Folate and Pregnancy: Neural Tube Defect Prevention
The discovery that folic acid supplementation prevents neural tube defects (NTDs) stands as one of the most significant achievements in preventive medicine. Since the landmark Medical Research Council Vitamin Study of 1991 demonstrated a 72% reduction in NTD recurrence with periconceptional folic acid supplementation, public health initiatives worldwide have dramatically reduced the incidence of these devastating birth defects. Understanding the biological mechanisms, clinical guidelines, and ongoing debates surrounding folate in pregnancy is essential for healthcare providers and expectant mothers alike.
Table of Contents
- Key Benefits at a Glance
- Neural Tube Closure (Days 21-28)
- Spina Bifida and Anencephaly
- Folic Acid Fortification History
- Preconception Supplementation Timing
- MTHFR Polymorphisms and Methylfolate
- Recommended Doses (400-800 mcg)
- High-Risk Pregnancies (4 mg)
- Folic Acid vs. Methylfolate Debate
- Placental Development
- Preterm Birth Risk Reduction
- Clinical Guidelines
- Research Papers
- Connections
- Featured Videos
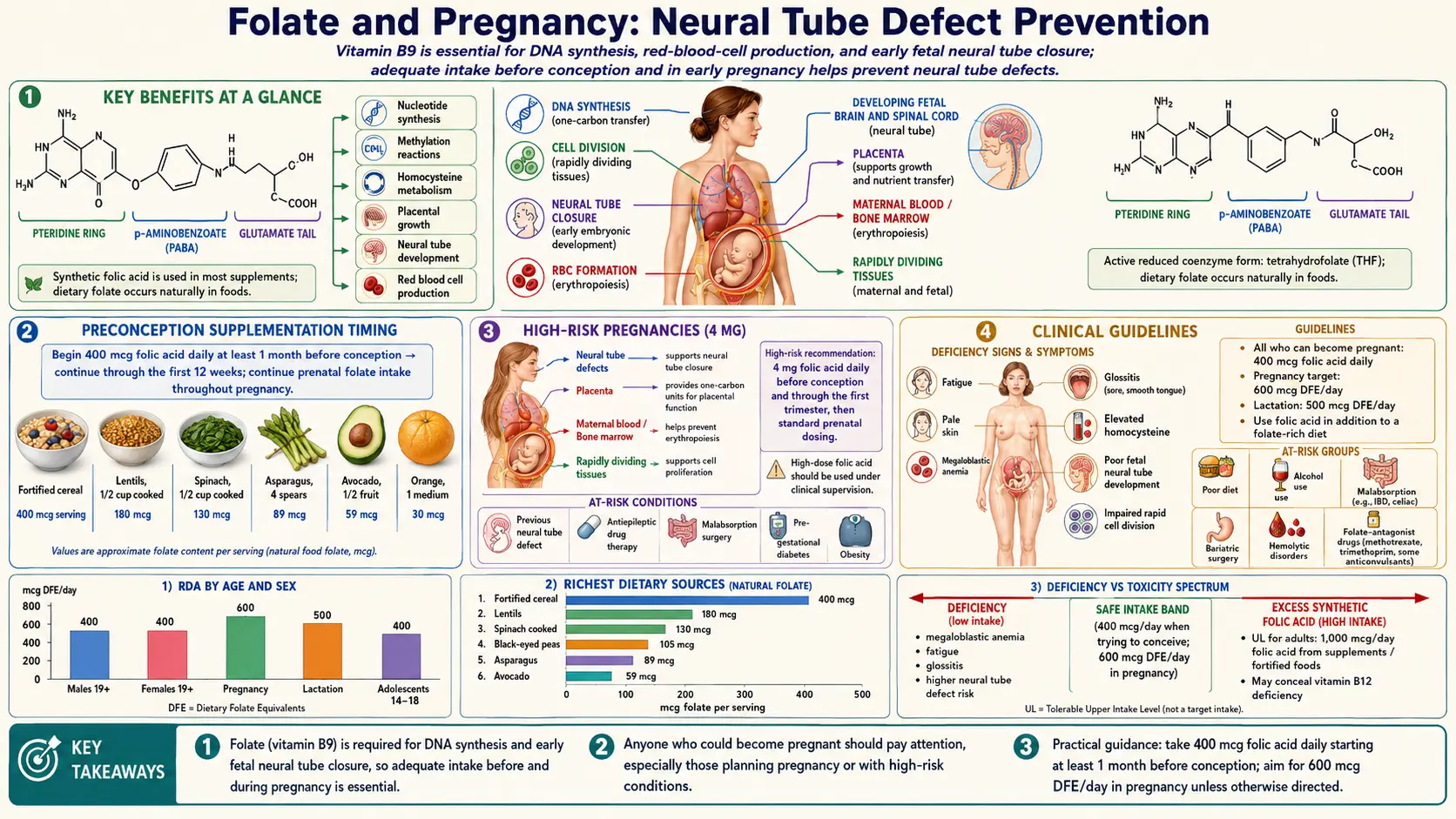
Key Benefits at a Glance
- Up to 72% reduction in NTD recurrence with periconceptional folic acid (MRC Vitamin Study, 1991).
