Aspirin Side Effects, Risks & Contraindications
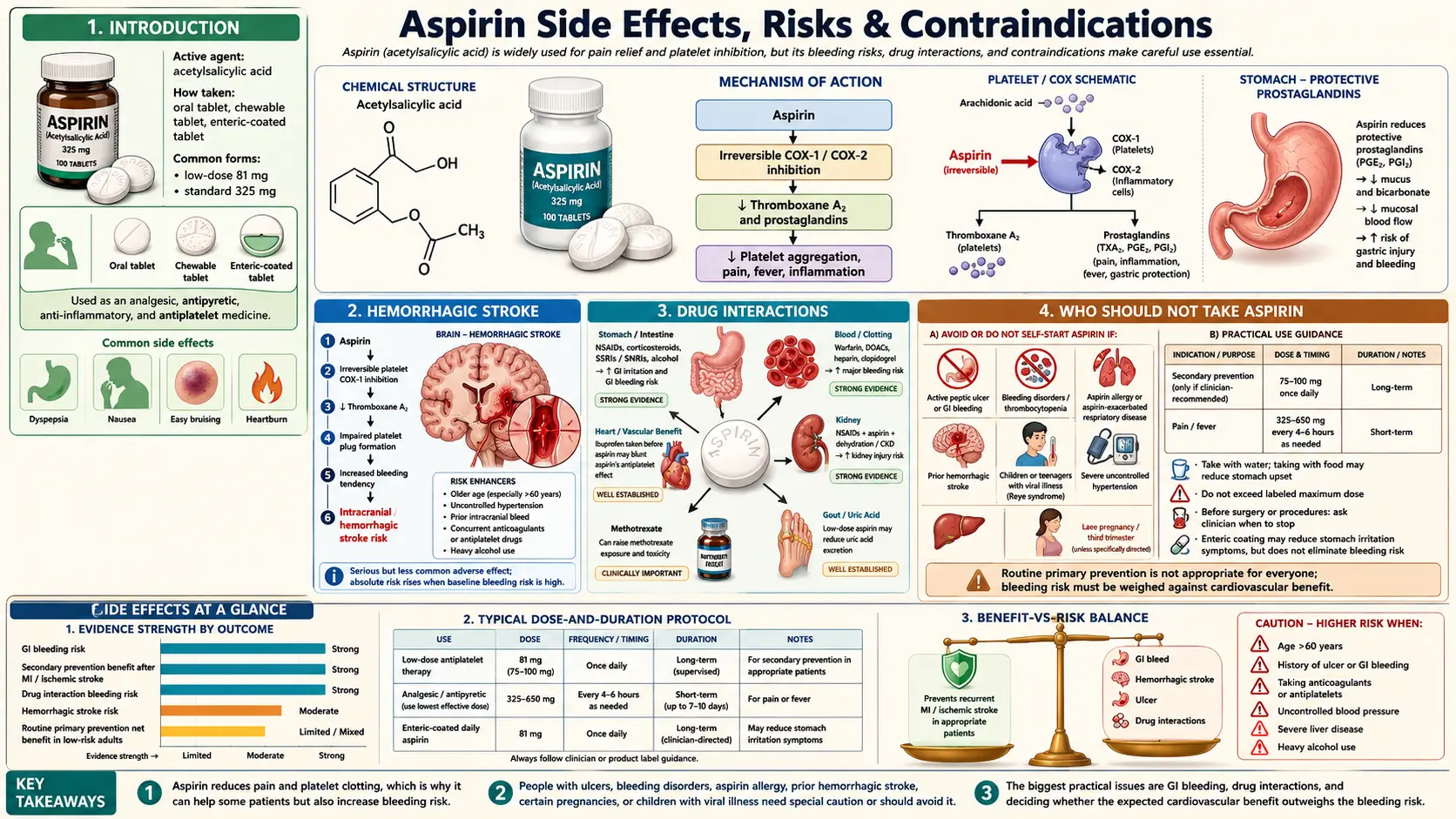
Aspirin (acetylsalicylic acid) is among the most extensively studied drugs in medical history, with over a century of clinical use and tens of thousands of randomized-trial participants contributing to its evidence base. That long track record, however, does not make it a benign medication. Even at low "cardioprotective" doses of 75–100 mg daily, aspirin carries real risks of gastrointestinal bleeding, intracranial hemorrhage, and a range of less common but serious adverse effects. A balanced understanding of these harms — and of the populations in whom they outweigh the benefits — is essential for any rational decision about starting, continuing, or stopping aspirin.
