Methylene Blue Mitochondrial Mechanism: How the Electron-Transport Bypass Works
The defining property of methylene blue as a therapeutic agent is its ability to act as an alternative electron carrier in the mitochondrial electron transport chain. To understand why this matters — and why the dose response is so unforgiving — you have to start with what mitochondria actually do, why they fail, and how a small dye molecule can prop them back up. This page is the molecular biology layer of the methylene blue story: how it works, where it works, and why low doses help while high doses harm.
Table of Contents
- What Mitochondria Do
- The Electron Transport Chain
- Where the Chain Fails
- Methylene Blue as an Electron Shuttle
- The MB / Leuco-MB Redox Couple
- The Hormetic Bell Curve
- Beyond Bypass: NOS, MAO, and Tau
- Tissue Distribution — Why the Brain
- Research Papers and References
- Connections
What Mitochondria Do
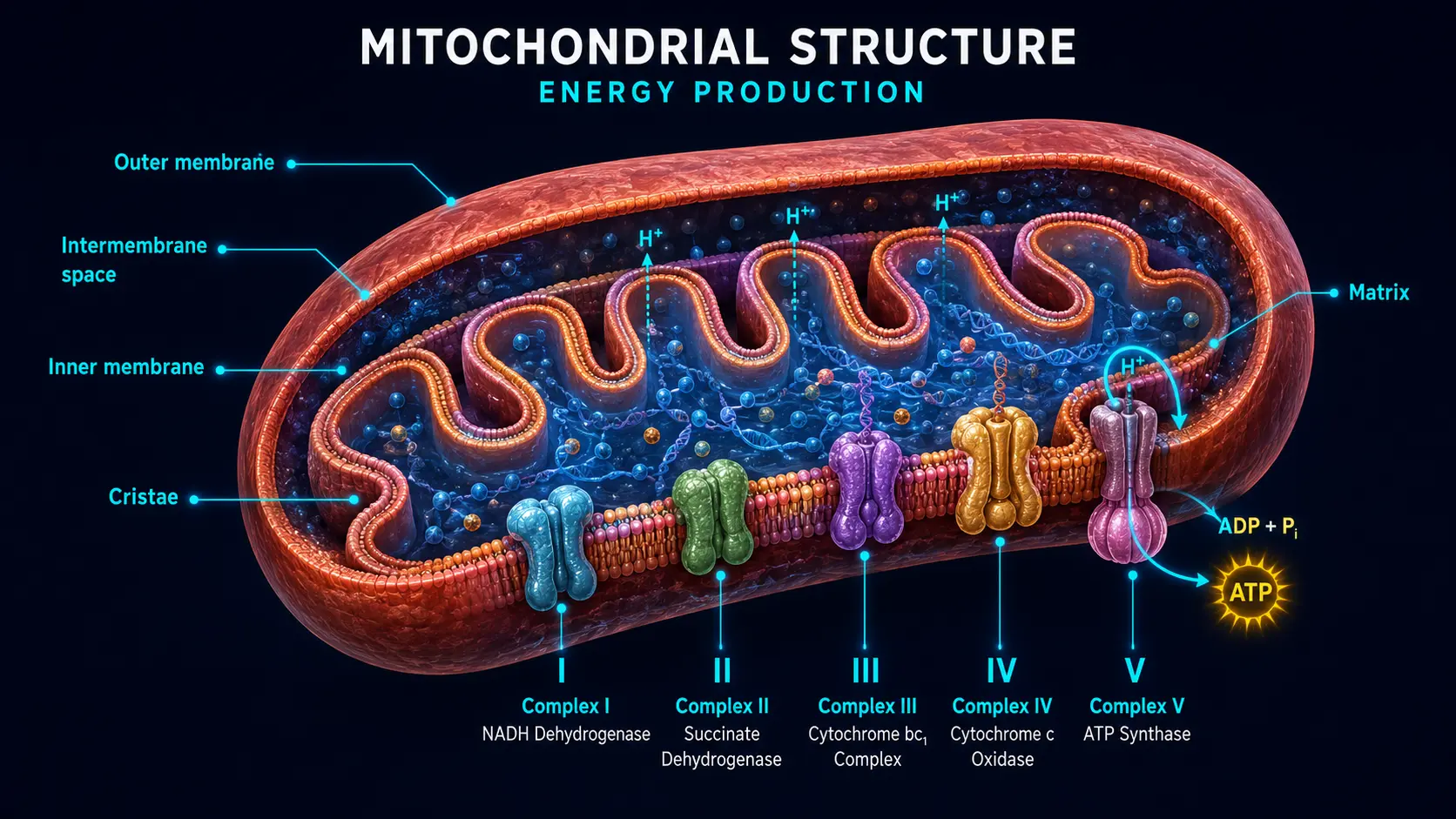
Mitochondria are the energy-producing organelles inside virtually every nucleated cell. A single neuron contains hundreds of them; a hardworking cardiac myocyte holds thousands. Their primary job is to convert the chemical energy stored in food (glucose, fatty acids, amino acids) into ATP — the universal cellular energy currency — through a sequence of oxidative reactions called cellular respiration. They also generate heat, regulate apoptosis, control calcium homeostasis, synthesize iron-sulfur clusters, modulate immune signaling, and create the small reactive-oxygen-species pulses cells use as second messengers.
