (very high risk < 50).
- Non-HDL: < 100 mg/dL (very high risk < 80).
Triglycerides
- < 150 mg/dL: optimal.
- 150-499: borderline-to-high; mostly a lifestyle and metabolic problem.
- > 500: pancreatitis risk — treat aggressively.
HDL and the Total/HDL Ratio
- HDL > 40 mg/dL men, > 50 women is the conventional bar — but raising HDL pharmacologically does not reduce events. Use HDL as a risk marker, not a treatment target.
- Total cholesterol / HDL ratio < 3.5 is generally favorable; > 5 unfavorable.
Lp(a)
- < 75 nmol/L (~30 mg/dL): low risk.
- 75-125 nmol/L: moderate.
- > 125 nmol/L: elevated — treat LDL/ApoB more aggressively to compensate.
- > 250 nmol/L: very high — consider PCSK9 inhibitor and screen first-degree relatives.
Statins and How They Work
Statins are the most-studied medication class in human history — tens of millions of patient-years across dozens of randomized trials. They are the foundation of LDL lowering and the only lipid drug with both consistent event reduction and proven plaque regression on intravascular ultrasound. The image above shows the molecular mechanism.
The Mechanism in Plain Language
The liver makes most of the body's cholesterol. The rate-limiting step in that synthesis pathway is an enzyme called HMG-CoA reductase. Statins are competitive inhibitors of HMG-CoA reductase — they sit in the enzyme's active site and block it.
Two things happen when liver cells make less cholesterol internally:
- The liver puts more LDL receptors on its surface to grab cholesterol from the blood.
- Those LDL receptors pull LDL particles (and VLDL remnants and IDL) out of circulation and recycle them.
The net result: blood LDL drops by 20-55 percent, depending on which statin and what dose. ApoB drops in parallel. Triglycerides drop modestly (10-30 percent). HDL nudges up slightly. Plaque often regresses on imaging when LDL stays below ~70 mg/dL for a year or more.
Beyond LDL Lowering — Pleiotropic Effects
Statins do more than lower cholesterol. They reduce vascular inflammation (CRP drops along with LDL), stabilize existing plaque so it is less likely to rupture, improve endothelial function, and have mild anti-thrombotic effects. The "JUPITER" trial showed that even people with normal LDL but elevated CRP benefited — suggesting some of the event reduction is independent of LDL.
Intensity Tiers (ACC/AHA classification)
High-Intensity Statins (LDL reduction ~50 percent or more)
- Atorvastatin 40-80 mg — first-line for secondary prevention. Used in PROVE-IT, TNT, IDEAL.
- Rosuvastatin 20-40 mg — most potent per milligram; least drug-interaction-prone of the high-potency statins. Used in JUPITER, GISSI-HF.
Moderate-Intensity Statins (LDL reduction ~30-50 percent)
- Atorvastatin 10-20 mg
- Rosuvastatin 5-10 mg
- Simvastatin 20-40 mg (40 only; 80 mg withdrawn for myopathy risk)
- Pravastatin 40-80 mg — least interactions, often used in elderly.
- Lovastatin 40 mg, Fluvastatin 80 mg, Pitavastatin 1-4 mg.
Low-Intensity Statins (LDL reduction < 30 percent)
- Reserved for statin-intolerant patients who can tolerate small doses, or as starter doses.
Landmark Statin Trials
- 4S (1994) — simvastatin in patients with prior MI cut total mortality 30 percent. The trial that changed cardiology.
- WOSCOPS, AFCAPS/TexCAPS — primary prevention works in elevated-LDL patients without prior events.
- PROVE-IT (2004) — intensive (atorvastatin 80) beat moderate (pravastatin 40) post-ACS. Established "lower is better."
- JUPITER (2008) — rosuvastatin in high-CRP patients with normal LDL still cut events 44 percent.
- HOPE-3 (2016) — primary prevention in intermediate-risk patients.
- FOURIER (2017) and ODYSSEY OUTCOMES (2018) — PCSK9 inhibitors on top of maximally tolerated statin further reduced events with no LDL floor seen.