Table of Contents
- Introduction
- Gastrointestinal Bleeding
- Peptic Ulcer Disease
- PPI Co-Prescription
- Hemorrhagic Stroke
- Reye's Syndrome
- Tinnitus & Hearing Loss
- Aspirin-Exacerbated Respiratory Disease (Samter's Triad)
- Aspirin Allergy & Hypersensitivity
- Drug Interactions
- Pregnancy Considerations
- Surgery & Dental Procedures
- Aspirin Overdose & Salicylate Toxicity
- Who Should Not Take Aspirin
- References & Research Papers
- Connections
- Featured Videos
Introduction
Aspirin's therapeutic effects — analgesia, antipyresis, anti-inflammatory activity, and irreversible inhibition of platelet cyclooxygenase-1 (COX-1) — are inseparable from its adverse effects. The same mechanism that makes aspirin a powerful antiplatelet agent (suppressing thromboxane A2 production for the 7–10 day lifespan of the platelet) also impairs hemostasis throughout the body. The same COX-1 inhibition that reduces prostaglandin-mediated pain and inflammation also strips away the gastroduodenal mucosal defenses those prostaglandins normally maintain.
For primary prevention in low-risk adults, contemporary meta-analyses have increasingly suggested that the bleeding harm offsets or exceeds the cardiovascular benefit — a conclusion that prompted the 2022 U.S. Preventive Services Task Force to recommend against routine initiation of low-dose aspirin for primary prevention in adults aged 60 and older. For secondary prevention (after a prior myocardial infarction, ischemic stroke, or coronary revascularization), the benefit-to-risk ratio remains favorable for most patients, but the adverse effects below still apply and must be monitored.
Gastrointestinal Bleeding
Gastrointestinal (GI) bleeding is the most common serious adverse effect of aspirin and the principal reason for discontinuation. It occurs through two complementary mechanisms:
- Systemic COX-1 inhibition depletes cytoprotective prostaglandins (PGE2, PGI2) that normally maintain gastric mucus secretion, bicarbonate production, and mucosal blood flow. This injury occurs regardless of the route of administration — enteric-coated, buffered, and even rectal or intravenous formulations all produce it.
- Topical mucosal damage from the acidic aspirin tablet dissolving in contact with the gastric epithelium causes direct chemical injury and impairs the epithelial barrier. This component is partially mitigated by enteric coating and by taking aspirin with food, but it does not eliminate bleeding risk.
Incidence
Pooled data from randomized trials suggest that low-dose aspirin (75–100 mg daily) approximately doubles the risk of major upper GI bleeding compared with placebo. Absolute risk varies with baseline characteristics but is in the range of 0.5–1.0 additional major GI bleeds per 1,000 person-years in the general primary-prevention population, rising substantially in older adults and those with other risk factors. The 2018 ASPREE trial in adults aged 70 and older found a hazard ratio for major hemorrhage of 1.38 compared with placebo, with the majority of that excess arising from the GI tract.
Low-Dose vs High-Dose
- The relationship between dose and GI bleeding is approximately linear in the 75–325 mg range; doses above 100 mg daily confer no additional cardiovascular benefit but do increase bleeding.
- Anti-inflammatory doses (1–4 g daily, as historically used for rheumatologic conditions) produce substantially higher rates of gastroduodenal injury, with endoscopic ulcer prevalence of 10–30% in chronic users.
- Enteric-coated and buffered preparations reduce endoscopic lesions but have not been shown in large trials to meaningfully reduce clinically significant bleeding.
Peptic Ulcer Disease
Aspirin is a well-established cause of peptic ulcers, including ulcer complications (perforation and bleeding). The relative risk of uncomplicated peptic ulcer disease in chronic aspirin users is approximately 2–4 times that of non-users, and higher still when combined with other NSAIDs or anticoagulants.