- Prevents spina bifida and anencephaly when adequate stores are present during days 21–28 post-conception.
- Grain fortification (1998 U.S. mandate) reduced NTD prevalence by ~28%; Canada saw a 46% drop.
- 400–800 mcg/day for all women of reproductive age; 4 mg/day for high-risk pregnancies.
- Supports placental angiogenesis and trophoblast proliferation.
- 40–60% lower preterm birth risk with adequate preconception folate status.
- RBC folate >906 nmol/L associated with lowest NTD risk.
- Reduces homocysteine, supporting maternal cardiovascular and placental health.
Neural Tube Closure (Days 21-28)
The neural tube is the embryonic precursor of the brain and spinal cord, and its formation is one of the earliest and most critical events in human development.
- Neurulation process: Neural tube closure begins around day 21 of embryonic development (approximately 5 weeks gestational age) and is completed by day 28. During this week-long process, the flat neural plate folds inward and fuses along the midline to form a hollow tube. Closure occurs at multiple initiation sites simultaneously and proceeds bidirectionally, creating a zipper-like fusion of the neural folds.
- Closure sites: In humans, neural tube closure is initiated at multiple discrete sites. Closure site 1 begins in the cervical region and proceeds both cranially and caudally. Additional closure sites at the cranial end facilitate closure of the developing brain. Failure of closure at different sites produces distinct clinical phenotypes.
- Cellular requirements: Successful neural tube closure requires precise coordination of cell proliferation, cell shape changes (apical constriction), cell migration, and cell adhesion. Each of these processes demands rapid DNA synthesis and methylation reactions for which folate is an essential cofactor.
- Critical timing: Because neural tube closure is complete by day 28 post-conception, often before a woman even knows she is pregnant, the window for folate's protective effect is extremely narrow. This timing underscores the critical importance of preconception supplementation and population-wide food fortification strategies.
Spina Bifida and Anencephaly
Neural tube defects represent a spectrum of malformations resulting from incomplete neural tube closure, with spina bifida and anencephaly being the most common.
- Spina bifida: Failure of caudal neural tube closure results in spina bifida, which ranges in severity from the mild spina bifida occulta (a defect in the vertebral arch without herniation of neural tissue) to the severe myelomeningocele (protrusion of the spinal cord and meninges through a vertebral defect). Myelomeningocele causes lifelong disability including paralysis below the level of the defect, bowel and bladder dysfunction, hydrocephalus, and Chiari II malformation.
- Anencephaly: Failure of cranial neural tube closure produces anencephaly, in which the forebrain and calvarium fail to develop. Anencephaly is uniformly fatal, with most affected infants stillborn or surviving only hours to days after birth. It occurs in approximately 1 in 4,600 pregnancies in the United States following fortification.
- Encephalocele: This less common NTD involves herniation of brain tissue and meninges through a defect in the skull, most commonly in the occipital region. Outcomes depend on the size and content of the herniation.
