Methylene Blue, Methemoglobinemia, and G6PD Deficiency: The Hemolytic Risk

Methylene blue has a paradoxical relationship with two related blood disorders: it is the FDA-approved treatment for methemoglobinemia at therapeutic doses, but at higher doses it can cause the same condition. And in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency — one of the most common enzyme deficiencies worldwide — methylene blue can precipitate severe hemolytic anemia. These two conditions are the most important hematologic concerns in any methylene blue protocol, and screening should happen before therapeutic dosing.
Table of Contents
- What Methemoglobinemia Is
- MB as a Treatment
- MB as a Cause
- G6PD Deficiency
- Why G6PD Matters with Methylene Blue
- G6PD Screening
- Recognizing Hemolytic Anemia
- High-Risk Populations
- Research Papers and References
- Connections
What Methemoglobinemia Is
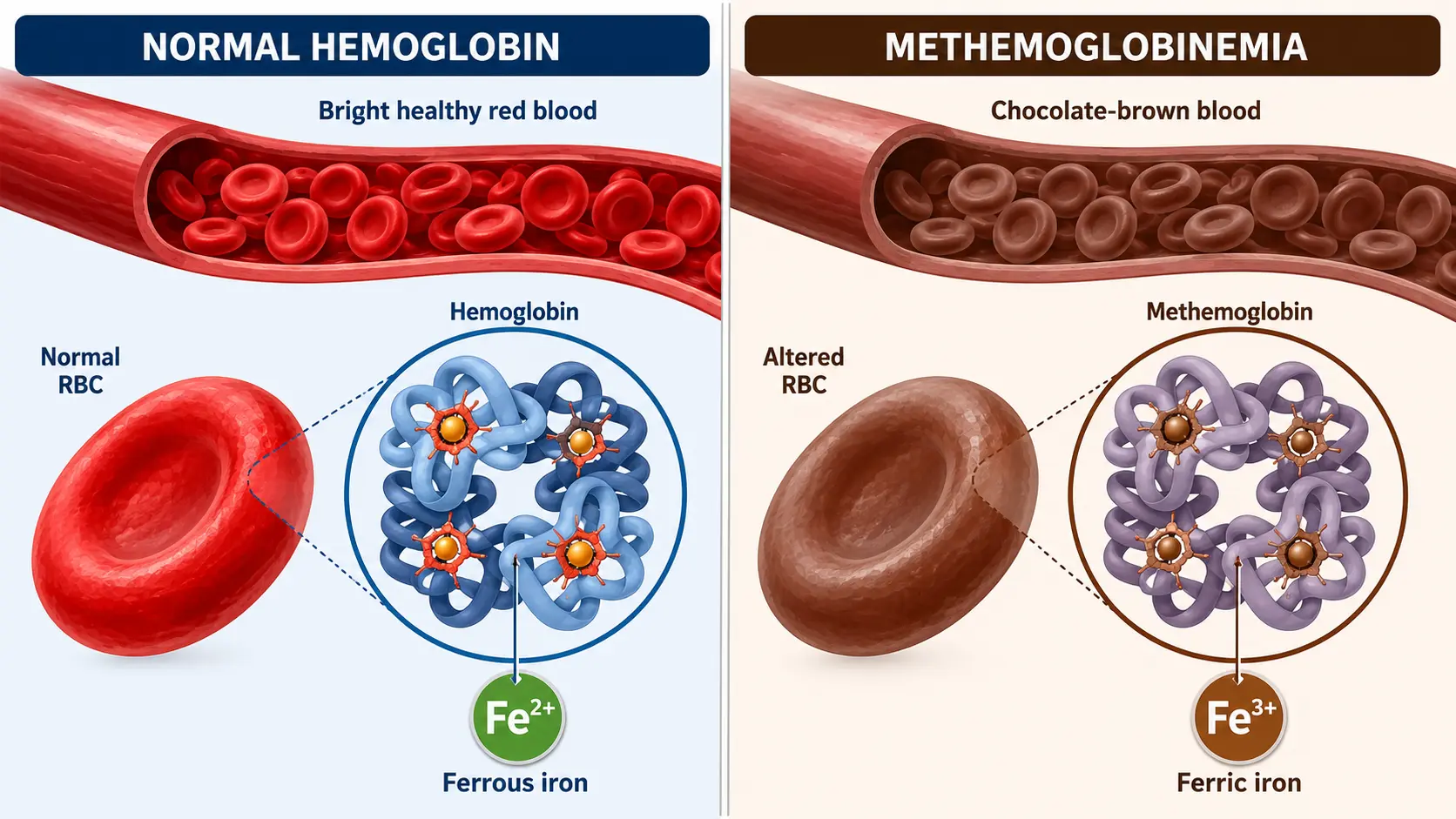
Hemoglobin in red blood cells contains iron in the ferrous (Fe²⁺) state, which is required to bind and release oxygen reversibly. About 1% of hemoglobin is normally oxidized to the ferric (Fe³⁺) form — methemoglobin — every day, and is reduced back by an enzyme system that depends on NADH and cytochrome b5 reductase. Methemoglobin cannot bind oxygen, so a high methemoglobin fraction reduces blood oxygen-carrying capacity.