When mitochondria function well, the cell makes ~30 ATP molecules per glucose, leaks minimal ROS, and signals fluently. When they don't, ATP production collapses, ROS production climbs, calcium handling becomes unstable, and the cell either compensates (through glycolysis), enters senescence, or dies. Mitochondrial dysfunction is now considered a driver of nearly every age-associated disease: heart failure, neurodegeneration, sarcopenia, type 2 diabetes, certain cancers, and the broad syndrome of "fatigue with no clear diagnosis" that occupies so much primary-care time.
The Electron Transport Chain
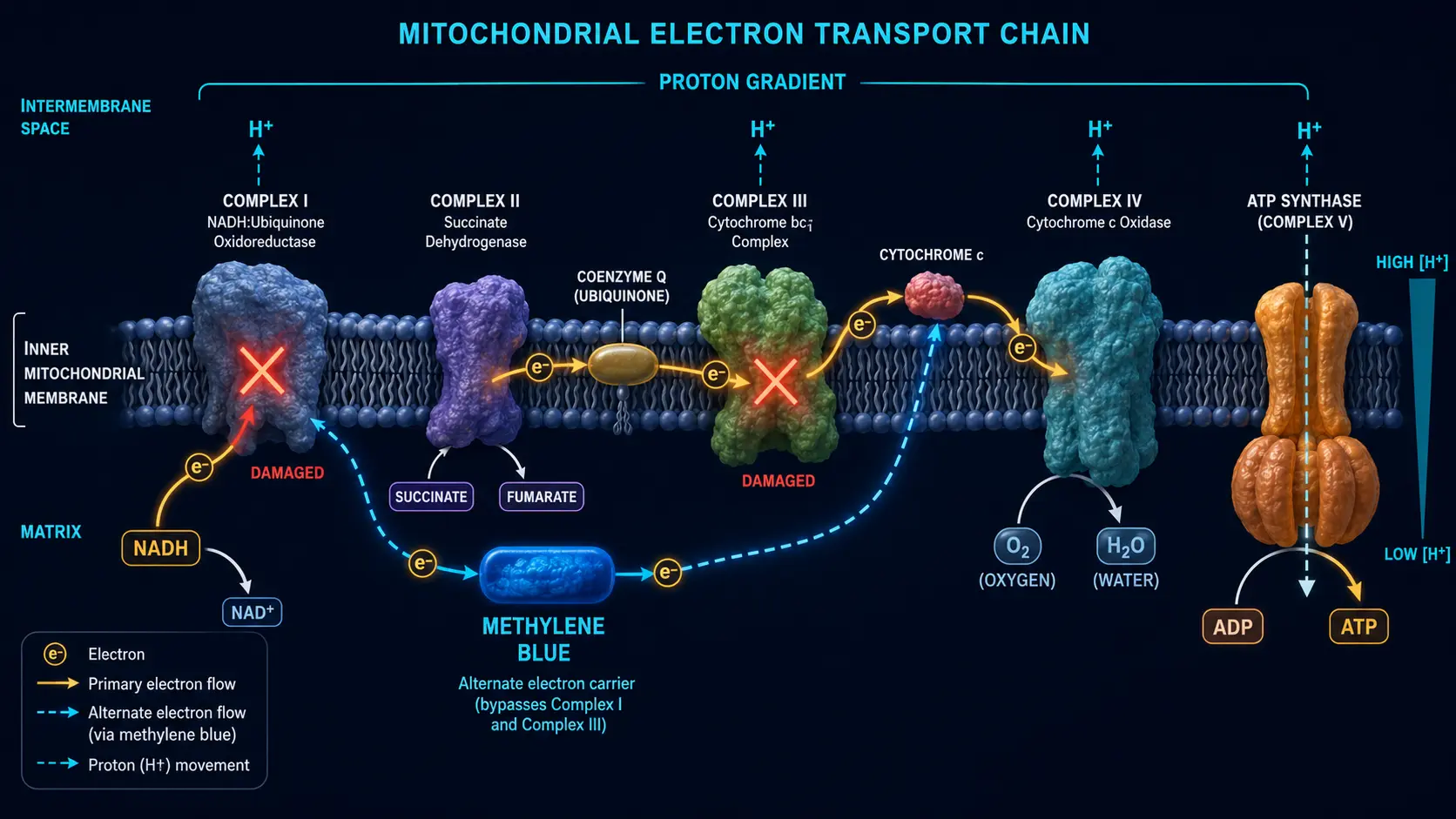
The final stage of cellular respiration is the electron transport chain (ETC), a series of four protein complexes embedded in the inner mitochondrial membrane:
- Complex I (NADH dehydrogenase) — accepts electrons from NADH; pumps protons. The largest complex; the most common site of inherited mitochondrial deficiency; the primary site of drug-induced toxicity (rotenone, MPP+, certain anticonvulsants).