- Global prevalence: Prior to folic acid fortification, NTD prevalence ranged from approximately 1 to 6 per 1,000 births, with significant geographic and ethnic variation. Rates were historically highest in parts of China, the British Isles, and among certain populations in Central America. Fortification has reduced prevalence by 25 to 50% in countries that have implemented mandatory programs.
Folic Acid Fortification History
The implementation of mandatory folic acid fortification represents a landmark public health intervention with demonstrable population-level impact.
- United States mandate: In 1998, the United States Food and Drug Administration mandated the addition of 140 mcg of folic acid per 100 grams to all enriched cereal grain products (flour, bread, pasta, rice, cornmeal). This policy was estimated to provide an average of 100 to 200 mcg of additional folic acid daily to women of reproductive age.
- Impact on NTD rates: Following fortification in the U.S., NTD prevalence decreased by approximately 28%, from 10.7 per 10,000 live births to 7.0 per 10,000. Similar reductions were observed in Canada (46% reduction), Chile (40%), and other countries with mandatory fortification programs.
- Global adoption: As of 2024, over 80 countries have implemented mandatory folic acid fortification of staple foods. However, many European countries have declined mandatory fortification due to concerns about potential adverse effects of excessive folic acid intake in the general population, relying instead on voluntary fortification and supplementation recommendations.
- Remaining disparities: Despite fortification, NTD rates remain higher in certain populations, including Hispanic women, women with lower socioeconomic status, and women with obesity. These disparities suggest that additional factors beyond dietary folate intake contribute to NTD risk.
Preconception Supplementation Timing
The timing of folate supplementation relative to conception is the most critical factor determining its effectiveness in NTD prevention.
- Optimal timing: Folic acid supplementation should ideally begin at least one month before conception and continue through the first 12 weeks of pregnancy (first trimester). This ensures that maternal folate stores are replete before the critical period of neural tube closure.
- Red blood cell folate: While serum folate levels respond rapidly to supplementation (within days), red blood cell (RBC) folate, which better reflects tissue folate stores, requires 8 to 14 weeks to reach optimal concentrations. RBC folate levels above 906 nmol/L (400 ng/mL) are associated with the lowest NTD risk.
- Unplanned pregnancies: Approximately 45% of pregnancies in the United States are unplanned, meaning that many women do not begin supplementation before conception. This reality is the primary justification for population-wide food fortification, which provides a baseline level of folic acid to all women of reproductive age regardless of their pregnancy planning status.
- Continuous supplementation recommendation: Major health organizations recommend that all women capable of becoming pregnant consume 400 mcg of folic acid daily, whether or not they are planning a pregnancy, to ensure adequate folate status at the time of conception.
MTHFR Polymorphisms and Methylfolate
Genetic variations in folate metabolism, particularly in the MTHFR gene, have emerged as important modifiers of NTD risk and response to folic acid supplementation.
- MTHFR C677T variant: The methylenetetrahydrofolate reductase (MTHFR) C677T polymorphism is the most clinically significant genetic variant in folate metabolism. The TT genotype, present in 10 to 15% of North American and European populations (and up to 25% in certain Mexican and Italian populations), results in a thermolabile enzyme with approximately 30% of normal activity. This leads to reduced conversion of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate, the primary circulating form of folate.
- NTD risk modification: Women with the MTHFR 677TT genotype have a 2 to 3-fold increased risk of having an NTD-affected pregnancy. When both mother and fetus carry the TT genotype, the risk may be even higher. This genetic risk is partially mitigated by adequate folate intake, making supplementation especially important for carriers.
- MTHFR A1298C variant: This second common MTHFR polymorphism reduces enzyme activity to a lesser degree than C677T. Compound heterozygosity (C677T/A1298C) produces an intermediate phenotype with moderately reduced MTHFR activity.
- Methylfolate consideration: Some practitioners advocate for the use of L-methylfolate (5-MTHF) rather than folic acid in women with MTHFR polymorphisms, arguing that it bypasses the impaired enzymatic step. While biochemically logical, clinical evidence demonstrating superior NTD prevention with methylfolate compared to folic acid is currently lacking.