Symptoms emerge as methemoglobin rises:
- 10–20%: bluish skin discoloration (cyanosis), often described as "chocolate-brown blood" on phlebotomy
- 20–30%: headache, fatigue, dizziness, dyspnea
- 30–50%: tachycardia, lethargy, confusion
- 50–70%: seizures, coma, lactic acidosis
- > 70%: typically fatal without treatment
Acquired methemoglobinemia is most often drug- or chemical-induced: dapsone, benzocaine (Hurricane spray, teething gels), nitroprusside, lidocaine in high doses, certain herbal teas, well-water nitrates, aniline dyes, and chlorate. Inherited methemoglobinemia (cytochrome b5 reductase deficiency or hemoglobin M variants) is rare but worth knowing about.
MB as a Treatment
At therapeutic doses (1–2 mg/kg IV given over 5 minutes), methylene blue rapidly reduces methemoglobin back to functional hemoglobin. The mechanism: MB is reduced to leuco-MB by NADPH (via the alternate NADPH-MB reductase pathway present in red blood cells), and leuco-MB then donates electrons to methemoglobin, restoring the ferrous iron state. Clinical response is usually visible within 30 minutes; methemoglobin typically falls from 30–40% back below 10%.
This is the single most established and life-saving use of methylene blue. The maximum cumulative dose for methemoglobinemia treatment is 7 mg/kg, after which alternative therapies (exchange transfusion, hyperbaric oxygen, ascorbic acid) are considered.
MB as a Cause
The same molecule that treats methemoglobinemia at 1–2 mg/kg can cause it at higher repeat doses or in vulnerable patients. The mechanism is the dose-dependent flip from antioxidant to pro-oxidant: at high doses, methylene blue accepts electrons from NADH but cannot effectively shuttle them to cytochrome c, generating superoxide and ultimately oxidizing hemoglobin's iron back to ferric form.
The threshold above which paradoxical methemoglobinemia becomes likely is around 7 mg/kg cumulative or 15 mg/kg single dose. Risk factors that lower this threshold:
- G6PD deficiency (see below) — the dominant risk factor
- NADPH-quinone oxidoreductase deficiency
- Pre-existing methemoglobinemia
- Concurrent oxidant exposure (dapsone, benzocaine, certain antibiotics)
- Severe sepsis or oxidant overload
For nootropic dosing (0.5–4 mg/kg/day), the risk in healthy non-G6PD-deficient adults is very low. The risk rises in vulnerable individuals or with sustained dosing well above the nootropic range.
G6PD Deficiency
Glucose-6-phosphate dehydrogenase (G6PD) is the rate-limiting enzyme of the pentose phosphate pathway, which generates NADPH — the reducing equivalent that protects red blood cells from oxidative damage. G6PD deficiency is the most common enzyme deficiency in humans, affecting approximately 400–500 million people globally. Prevalence is high in:
- Mediterranean populations (Italian, Greek, Spanish, Middle Eastern) — up to 10–30%
- African populations (especially West African and African-American men) — 10–15%
- Southeast Asian populations — 5–10%
- South Asian populations
The trait is X-linked. Hemizygous males (one X chromosome with the affected gene) and homozygous females have full-blown deficiency. Heterozygous females have intermediate expression with mosaic red cell populations.