- Complex II (succinate dehydrogenase) — accepts electrons from FADH2 produced by the Krebs cycle. Does not pump protons; the only ETC complex that is also a Krebs cycle enzyme.
- Complex III (cytochrome bc1) — transfers electrons from coenzyme Q (ubiquinone) to cytochrome c; pumps protons. Inhibited by antimycin A; site of significant ROS leak when impaired.
- Complex IV (cytochrome c oxidase) — the terminal step; transfers electrons to molecular oxygen, forming water; pumps protons. Inhibited by cyanide and carbon monoxide.
- ATP synthase (Complex V) — uses the proton gradient established by I, III, and IV to make ATP from ADP + Pi.
Electrons travel through the chain like cars on a downhill highway. The "downhill" is the redox potential difference between NADH (very reduced, electron-rich) and oxygen (very oxidized, electron-hungry). At each complex, some of that energy is used to pump protons into the intermembrane space, and the proton gradient powers ATP synthesis. The system is exquisitely tuned: small disruptions can collapse the gradient, halt ATP production, and turn the ETC into a ROS generator.
Where the Chain Fails
The ETC fails in characteristic, well-studied ways:
- Complex I deficiency — the most common acquired and inherited mitochondrial defect. Seen in Parkinson's, certain leukodystrophies, post-stroke tissue, aging brain, ischemia-reperfusion injury, sepsis. Drug exposures (statins, certain antibiotics, valproate) and toxins (rotenone, MPP+) target Complex I.
- Complex III dysfunction — characteristic in some hereditary mitochondrial disorders and a major source of superoxide leak when the chain is partially blocked.
- Complex IV deficiency — classic in Leigh syndrome and some forms of cardiomyopathy.
- Generalized dysfunction — the broad picture in aging and chronic disease, where complexes are partially impaired across the board, the membrane is leakier, mitochondrial DNA accumulates damage, and the system as a whole produces less ATP and more ROS.
The clinical signature of mitochondrial dysfunction includes fatigue (especially exercise intolerance), cognitive slowing, neurodegenerative symptoms, cardiomyopathy, exercise-induced lactic acidosis, and chronic muscle pain. Many "fibromyalgia," "chronic fatigue," and "long COVID" presentations show measurable mitochondrial dysfunction on muscle biopsy or specialty testing.