Recommended Doses (400-800 mcg)
- Standard recommendation: The U.S. Preventive Services Task Force, American College of Obstetricians and Gynecologists (ACOG), and World Health Organization recommend 400 to 800 mcg of folic acid daily for all women of reproductive age to prevent NTDs. Most prenatal vitamins contain 800 to 1,000 mcg.
- Dietary folate equivalents (DFE): Due to differences in bioavailability, 1 mcg of food folate equals 1 DFE, while 1 mcg of supplemental folic acid taken with food equals 1.7 DFE. The recommended dietary allowance (RDA) for pregnant women is 600 mcg DFE per day.
- Upper limit considerations: The tolerable upper intake level (UL) for folic acid from supplements and fortified foods is set at 1,000 mcg (1 mg) per day for adults. This limit was established primarily based on concerns about masking vitamin B12 deficiency. Exceeding the UL is not recommended except under medical supervision for high-risk pregnancies.
High-Risk Pregnancies (4 mg)
Certain women at elevated NTD risk require substantially higher folic acid doses than the standard recommendation.
- Previous NTD-affected pregnancy: Women who have had a prior pregnancy affected by an NTD have a recurrence risk of 2 to 5%, approximately 10 to 20 times the general population risk. These women are recommended to take 4 mg (4,000 mcg) of folic acid daily, beginning at least one month before conception and continuing through the first trimester.
- Antiepileptic drug use: Women taking valproic acid or carbamazepine have a significantly elevated NTD risk (1 to 2% with valproic acid). The 4 mg dose is recommended for these patients, though the evidence that high-dose folic acid can fully overcome the teratogenic effect of these medications is limited.
- Diabetes mellitus: Pre-gestational diabetes (both type 1 and type 2) is associated with a 2 to 10-fold increased risk of NTDs. Many guidelines recommend the 4 mg high-dose regimen for diabetic women planning pregnancy.
- Obesity: Maternal obesity (BMI greater than 30) is an independent risk factor for NTDs, with risk increasing proportionally with BMI. Some guidelines recommend higher folic acid doses for obese women, though the optimal dose has not been established.
- Family history: Women with a first-degree relative (parent or sibling) who was affected by an NTD, or whose partner has an NTD or previous NTD-affected offspring, may be considered for the 4 mg high-dose regimen.
Folic Acid vs. Methylfolate Debate
A growing controversy in prenatal nutrition concerns whether L-methylfolate should replace folic acid as the recommended supplement form.
- Arguments for methylfolate: Proponents argue that L-methylfolate is the biologically active form that directly enters the folate cycle, bypasses potentially impaired MTHFR enzyme activity, does not produce unmetabolized folic acid (UMFA) in the circulation, and may be more effective in women with MTHFR polymorphisms.
- Arguments for folic acid: The NTD prevention evidence base is built entirely on folic acid, not methylfolate. Folic acid is inexpensive, highly stable, has well-characterized pharmacokinetics, and has decades of safety data in pregnancy. No randomized controlled trial has demonstrated NTD prevention with methylfolate supplementation.
- Unmetabolized folic acid (UMFA): When folic acid intake exceeds the capacity of dihydrofolate reductase (approximately 200-400 mcg in a single dose), unmetabolized folic acid appears in the circulation. Concerns have been raised about potential adverse effects of chronic UMFA exposure, including impaired natural killer cell function and acceleration of pre-existing pre-neoplastic lesions. However, the clinical significance of these findings remains debated.
- Current consensus: Most major medical organizations continue to recommend folic acid for NTD prevention, noting the lack of clinical trial evidence for methylfolate in this indication. However, some professional guidelines acknowledge methylfolate as an acceptable alternative, particularly for women with documented MTHFR polymorphisms who demonstrate inadequate response to folic acid.
Placental Development
- Trophoblast proliferation: Folate is essential for the rapid cell division that characterizes early placental development. The cytotrophoblast, the proliferative compartment of the placenta, has high folate requirements for DNA synthesis and methylation during the first trimester.