Risk Factors That Amplify Aspirin-Induced Ulcer Risk
- Age >65, particularly >75
- Prior history of peptic ulcer disease or GI bleeding (the single strongest predictor)
- Concurrent use of another NSAID, a corticosteroid, an anticoagulant, or an SSRI
- High-dose aspirin or multiple antiplatelet therapy (e.g., aspirin plus clopidogrel)
- Heavy alcohol use and smoking
- Helicobacter pylori infection
Helicobacter pylori Co-Infection
The interaction between H. pylori infection and aspirin use is more than additive. In patients with a prior history of ulcer bleeding who test positive for H. pylori, eradication of the organism with triple therapy markedly reduces the rate of recurrent ulcer bleeding on aspirin — to levels comparable with aspirin users who never had H. pylori. Current guidelines therefore recommend H. pylori testing and eradication before starting long-term aspirin in any patient with a prior ulcer history.
PPI Co-Prescription
Proton pump inhibitors (PPIs) such as omeprazole, pantoprazole, and esomeprazole markedly reduce the incidence of aspirin-induced upper GI injury by suppressing gastric acid secretion and allowing mucosal healing. The landmark OBERON and related trials demonstrated that PPI co-therapy reduces endoscopic gastric and duodenal ulcers in chronic low-dose aspirin users by approximately 70–80%.
When PPI Co-Therapy Is Recommended
- Prior history of peptic ulcer or upper GI bleeding — a clear indication; PPIs are strongly recommended alongside aspirin in this population.
- Age ≥ 65 combined with one additional risk factor (concurrent NSAID, corticosteroid, anticoagulant, SSRI, or dyspepsia).
- Dual antiplatelet therapy (aspirin plus clopidogrel, ticagrelor, or prasugrel) — routinely recommended in the United States, particularly for the first 12 months after coronary stenting.
- Concurrent anticoagulation (warfarin or a direct oral anticoagulant) with aspirin for a specific indication.
PPI therapy itself carries potential long-term considerations (hypomagnesemia, possible increased risk of Clostridioides difficile, modest effects on bone density, and pharmacokinetic interactions with clopidogrel through CYP2C19). These trade-offs are generally outweighed by the reduction in bleeding in patients who genuinely need long-term aspirin.
Hemorrhagic Stroke
By impairing platelet aggregation, aspirin increases the risk of intracranial hemorrhage (ICH), including hemorrhagic stroke and chronic subdural hematoma. Although the absolute risk is small, it is one of the most feared adverse effects because of the high mortality and disability associated with intracerebral bleeding.
Quantifying the Excess Risk
- Meta-analyses of primary-prevention trials indicate that low-dose aspirin increases hemorrhagic stroke risk by roughly 30–40% in relative terms.
- In absolute terms, this corresponds to approximately 1–2 additional hemorrhagic strokes per 10,000 person-years in middle-aged adults — rising with age and with the presence of uncontrolled hypertension.
- In the ASPREE trial in healthy adults aged ≥70, the rate of intracranial bleeding was 8.6 vs 5.0 events per 1,000 person-years (aspirin vs placebo).
- Aspirin also increases the risk of hemorrhagic transformation of ischemic stroke and of spontaneous subdural hematoma, particularly in older adults prone to falls.
For secondary prevention after a prior ischemic stroke or TIA, the absolute reduction in recurrent ischemic events generally exceeds the absolute increase in intracranial hemorrhage, so aspirin remains indicated. For primary prevention, the calculus is much less favorable, which is a major driver of the current movement away from routine primary-prevention aspirin in older adults.
Reye's Syndrome
Reye's syndrome is a rare but frequently fatal condition characterized by acute non-inflammatory encephalopathy and fatty degeneration of the liver, occurring almost exclusively in children and adolescents recovering from a viral illness — most classically influenza or varicella (chickenpox). The association with aspirin is one of the strongest and most consequential pharmacoepidemiologic findings of the 20th century.
Clinical Features
- Biphasic illness: a typical viral prodrome (fever, upper respiratory symptoms, or varicella), apparent recovery, then abrupt onset of intractable vomiting and progressive neurologic deterioration — lethargy, confusion, agitation, seizures, and coma.
- Laboratory findings: marked elevation of liver transaminases (AST/ALT) without significant jaundice or hyperbilirubinemia, hyperammonemia, prolonged prothrombin time, and hypoglycemia.