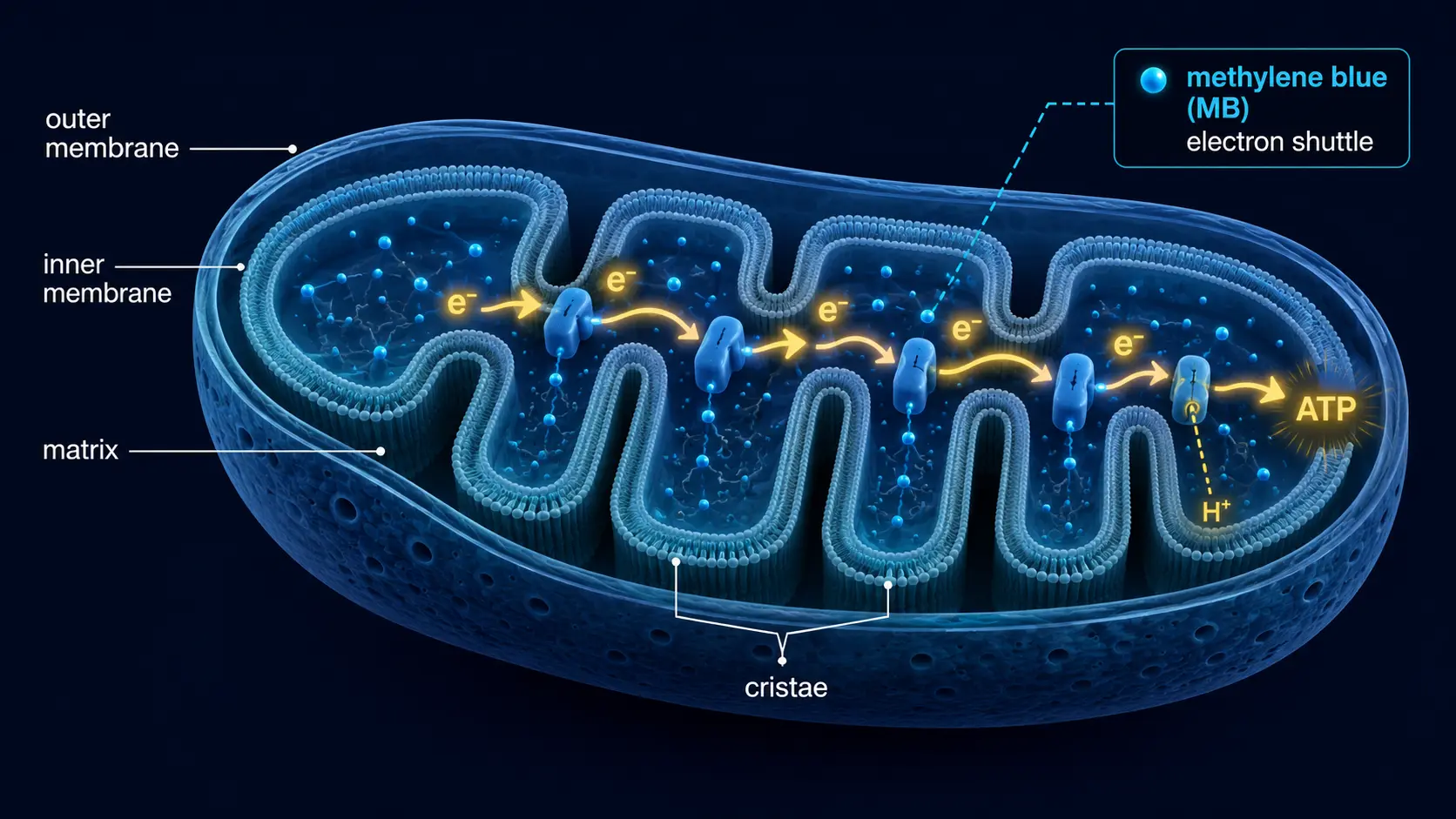
Methylene Blue as an Electron Shuttle
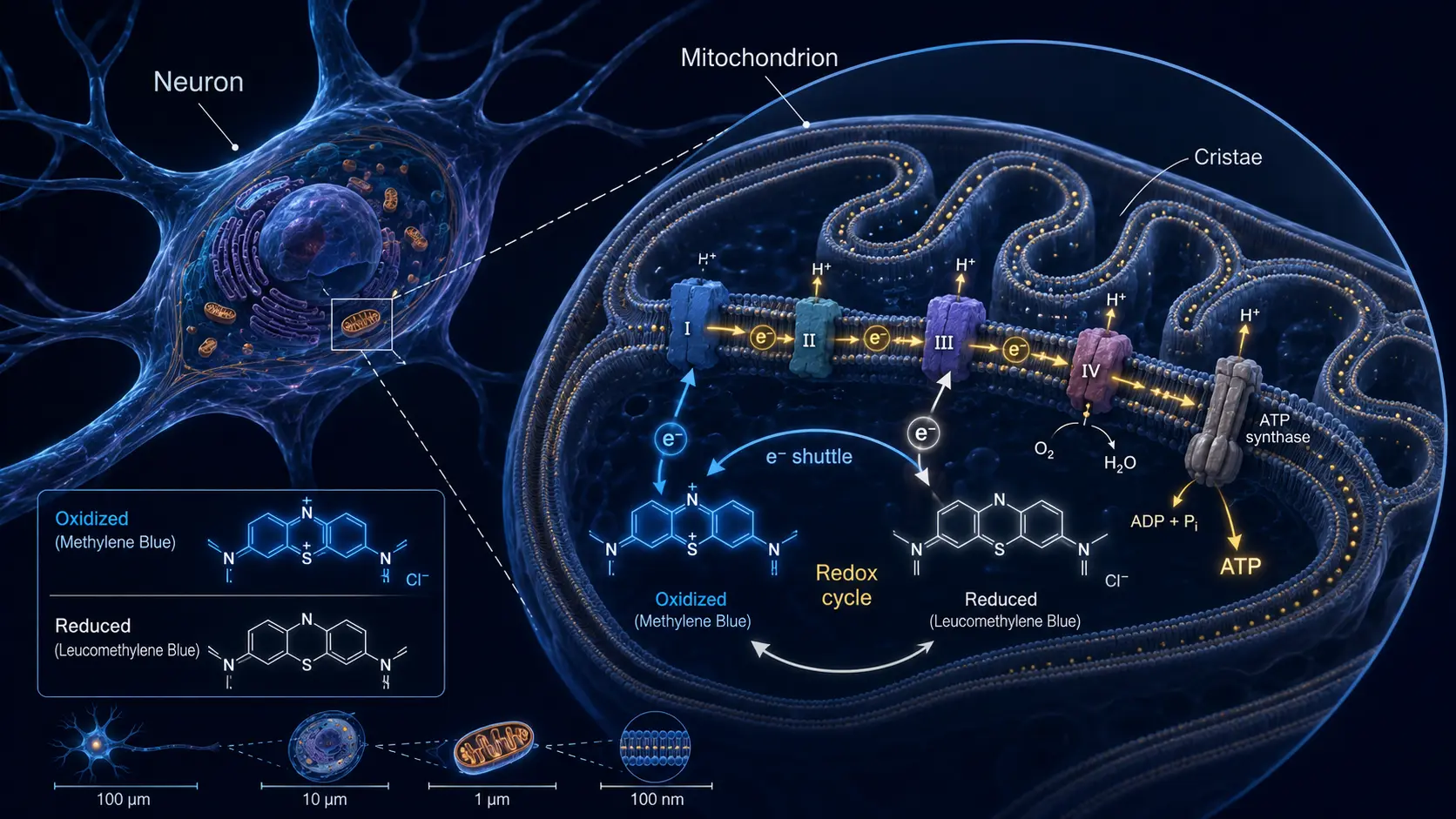
Methylene blue (MB) sits at the intersection of two unusual properties: it is highly water-soluble (it crosses biological membranes efficiently as the protonated form), and it has a redox potential between NADH and cytochrome c. That redox geometry is what lets it act as an alternate electron carrier:
- Inside the mitochondrial matrix, MB accepts two electrons (and a proton) from NADH, becoming the colorless reduced form, leuco-methylene blue (LMB). This is the same chemistry that drives the visible color change in classic chemistry-class titration demonstrations.
- LMB then donates electrons directly to cytochrome c (sitting between Complexes III and IV), regenerating the oxidized blue MB.
- The oxidized MB cycles back to accept more electrons from NADH.
The net effect: electrons that would otherwise be stuck in a partially impaired Complex I or III can take a shortcut directly to Complex IV. Cells with mitochondrial dysfunction recover ATP production. Reactive oxygen species generated by stalled-out Complex I and III drop. The chain runs again.
Critically, MB does not bypass Complex IV (cytochrome c oxidase) — the terminal step still has to function for the chain to deliver electrons to oxygen. This is why MB is not a useful intervention in cyanide poisoning (which inhibits Complex IV) but is useful in many other mitochondrial impairments.
The MB / Leuco-MB Redox Couple
The catalytic capacity of MB depends on the rapid interconversion between the oxidized blue form and the reduced colorless leuco form. This is one of the cleanest reversible biological redox systems known. The midpoint potential is about +11 mV at pH 7, comfortably between NADH (-320 mV) and cytochrome c (+254 mV). The reaction is fast, the interconversion is freely reversible, and a small amount of MB can shuttle a large number of electrons over time.
Several practical implications follow from the chemistry:
- Vitamin C interaction — ascorbate (vitamin C) reduces MB to LMB, both in solution and in vivo. Co-administration of vitamin C is sometimes used to enhance the bioavailability and tissue distribution of MB.
- Color as a marker of redox state — blue urine after MB administration reflects the oxidized form; the kidneys produce a fair amount of leuco-MB which oxidizes back to blue on contact with air or oxidizing agents in the bladder.