- Placental vascular development: Adequate folate status supports proper angiogenesis and vasculogenesis within the developing placenta. Folate deficiency may impair the formation of the villous vascular network, potentially contributing to placental insufficiency and adverse pregnancy outcomes including preeclampsia and intrauterine growth restriction.
- Folate transport: The placenta actively concentrates folate from maternal to fetal circulation via folate receptor alpha (FRa) and the proton-coupled folate transporter (PCFT). Maternal folate levels must be adequate to maintain the concentration gradient that drives transplacental folate transfer.
Preterm Birth Risk Reduction
- Epidemiological evidence: Multiple observational studies and meta-analyses have reported that folic acid supplementation, particularly when begun before conception and continued through pregnancy, is associated with a 40 to 60% reduction in preterm birth (delivery before 37 weeks of gestation).
- Dose-response relationship: Some studies suggest a dose-response effect, with longer duration of supplementation before conception associated with greater preterm birth risk reduction. Women who supplemented for more than one year before conception showed the greatest benefit in certain analyses.
- Proposed mechanisms: Folate may reduce preterm birth risk through multiple mechanisms, including improved placental implantation, reduced homocysteine-mediated endothelial damage, enhanced DNA repair capacity, and reduced systemic inflammation.
Clinical Guidelines
- Universal recommendation: All women of reproductive age should consume 400 to 800 mcg of folic acid daily from supplements or fortified foods, in addition to consuming food sources of folate from a varied diet.
- Prenatal vitamins: Once pregnancy is confirmed, women should continue folic acid supplementation as part of a comprehensive prenatal vitamin throughout the entire pregnancy and into the postpartum period, particularly if breastfeeding.
- Genetic screening: Routine MTHFR genotyping is not currently recommended by ACOG or the American College of Medical Genetics, as the clinical utility of this information for modifying NTD prevention strategies is not established. However, targeted testing may be considered in women with recurrent NTD-affected pregnancies or other indications.
- Monitoring: Serum folate and RBC folate levels can be measured to assess folate status, but routine monitoring is not recommended in uncomplicated pregnancies when adequate supplementation is confirmed. Monitoring may be warranted in high-risk populations or when compliance is uncertain.
- Counseling: Healthcare providers should counsel all women of reproductive age about the importance of adequate folate intake, ideally before conception, and document this counseling in the medical record.
Summary
Folate's role in preventing neural tube defects is one of the best-established nutrient-disease relationships in medicine. Supplementation with 400 to 800 mcg of folic acid before conception and during early pregnancy reduces NTD risk by up to 72%. Women at high risk should receive 4 mg daily. While the folic acid versus methylfolate debate continues, clinical evidence overwhelmingly supports folic acid for NTD prevention. Preconception planning and population-wide fortification remain essential strategies to ensure adequate folate status during the critical window of neural tube closure.
Research Papers
- MRC Vitamin Study Research Group. Prevention of neural tube defects: results of the Medical Research Council Vitamin Study. Lancet. 1991;338(8760):131-137.
- Czeizel AE, Dudas I. Prevention of the first occurrence of neural-tube defects by periconceptional vitamin supplementation. N Engl J Med. 1992;327(26):1832-1835.
- PubMed — Folic acid fortification and NTD prevalence (topic search)
- PubMed — MTHFR C677T and NTD risk (topic search)
- PubMed — RBC folate threshold and NTD risk (topic search)
- PubMed — Unmetabolized folic acid in pregnancy (topic search)
- PubMed — Folic acid and preterm birth (topic search)
- PubMed — Valproate, folic acid, and NTDs (topic search)
- NIH Office of Dietary Supplements — Folate Fact Sheet for Health Professionals
- Linus Pauling Institute — Folate
- CDC — Folic Acid
- Harvard T.H. Chan School of Public Health — Folate (Folic Acid)
Connections
- Vitamin B9
- Folate and Mental Health
- Vitamin B12
- Vitamin B6
- Choline
- Vitamin B6 and Homocysteine
- Iron
- Vitamin D3
- Anemia
- Homocysteine
- Preeclampsia
- Gestational Diabetes
- B12 Deficiency Diagnosis