- Histopathology: microvesicular steatosis of hepatocytes with swollen mitochondria — the pathologic signature of mitochondrial injury.
- Mortality historically ranged from 30% to over 50%; survivors may suffer permanent neurologic sequelae.
Why Aspirin Is Contraindicated Under Age ~16 in Febrile Viral Illness
Case-control studies in the early 1980s — most notably the Centers for Disease Control pilot study and the U.S. Public Health Service study — found that children with Reye's syndrome were far more likely to have received aspirin during their preceding viral illness than matched controls, with odds ratios typically in the range of 20–40. The proposed mechanism involves salicylate-induced impairment of mitochondrial beta-oxidation of fatty acids and disruption of the urea cycle, producing the characteristic hepatic steatosis and hyperammonemia.
Following the public health warnings issued between 1980 and 1982, and the FDA's 1986 mandate that aspirin-containing products carry a Reye's syndrome warning, the incidence of Reye's syndrome in the United States fell dramatically — from several hundred cases per year in the early 1980s to fewer than two per year by the late 1990s. This decline is one of the most compelling demonstrations in pharmacovigilance that a population-wide change in medication use can eliminate a serious disease.
FDA Labeling History
- 1980: CDC and state health departments issue the first warnings linking aspirin to Reye's syndrome.
- 1982: U.S. Surgeon General advisory against aspirin for children with viral illnesses.
- 1986: FDA requires a Reye's syndrome warning on all aspirin-containing OTC products.
- 2003: FDA extends the warning to include children and teenagers up to age 19, reflecting documented cases in adolescents.
In current pediatric practice, aspirin should generally not be given to anyone under the age of approximately 16 with a febrile illness of possible viral origin. Acetaminophen or ibuprofen are preferred antipyretics. The principal exceptions — where pediatric aspirin is used under specialist supervision — are Kawasaki disease and selected rheumatologic or post-cardiac-surgery indications; in these cases, the clinician weighs the risk of Reye's syndrome against the specific indication, and families are counseled to withhold aspirin and seek guidance at the first sign of influenza or varicella.
Tinnitus & Hearing Loss
Salicylates have been recognized as ototoxic for more than a century. Aspirin-induced auditory effects are strongly dose-related and, in most cases, fully reversible once the drug is withdrawn.
- Tinnitus (typically bilateral, high-pitched ringing) is among the earliest clinical signs of salicylate toxicity. It appears in a small percentage of patients at doses of 1–2 g daily and becomes common at doses of 3–4 g daily.
- Sensorineural hearing loss, usually symmetric and affecting higher frequencies first, accompanies tinnitus in more pronounced cases. Hearing typically returns to baseline within 24–72 hours of discontinuation.
- The underlying mechanism involves salicylate binding to the outer hair cell motor protein prestin, impairing cochlear amplification, together with effects on cochlear blood flow.
- At the low doses used for cardiovascular prevention (75–100 mg daily), tinnitus is uncommon; when it does occur at low doses it may signal underlying hypersensitivity or drug accumulation from impaired renal clearance.
Persistent tinnitus on low-dose aspirin warrants evaluation for an alternative cause and consideration of discontinuation.
Aspirin-Exacerbated Respiratory Disease (Samter's Triad)
Aspirin-exacerbated respiratory disease (AERD), also known as Samter's triad or NSAID-exacerbated respiratory disease (N-ERD), is a distinct clinical syndrome affecting approximately 7–10% of adults with asthma and up to 30–40% of those with severe asthma and nasal polyposis.
The Classic Triad
- Asthma, often late-onset, typically severe and difficult to control
- Chronic rhinosinusitis with nasal polyps, frequently recurrent despite surgery
- Respiratory hypersensitivity reactions to aspirin and other COX-1 inhibitors — characteristically bronchospasm, rhinorrhea, and nasal congestion developing within 30–180 minutes of ingestion
Pathophysiology
AERD is not a classical IgE-mediated allergy. It is a pharmacologic reaction driven by dysregulation of arachidonic acid metabolism: COX-1 inhibition diverts substrate toward the 5-lipoxygenase pathway, producing cysteinyl leukotrienes (LTC4, LTD4, LTE4) that drive bronchoconstriction, mucus secretion, and eosinophilic inflammation. Patients show abnormally high baseline urinary LTE4 and a dramatic further increase after aspirin challenge.