- Light sensitivity — visible light (especially red, around 660 nm) excites MB to a triplet state that produces singlet oxygen. This is the basis for photodynamic therapy (see Photodynamic Therapy).
The Hormetic Bell Curve
The most clinically important property of methylene blue is its biphasic dose-response. Low doses (0.5–4 mg/kg) are net antioxidant: they accelerate the ETC, reduce ROS leak, and improve cellular energetics. High doses (above ~10 mg/kg in humans) become net pro-oxidant: MB accepts electrons but cannot effectively shuttle them all to cytochrome c, generating superoxide instead and contributing to methemoglobinemia (oxidation of hemoglobin's iron to ferric form).
The shape of the curve is one of the cleanest examples of hormesis — a stress that is beneficial at low doses and harmful at higher doses — in pharmacology. Practical consequences:
- Doubling a nootropic dose does not double the cognitive effect; it can reverse it
- The therapeutic dose for methemoglobinemia (1–2 mg/kg IV as a single dose) sits well above the nootropic range and is given for an entirely different indication
- Doses above 7 mg/kg can paradoxically cause methemoglobinemia, especially in repeated dosing
- The optimal dose for any given individual depends on body weight, antioxidant status, hemoglobin reserve, and concurrent medications
This is why dosing protocols matter. See the Dosing Guide for practical numbers.
Beyond Bypass: NOS, MAO, and Tau
Methylene blue has at least four mechanisms of action besides the electron-transport bypass, and the relative importance of each depends on the dose and the indication.
Nitric oxide synthase (NOS) inhibition
MB inhibits all three isoforms of nitric oxide synthase, particularly inducible NOS (iNOS) and the soluble form of guanylate cyclase that NO activates. This is the basis for its use in vasoplegic shock (catecholamine-resistant low blood pressure during cardiac surgery), where it restores vascular tone. The same mechanism contributes to its blood-pressure raising effect in some patients.
Monoamine oxidase A (MAO-A) inhibition
MB is a reversible inhibitor of MAO-A at clinically relevant concentrations. This accounts for both its mood effects (serotonin and norepinephrine accumulate) and its most dangerous interaction (with serotonergic drugs — see Drug Interactions). The MAO-A inhibition is largely responsible for the FDA black-box warning on parenteral methylene blue.
Tau aggregation inhibition
Methylene blue and its more soluble derivative LMTX disrupt the abnormal aggregation of tau protein implicated in Alzheimer's disease and frontotemporal dementia. Phase-3 trials of LMTX missed primary endpoints but showed potentially meaningful effects in monotherapy subgroups. Research continues, particularly in primary tauopathies.
Antimicrobial photodynamic activity
Under visible light (especially red), MB generates singlet oxygen that damages bacterial, fungal, and viral structures. This is the basis for antimicrobial photodynamic therapy (aPDT) used in dentistry and dermatology, and it explains MB's historical use as an antiseptic before modern antibiotics.
Tissue Distribution — Why the Brain
Methylene blue is unusual among small-molecule drugs in that it crosses the blood-brain barrier readily and accumulates in mitochondria-rich tissues. Brain tissue concentrations are roughly 5–10 times plasma; the highest concentrations are in regions with the densest mitochondrial populations — cortex, hippocampus, basal ganglia. Heart, kidney, and liver also accumulate methylene blue, which is part of why both the brain effects and the renal effects (blue urine) are so prominent.
The tissue distribution and the mitochondrial mechanism together explain why MB has been studied for cognitive effects, mood, neurodegeneration, post-cardiac-surgery delirium, and ischemia-reperfusion injury — tissues that are mitochondria-dense and ETC-dependent are the ones that respond.
Research Papers and References
- MB and electron transport chain — PubMed search
- MB and mitochondrial ATP — PubMed search
- MB hormetic dose response — PubMed search
- MB and NOS in vasoplegia — PubMed search
- LMTX and tau aggregation — PubMed search
- MB brain penetration — PubMed search
- MB / leuco-MB redox cycling — PubMed search
Connections
- Methylene Blue Deep-Dive Articles:
- Methylene Blue Overview
- Dosing Guide
- Cognitive Enhancement & ADHD
- Methylene Blue & Long COVID
- Drug Interactions & Serotonin Syndrome
- Methemoglobinemia & G6PD
- Photodynamic Therapy & Cancer
- Pharmaceutical vs Industrial
- History
- NAD+ and NMN
- Longevity Protocols
- Creatine
- Oxidative Stress
- Depression
- Dementia