Beyond Statins
If maximally tolerated statin therapy does not reach your LDL-C / ApoB target — or you cannot tolerate statins at all — there are six well-studied add-on or alternative classes.
1. Ezetimibe
- Mechanism: Blocks the NPC1L1 cholesterol transporter in the small intestine, reducing dietary and biliary cholesterol absorption.
- LDL effect: Additional 18-25 percent on top of statin.
- Evidence: IMPROVE-IT (2015) added ezetimibe to simvastatin in post-ACS patients and cut events 6 percent — the first non-statin LDL drug to prove the lower-is-better hypothesis on outcomes.
- Use case: Cheap, generic, well-tolerated. Almost everyone on a statin who isn't at goal should add ezetimibe before escalating further.
2. PCSK9 Inhibitors (Evolocumab, Alirocumab)
- Mechanism: Monoclonal antibodies that block PCSK9, a protein that destroys LDL receptors. Block PCSK9 and LDL receptors live longer, clearing more LDL.
- LDL effect: 50-60 percent reduction on top of statin. Lp(a) drops 20-30 percent as a bonus.
- Evidence: FOURIER and ODYSSEY OUTCOMES proved cardiovascular benefit. Driving LDL into the 20s and 30s reduced events further with no safety floor.
- Use case: Familial hypercholesterolemia, secondary prevention not at goal on statin + ezetimibe, statin intolerance, elevated Lp(a). Injection every 2-4 weeks. Expensive but increasingly covered.
3. Bempedoic Acid (Nexletol)
- Mechanism: Inhibits ATP-citrate lyase, an enzyme upstream of HMG-CoA reductase in the cholesterol synthesis pathway. Activated only in the liver, not muscle — so it should not cause myopathy.
- LDL effect: ~17 percent monotherapy, ~38 percent combined with ezetimibe.
- Evidence: CLEAR Outcomes (2023) showed bempedoic acid reduced major cardiovascular events 13 percent in statin-intolerant patients.
- Use case: Statin-intolerant patients with established ASCVD or familial hypercholesterolemia.
4. Inclisiran (Leqvio)
- Mechanism: Small interfering RNA (siRNA) that silences PCSK9 production in the liver. A "PCSK9 blocker" with twice-yearly dosing.
- LDL effect: ~50 percent reduction on top of statin, similar to monoclonal PCSK9 inhibitors.
- Evidence: ORION-9, ORION-10, ORION-11 confirmed durable LDL lowering. Outcome trials (ORION-4, VICTORION-2) ongoing.
- Use case: Patients who would benefit from PCSK9 inhibition but prefer two injections a year (administered in clinic) over self-injecting every 2-4 weeks.
5. Niacin (Nicotinic Acid)
- Effect: Lowers LDL ~15 percent, raises HDL 15-35 percent, lowers Lp(a) 20-30 percent, lowers triglycerides 20-50 percent.
- Evidence: AIM-HIGH and HPS2-THRIVE failed to show event reduction when added to statins, despite improving the lipid panel. Side effects (flushing, glucose intolerance, hepatotoxicity) are common.
- Current role: Niche — mainly for severe hypertriglyceridemia or selected Lp(a) cases when nothing else is available. Not a routine add-on.
6. Fibrates (Fenofibrate, Gemfibrozil)
- Mechanism: PPAR-alpha agonists. Lower triglycerides 30-50 percent, raise HDL 10-15 percent. Modest LDL effect.
- Evidence: ACCORD-Lipid suggested benefit only in the high-triglyceride / low-HDL subgroup. PROMINENT (pemafibrate, 2022) was negative.
- Use case: Triglycerides > 500 mg/dL to prevent pancreatitis; selective use in mixed dyslipidemia.
7. Omega-3 Fatty Acids (EPA / DHA)
- Effect: Lower triglycerides 20-30 percent at high doses (4 g/day). Modest LDL effects.
- Evidence: REDUCE-IT (icosapent ethyl 4 g/day) cut events 25 percent in statin-treated patients with elevated triglycerides. STRENGTH and VITAL (general fish oil) were largely negative.
- Use case: Prescription-grade EPA-only (icosapent ethyl) on top of statin for residual risk; high triglycerides.