Because the reaction is pharmacologic rather than immunologic, all COX-1 inhibitors — ibuprofen, naproxen, ketorolac, and others — will cross-react. Selective COX-2 inhibitors (celecoxib) and acetaminophen at low-to-moderate doses are generally tolerated. Patients with AERD who require aspirin for cardiovascular indications can undergo aspirin desensitization under allergist supervision, which also has therapeutic benefit for the underlying airway disease.
Aspirin Allergy & Hypersensitivity
"Aspirin allergy" is a clinically imprecise label that encompasses several distinct mechanisms. Careful phenotyping matters, because management and the degree of cross-reactivity with other NSAIDs differ substantially.
True IgE-Mediated Hypersensitivity
- Classical immediate-type allergy mediated by aspirin-specific IgE antibodies.
- Manifestations: urticaria, angioedema, bronchospasm, and in severe cases anaphylaxis, occurring within minutes to an hour of exposure.
- Typically reaction is to aspirin specifically, not all NSAIDs — structurally unrelated NSAIDs are often tolerated.
- Rare; confirmation usually requires specialist evaluation.
Pseudo-Allergy (Pharmacologic Hypersensitivity)
- Not antibody-mediated. Driven by COX-1 inhibition and leukotriene shunting.
- Includes AERD (above), NSAID-induced urticaria/angioedema in patients with chronic spontaneous urticaria, and blended respiratory/cutaneous reactions.
- Cross-reactivity with other COX-1 inhibitors is the rule; selective COX-2 inhibitors and acetaminophen are usually tolerated.
Desensitization Protocols
For patients with AERD or with prior urticarial reactions who require aspirin for cardiovascular indications, supervised desensitization induces a temporary state of tolerance. Standard protocols administer escalating oral doses (typically starting at 20–40 mg and doubling every 90–180 minutes up to 325 mg) in a monitored setting with resuscitation equipment available. Once tolerance is established, it must be maintained by uninterrupted daily dosing — a lapse of more than 48 hours generally requires repeat desensitization.
Drug Interactions
Aspirin interacts with numerous drug classes. The most clinically important interactions involve additive bleeding risk, renal hemodynamic effects, and reduced renal clearance of narrow-therapeutic-index drugs.
Warfarin
- Aspirin adds platelet inhibition to warfarin's inhibition of coagulation factors; the combination substantially increases major bleeding, particularly intracranial and GI.
- Combination is appropriate in specific indications (e.g., mechanical heart valve with coronary disease) but requires careful risk-benefit assessment and tight INR control.
Direct Oral Anticoagulants (DOACs)
- Apixaban, rivaroxaban, dabigatran, and edoxaban all show meaningfully increased bleeding when combined with aspirin.
- In atrial fibrillation without recent coronary stenting, routine addition of aspirin to a DOAC offers no ischemic benefit but clearly increases bleeding — and should generally be avoided.
SSRIs and SNRIs
- Serotonin reuptake inhibitors deplete platelet serotonin and impair platelet activation, producing an antiplatelet effect of their own.
- Addition of aspirin (or an NSAID) to an SSRI roughly triples the risk of upper GI bleeding compared with either agent alone.
- When the combination is necessary, PPI co-therapy should be strongly considered.
Methotrexate
- Salicylates displace methotrexate from plasma protein binding and compete for renal tubular secretion, raising methotrexate levels.
- At the high oncologic doses of methotrexate, this interaction can precipitate severe myelosuppression, mucositis, and nephrotoxicity — a well-recognized and potentially fatal combination.
- At low doses of methotrexate (e.g., weekly 10–25 mg for rheumatoid arthritis) combined with low-dose aspirin, the interaction is usually clinically manageable but warrants monitoring.
ACE Inhibitors / ARBs Plus a Diuretic (the "Triple Whammy")
- The combination of (1) an ACE inhibitor or ARB, (2) a diuretic, and (3) an NSAID (including aspirin at anti-inflammatory doses) is a well-documented cause of acute kidney injury.