Most G6PD-deficient individuals are asymptomatic until exposed to an oxidative trigger. Common triggers include fava beans (favism), certain antimalarials (primaquine, dapsone), sulfa antibiotics, nitrofurantoin, certain henna and dye products, and — importantly — methylene blue.
Why G6PD Matters with Methylene Blue
In G6PD-deficient red cells, the NADPH-dependent reduction of methylene blue to leuco-MB is impaired. Methylene blue therefore stays in the oxidized blue form longer, generating direct oxidative stress on already-vulnerable red cells. The result can be:
- Acute hemolytic anemia — massive red cell destruction within hours to days of MB exposure
- Paradoxical methemoglobinemia — MB fails to reduce methemoglobin and can worsen it
- Hemoglobinuria — dark-red or brown urine from free hemoglobin
- Acute kidney injury — from hemoglobin pigment nephropathy
The risk is dose-dependent. Severe hemolysis is most often reported with intravenous methylene blue at therapeutic doses (1–2 mg/kg) in G6PD-deficient patients. The risk at low oral nootropic doses (0.5–1 mg/kg) is lower but not zero, and increases with sustained daily dosing.
G6PD Screening
The good news: G6PD deficiency is easily detected with a simple blood test. The qualitative fluorescent spot test or quantitative spectrophotometric assay are both inexpensive, widely available, and definitive in non-acute settings.
Important caveat: testing during or immediately after an acute hemolytic episode can give a false-negative result because the most-deficient red cells have already been destroyed. Wait at least 4–6 weeks after a hemolytic event before screening, or test the patient's relatives.
Practical recommendation: any patient considering therapeutic doses of methylene blue should have G6PD status documented before initiation. The cost is low (typically $30–75 out of pocket), and the information is permanent. For patients of Mediterranean, African, Middle Eastern, or Southeast Asian descent, the test is particularly important given baseline prevalence.
Recognizing Hemolytic Anemia
Symptoms typically develop 24–72 hours after exposure to a triggering agent:
- Sudden fatigue, weakness, pallor
- Jaundice (yellow eyes/skin from elevated bilirubin)
- Dark-red, cola-colored, or brown urine (hemoglobinuria)
- Back pain or flank pain
- Tachycardia, shortness of breath on exertion
- Fever, chills
Laboratory features: drop in hemoglobin and hematocrit, rise in indirect bilirubin and LDH, fall in haptoglobin, elevated reticulocyte count (compensatory), Heinz bodies and bite cells on peripheral smear. Severe cases require IV hydration to protect the kidneys, blood transfusion if hemoglobin is critically low, and stopping the offending agent.
High-Risk Populations
Patients in whom particular caution is warranted:
- Known G6PD deficiency — absolute contraindication for therapeutic methylene blue; nootropic doses possible only with hematology input
- Mediterranean, African, Middle Eastern, or Southeast Asian descent without documented G6PD status — screen first
- Family history of favism or jaundice on antimalarials
- Newborns — immature glucuronidation increases bilirubin toxicity from any hemolysis
- Patients on other oxidant-stressing drugs — dapsone, primaquine, nitrofurantoin, sulfa drugs
- Severe acute illness, sepsis, DKA — oxidant load is already high
For all of these, the right answer is to confirm G6PD status, work with a clinician, and consider whether the methylene blue indication justifies the additional risk.
Research Papers and References
- G6PD and methylene blue hemolysis — PubMed search
- Methemoglobinemia treatment — PubMed search
- Paradoxical methemoglobinemia from MB — PubMed search
- G6PD deficiency global epidemiology — PubMed search
- G6PD screening tests — PubMed search
- Hemoglobin pigment nephropathy — PubMed search
External Authoritative Resources
Connections
- Methylene Blue Deep-Dive Articles:
- Methylene Blue Overview
- Mitochondrial Mechanism
- Dosing Guide
- Drug Interactions
- Pharmaceutical vs Industrial
- Hematology
- Anemia
- Complete Blood Count
- Iron Panel
- Kidney Function Tests