Lifestyle Cornerstones
Drugs do not replace lifestyle — they layer on top of it. Lifestyle changes alone can lower LDL 15-30 percent, drop triglycerides 30-50 percent, and reduce all-cause mortality independently of any pill. The compounding effect of lifestyle plus medication is much greater than either alone.
1. Mediterranean Diet
- The most-studied dietary pattern for cardiovascular disease. PREDIMED (2013, re-published 2018) randomized 7,400 high-risk adults to Mediterranean diet plus extra-virgin olive oil or mixed nuts vs. low-fat control. The Mediterranean groups had ~30 percent fewer major cardiovascular events.
- Pattern: heavy on vegetables, legumes, whole grains, fruit, nuts, olive oil, fish; moderate dairy and poultry; low red and processed meat; minimal added sugar and refined flour.
- The benefit is most likely a combination of monounsaturated fat replacing saturated fat, polyphenols, fiber, and the displacement of ultra-processed food.
2. Soluble Fiber and Whole Grains
- Soluble fiber binds bile acids in the gut. The liver pulls cholesterol out of the blood to make replacement bile acids — net LDL drops.
- 5-10 g/day of soluble fiber lowers LDL by 5-11 mg/dL. Sources: oats (beta-glucan), psyllium husk, beans, lentils, apples, citrus, brussels sprouts.
- Psyllium 7-10 g/day is the most concentrated, evidence-based form.
3. Plant Sterols and Stanols
- Compete with cholesterol for absorption in the intestine. 2 g/day lowers LDL ~10 percent. Found in fortified spreads, yogurts, and supplements.
- Effect stacks additively with statins, ezetimibe, and fiber.
4. Soy Protein
- 25 g/day of soy protein (in place of animal protein) lowers LDL 4-6 percent. Modest but real, and additive to other measures.
- Soy also has a small blood-pressure-lowering effect.
5. Reduce Saturated Fat, Trans Fat, and Refined Carbohydrate
- Replacing saturated fat (red meat, butter, full-fat dairy) with mono- and polyunsaturated fat (olive oil, nuts, seeds, fish) consistently lowers LDL.
- Trans fats raise LDL and lower HDL — the worst macronutrient ever measured. Largely banned but still lurking in some packaged foods.
- Refined carbs and added sugar drive triglycerides and small-dense LDL through hepatic VLDL production. Cutting them lowers triglycerides faster than cutting fat does.
6. Exercise
- Aerobic exercise (150 min/week of moderate, or 75 min/week vigorous) raises HDL ~5 mg/dL, lowers triglycerides 10-20 percent, and shifts LDL toward larger, more buoyant subtypes.
- Resistance training adds independent metabolic benefit and improves insulin sensitivity.
- Effect on LDL-C alone is modest, but the cardiovascular event reduction is large — exercise reduces mortality more than most cholesterol drugs.
7. Weight Loss
- Every 10 pounds lost reduces LDL by ~5-8 mg/dL, triglycerides by 20 mg/dL, and raises HDL by 1-2 mg/dL.
- Visceral fat loss is the biggest lever — it drops VLDL output and reverses the small-dense-LDL pattern.
8. Smoking Cessation
- Smoking lowers HDL, oxidizes LDL, damages endothelium, and roughly doubles cardiovascular risk independent of cholesterol numbers.
- Within one year of quitting, cardiovascular risk drops by half. Within 10-15 years it approaches that of a never-smoker.
9. Alcohol Moderation
- Alcohol raises HDL but also raises triglycerides and blood pressure. The "J-curve" benefit is now considered overstated.
- For lipid management: keep alcohol low; eliminate it entirely if triglycerides are elevated.
Special Populations
Women
- Estrogen is mildly cardioprotective — women's cardiovascular risk lags men by roughly 10 years until menopause, then catches up.
- After menopause, LDL typically rises 10-15 percent. Hormone replacement therapy is not a cholesterol treatment — treat lipids per the same risk-based framework as men.
- Pregnancy: stop statins (Category X). Bile acid sequestrants (cholestyramine, colesevelam) are pregnancy-safe alternatives if needed.