- ACEi/ARBs dilate the efferent renal arteriole; diuretics reduce circulating volume; NSAIDs constrict the afferent arteriole via prostaglandin inhibition. Together they can collapse glomerular filtration pressure.
- Low-dose cardiovascular aspirin (75–100 mg) carries far less renal risk than full-dose NSAIDs, but the combination still deserves caution in older adults, in dehydration, and during acute illness.
Corticosteroids
- Systemic corticosteroids alone are not strongly ulcerogenic, but their combination with aspirin or another NSAID markedly increases peptic ulcer and GI bleeding risk (roughly a four-fold increase over NSAID alone).
- When the combination is required, PPI co-therapy is generally indicated.
Other Notable Interactions
- Ibuprofen: regular ibuprofen dosing can block access of aspirin to the platelet COX-1 active site, antagonizing aspirin's cardioprotective effect. If both are needed, aspirin should be taken at least 30 minutes before ibuprofen, or at least 8 hours after.
- Alcohol: heavy intake compounds gastric mucosal injury and bleeding risk.
- Uricosurics (probenecid, sulfinpyrazone): salicylates antagonize their urate-lowering effect.
- Oral hypoglycemics: high-dose salicylates can potentiate hypoglycemia.
Pregnancy Considerations
The role of aspirin in pregnancy depends critically on dose and trimester.
Low-Dose Aspirin (75–162 mg) for Pre-Eclampsia Prevention
- Multiple large randomized trials and the ASPRE trial in high-risk women have shown that low-dose aspirin started before 16 weeks of gestation significantly reduces the incidence of preterm pre-eclampsia.
- Current guidelines from ACOG, USPSTF, and NICE recommend low-dose aspirin (typically 81 mg or 150 mg daily, depending on the guideline) from ~12–16 weeks through ~36 weeks in women at elevated risk of pre-eclampsia.
- Low-dose aspirin has not been associated with increased rates of congenital malformations, bleeding, or other adverse outcomes in this context.
High-Dose Aspirin in the Third Trimester — Contraindicated
- Full anti-inflammatory doses of aspirin (and other non-selective NSAIDs) are contraindicated from approximately 30 weeks of gestation onward.
- Risks include premature closure of the fetal ductus arteriosus, pulmonary hypertension of the newborn, oligohydramnios from fetal renal effects, and increased maternal and neonatal bleeding at delivery.
- The FDA in 2020 extended its NSAID warning to 20 weeks of gestation and beyond, citing fetal renal dysfunction and oligohydramnios concerns at lower gestational ages than previously thought.
Breastfeeding
- Small amounts of salicylate are excreted in breast milk. Occasional low-dose aspirin is generally regarded as compatible with breastfeeding.
- Chronic high-dose use should be avoided because of theoretical risk of Reye's syndrome in a nursing infant who develops a viral illness.
Surgery & Dental Procedures
Because aspirin irreversibly acetylates platelet COX-1, its antiplatelet effect persists for the 7–10-day lifespan of the exposed platelets. Historical practice was to hold aspirin for 7–10 days before any surgical or dental procedure; contemporary guidance is considerably more nuanced.
Continuing Low-Dose Aspirin Perioperatively
- For patients on aspirin for secondary cardiovascular prevention — prior myocardial infarction, prior ischemic stroke, coronary stents, or established coronary disease — current cardiology and surgical guidelines generally favor continuing low-dose aspirin through most non-cardiac surgeries and most dental procedures.
- The POISE-2 trial found no cardiovascular benefit but a modest increase in major bleeding when aspirin was continued perioperatively in a broad non-cardiac surgery population — but subsequent analyses and guideline bodies still favor continuation in clearly secondary-prevention patients, where the risk of stent thrombosis or recurrent MI off aspirin is substantial.
- Dental extractions, endoscopy with biopsy, cataract surgery, and most dermatologic procedures are generally safe to perform on uninterrupted low-dose aspirin.
Holding Aspirin
- Neurosurgery, posterior eye surgery, and some urologic procedures — where even small amounts of bleeding in a closed compartment can be catastrophic — remain indications for holding aspirin, typically for 5–7 days preoperatively.
- For patients taking aspirin solely for primary prevention, holding it for 5–7 days before elective surgery is reasonable because the acute cardiovascular benefit of uninterrupted therapy is small.