- Women have historically been undertreated for cholesterol — symptoms of heart attack are also more atypical. Treat aggressively.
Elderly (75+)
- Statin benefit persists into the 80s for secondary prevention. Primary prevention in older adults is less clear-cut — weigh against polypharmacy, frailty, and competing causes of death.
- STAREE (in progress) and PREVENTABLE (in progress) are filling the evidence gap.
- Use lower starting doses; renal and hepatic function decline with age.
Type 2 Diabetes
- Diabetes is a "coronary heart disease equivalent" — treat to secondary-prevention targets even without prior events.
- Diabetic dyslipidemia is the small-dense-LDL / high-TG / low-HDL pattern. Statins are foundational; ezetimibe and PCSK9 stack on top.
- SGLT2 inhibitors and GLP-1 agonists (e.g., semaglutide) reduce cardiovascular events independently of LDL — add them when indicated.
Chronic Kidney Disease (CKD)
- CKD accelerates atherosclerosis. Most CKD patients qualify as high or very-high risk regardless of other factors.
- Statins and ezetimibe are first-line; SHARP trial (2011) showed 17 percent event reduction.
- Dose-adjust for kidney function (rosuvastatin in particular). Avoid PCSK9 dose changes — renal-cleared metabolism is minimal.
Familial Hypercholesterolemia (FH)
- Genetic disorder — LDL receptor or ApoB or PCSK9 mutation. Heterozygous FH affects ~1 in 250; homozygous ~1 in 250,000-1,000,000.
- LDL is typically > 190 mg/dL in heterozygous, > 400 in homozygous. Premature ASCVD, often first event in 30s-40s for men, 40s-50s for women.
- Treatment: high-intensity statin + ezetimibe almost always; PCSK9 inhibitor or inclisiran routinely added; LDL apheresis or evinacumab for homozygous FH.
- Cascade-screen first-degree relatives once a proband is identified.
Statin Intolerance
- True statin myopathy (CK-elevating muscle damage) is rare — well under 1 percent. Statin-associated muscle symptoms (SAMS) without CK rise are reported in 5-15 percent of users in observational studies, but blinded crossover trials (SAMSON, StatinWise) show most symptoms are nocebo-driven and resolve on placebo as readily as on statin.
- Practical approach: rechallenge with a different statin; try every-other-day dosing of long-half-life statins (rosuvastatin, atorvastatin); switch to bempedoic acid or PCSK9 inhibitor if symptoms persist on multiple agents.
- Real intolerance is uncommon but real — the goal is to keep LDL down, not to win an argument about which drug.
Elevated Lp(a)
- About 20 percent of the population. Currently no targeted therapy — lower the LDL/ApoB you can control to compensate.
- Aspirin for primary prevention may help in elevated-Lp(a) patients (uncertain — trials ongoing).
- PCSK9 inhibitors lower Lp(a) 20-30 percent as a side benefit. Niacin lowers it ~30 percent.
- Specific Lp(a) lowering drugs (pelacarsen, olpasiran, lepodisiran) are in late-stage outcome trials — potentially transformative if positive.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- 4S simvastatin survival study
- PROVE-IT atorvastatin intensive
- FOURIER evolocumab outcomes
- PCSK9 inhibitor cardiovascular outcomes
- ApoB cardiovascular risk
- Lipoprotein(a) management
- Ezetimibe IMPROVE-IT
- Bempedoic acid CLEAR Outcomes
- Niacin HDL cardiovascular
- Familial hypercholesterolemia treatment
- Statin myopathy intolerance
- Mediterranean diet LDL cholesterol
- Plant sterols cholesterol lowering
- Soluble fiber LDL reduction
Connections
- Atherosclerosis
- ApoB
- Lipid Panel
- Coronary Calcium Score
- Lipoprotein(a)
- Cardiovascular Disease
- Coronary Artery Disease
- Hypertension
- Stroke
- Diabetes
- Metabolic Syndrome
- Insulin Resistance
- Berberine
- Garlic
- Omega-3 Fatty Acids
- Magnesium
- Anti-Inflammatory Diet
- Olive Oil