- Coronary artery bypass grafting is a special case: aspirin is often continued up to the day of surgery in stable patients to reduce graft occlusion.
Decisions should be individualized in consultation with the prescriber, particularly in patients with recent coronary stenting — where premature discontinuation of antiplatelet therapy is a major risk factor for stent thrombosis.
Aspirin Overdose & Salicylate Toxicity
Acute salicylate poisoning remains a significant cause of toxicologic morbidity and mortality, particularly in the setting of intentional self-harm and inadvertent chronic over-dosing in elderly patients with impaired renal function. Chronic toxicity is often unrecognized on initial presentation and carries higher mortality than acute overdose of comparable serum levels.
Clinical Presentation
- Early: tinnitus, nausea and vomiting, hyperventilation, diaphoresis, mild lethargy.
- Moderate: tachypnea, tachycardia, fever, volume depletion, confusion.
- Severe: coma, seizures, cerebral edema, non-cardiogenic pulmonary edema, and circulatory collapse.
The Mixed Acid-Base Picture
Salicylate toxicity produces a distinctive and classic acid-base disturbance:
- Respiratory alkalosis — salicylate directly stimulates the medullary respiratory center, causing hyperventilation and a primary decrease in PaCO2.
- Metabolic acidosis — salicylate uncouples mitochondrial oxidative phosphorylation, driving anaerobic metabolism and accumulation of lactate, ketoacids, and salicylate itself, producing a high-anion-gap metabolic acidosis.
- In adults, the typical arterial blood gas shows a mixed respiratory alkalosis with primary metabolic acidosis; children often progress rapidly to pure metabolic acidosis, which is a more ominous sign.
Management
- Airway protection, aggressive fluid resuscitation, glucose administration (even with "normal" blood glucose, because CNS glucose can be depleted).
- Activated charcoal if presentation is within 1–2 hours of ingestion and airway is protected.
- Serum alkalinization with intravenous sodium bicarbonate — converts salicylate to its ionized form, trapping it outside the CNS and enhancing renal elimination. Urinary pH target >7.5.
- Correction of hypokalemia is essential to achieve urinary alkalinization.
- Hemodialysis is indicated for severe toxicity: altered mental status, renal failure, pulmonary edema, refractory acidosis, or serum salicylate concentrations above approximately 90–100 mg/dL (acute) or 40–60 mg/dL (chronic).
- Intubation should be approached with extreme caution: any fall in minute ventilation after sedation and paralysis can abolish the compensatory hyperventilation and precipitate rapid, fatal acidemia. If intubation is necessary, ventilator settings must match or exceed the patient's prior spontaneous minute ventilation.
Who Should Not Take Aspirin
Aspirin is contraindicated or should be avoided in the following groups:
- Children and adolescents under approximately 16 years of age with a febrile illness or suspected viral infection — due to the risk of Reye's syndrome. Specialist-supervised pediatric indications (e.g., Kawasaki disease) are exceptions.
- Known aspirin or NSAID hypersensitivity, including AERD (Samter's triad), NSAID-induced urticaria/angioedema, and prior anaphylaxis — unless desensitized under allergist supervision.
- Active peptic ulcer disease or active GI bleeding.
- Severe uncontrolled hypertension — which markedly amplifies the risk of hemorrhagic stroke.
- Hemorrhagic stroke or recent intracranial hemorrhage.
- Severe hepatic impairment or severe chronic kidney disease (creatinine clearance below ~10 mL/min, or on dialysis without a specific indication).
- Bleeding disorders such as hemophilia, severe thrombocytopenia (platelets <50 × 109/L), or von Willebrand disease.
- Pregnant women in the third trimester at anti-inflammatory doses (low-dose aspirin for pre-eclampsia prevention has a separate, evidence-based role).
- Glucose-6-phosphate dehydrogenase (G6PD) deficiency, where high-dose salicylates can precipitate hemolysis.
- Gout, where low-to-moderate aspirin doses reduce uric acid excretion and can precipitate an attack.
- Older adults with high fall risk and no strong cardiovascular indication — subdural hematoma risk often outweighs uncertain primary-prevention benefit.
Any decision to start, continue, or stop aspirin should be individualized. Patients on long-term aspirin should not discontinue abruptly without discussing the rationale with their prescriber, particularly in the first year after coronary stenting or a cardiovascular event.
References & Research Papers
GI Bleeding, Peptic Ulcer & PPI Co-Therapy
- Derry S, Loke YK. Risk of gastrointestinal haemorrhage with long term use of aspirin: meta-analysis. BMJ. 2000;321(7270):1183-1187.
- Chan FKL, Chung SCS, Suen BY, et al. Preventing recurrent upper gastrointestinal bleeding in patients with Helicobacter pylori infection who are taking low-dose aspirin or naproxen. N Engl J Med. 2001;344(13):967-973.
- Lanas A, García-Rodríguez LA, Arroyo MT, et al. Risk of upper gastrointestinal ulcer bleeding associated with selective cyclo-oxygenase-2 inhibitors, traditional non-aspirin non-steroidal anti-inflammatory drugs, aspirin and combinations. Gut. 2006;55(12):1731-1738.
- Chan FKL, Ching JYL, Hung LCT, et al. Clopidogrel versus aspirin and esomeprazole to prevent recurrent ulcer bleeding. N Engl J Med. 2005;352(3):238-244.
Cardiovascular Benefit-Risk & Hemorrhagic Stroke
- McNeil JJ, Wolfe R, Woods RL, et al.; ASPREE Investigator Group. Effect of aspirin on cardiovascular events and bleeding in the healthy elderly. N Engl J Med. 2018;379(16):1509-1518.
- Antithrombotic Trialists' (ATT) Collaboration. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet. 2009;373(9678):1849-1860.
- US Preventive Services Task Force. Aspirin use to prevent cardiovascular disease: US Preventive Services Task Force recommendation statement. JAMA. 2022;327(16):1577-1584.
Reye's Syndrome
- Hurwitz ES, Barrett MJ, Bregman D, et al. Public Health Service study on Reye's syndrome and medications: report of the pilot phase. N Engl J Med. 1985;313(14):849-857.
- Belay ED, Bresee JS, Holman RC, et al. Reye's syndrome in the United States from 1981 through 1997. N Engl J Med. 1999;340(18):1377-1382.
- Starko KM, Ray CG, Dominguez LB, Stromberg WL, Woodall DF. Reye's syndrome and salicylate use. Pediatrics. 1980;66(6):859-864.
Aspirin-Exacerbated Respiratory Disease & Hypersensitivity
- Stevenson DD, Szczeklik A. Clinical and pathologic perspectives on aspirin sensitivity and asthma. Chest. 2006;130(2):593-598.
- Kowalski ML, Agache I, Bavbek S, et al. Diagnosis and management of NSAID-exacerbated respiratory disease (N-ERD) — a EAACI position paper. Allergy. 2019;74(1):28-39.
Pregnancy & Pre-Eclampsia Prevention
- Rolnik DL, Wright D, Poon LC, et al. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N Engl J Med. 2017;377(7):613-622.
Perioperative Aspirin
- Devereaux PJ, Mrkobrada M, Sessler DI, et al.; POISE-2 Investigators. Aspirin in patients undergoing noncardiac surgery. N Engl J Med. 2014;370(16):1494-1503.
Salicylate Toxicity
- O'Malley GF. Emergency department management of the salicylate-poisoned patient. Emergency Medicine Clinics of North America. 2007;25(2):333-346.
- Juurlink DN, Gosselin S, Kielstein JT, et al.; EXTRIP Workgroup. Extracorporeal treatment for salicylate poisoning: systematic review and recommendations from the EXTRIP Workgroup. Annals of Emergency Medicine. 2015;66(2):165-181.
Connections
- Aspirin Overview
- Glycine & Aspirin Metabolism
- Glycine
- Peptic Ulcer Disease
- Aspirin for Heart Attack Prevention
- Aspirin Health Benefits
- Aspirin and the Kidneys
- Aspirin and Cancer Prevention
- Aspirin for Stroke Prevention
- NAC (N-Acetylcysteine)
- Stroke
- Tinnitus
- Tinnitus Relief
- Asthma
- Kidney Disease
- Hypertension
- Hemophilia
- Gout