History of Methylene Blue: Ehrlich, Malaria, and the Phenothiazine Revolution

Methylene blue's history is one of the most consequential in modern pharmacology. Synthesized in 1876 by a 22-year-old German chemistry student named Heinrich Caro as a textile dye, it became the first synthetic compound used as a medication, the molecule that proved Paul Ehrlich's "magic bullet" hypothesis of selective drug action, the structural template for the phenothiazine class of antipsychotics that revolutionized psychiatry in the 1950s, and now — nearly 150 years after its synthesis — an active subject of mitochondrial and cognitive research. Few molecules in chemistry have a longer, more continuous medical career.
Table of Contents
- 1876: Synthesis
- Paul Ehrlich and the Magic Bullet
- Malaria: The First Synthetic Antimalarial
- Antiseptic and Urinary Antiseptic Era
- The Phenothiazine Revolution
- Chlorpromazine and Modern Psychiatry
- A Second Life as Antidote
- Modern Cognitive and Mitochondrial Research
- Legacy
- Research Papers and References
- Connections
1876: Synthesis
Methylene blue was first synthesized in 1876 by Heinrich Caro at the German dye company BASF. Caro was working on aniline-based dyes for the textile industry, which was then the largest application of synthetic chemistry. Methylene blue's deep-blue color, light-fastness, and affinity for cotton made it commercially valuable as a fabric dye almost immediately. The chemical formula, methylthioninium chloride, locates the compound in the phenothiazine class — a sulfur-containing tricyclic aromatic system that would prove unexpectedly versatile.
Within a few years of its commercial introduction, biologists noticed that methylene blue selectively stained certain cellular structures — particularly nuclei and mitochondria — under the microscope. This made it one of the first reliable cytological dyes, and the routine staining of biological tissues with MB became a standard 19th-century laboratory technique. The 1882 isolation of the tubercle bacillus by Robert Koch used a methylene-blue-based stain. The "vital staining" of living cells — revealing dynamic structures within still-living organisms — was pioneered using methylene blue.
Paul Ehrlich and the Magic Bullet
Paul Ehrlich (1854–1915), the German immunologist and Nobel laureate who would later be called the founder of chemotherapy, became fascinated with methylene blue as a graduate student. His PhD thesis (1878) was on the staining of biological tissues with aniline dyes. He noticed that methylene blue accumulated preferentially in living nerve tissue (the basis for vital staining of nerves) and in malaria parasites within infected red blood cells.
The selectivity led to Ehrlich's foundational hypothesis: a chemical that selectively binds to a pathogen could selectively kill it — the "magic bullet" or "Zauberkugel." This idea, that drugs could be designed to target specific molecular features of pathogens or diseased cells, became the conceptual foundation for all subsequent rational drug discovery. Ehrlich's salvarsan (the first effective syphilis treatment, 1909) was the most direct expression of the principle, but methylene blue was the first.
Malaria: The First Synthetic Antimalarial
In 1891, Ehrlich and his collaborator Paul Guttmann published a study showing that methylene blue could cure malaria in patients. The doses were uncomfortable (causing the characteristic blue urine) and the drug was eventually displaced by quinine and later by synthetic antimalarials, but methylene blue retained a place in malaria treatment for decades. It was the first wholly synthetic compound used to treat a human infection — the proof of concept for chemotherapy.
Methylene blue's antimalarial activity remains relevant today. In the era of artemisinin-resistant Plasmodium falciparum, methylene blue has been investigated as a partner drug in combination therapies, particularly in West Africa. Trials in Burkina Faso and elsewhere have shown that MB combined with artemisinin or amodiaquine is effective and may reduce gametocytemia (the form of the parasite that infects mosquitoes), potentially limiting transmission.
Antiseptic and Urinary Antiseptic Era
By the early 20th century, methylene blue had found multiple medical applications:
- Urinary tract antiseptic — oral methylene blue was prescribed for urinary tract infections (the antimicrobial activity is real, though limited; the blue urine was reassuring to patients)
- Topical antiseptic — for minor wounds and skin infections
- Diagnostic dye — for chromoendoscopy, surgical mapping, and dye dilution studies of cardiac function
- Carbon monoxide and cyanide poisoning antidote — methylene blue was widely used during World War I for these toxicities
- Methemoglobinemia treatment — the application that would eventually become the FDA-approved indication
From roughly 1900 to 1950, methylene blue was a workhorse in the medical pharmacopeia. Its use waned as more specific antibiotics and modern drugs replaced it for most indications, but it never disappeared.
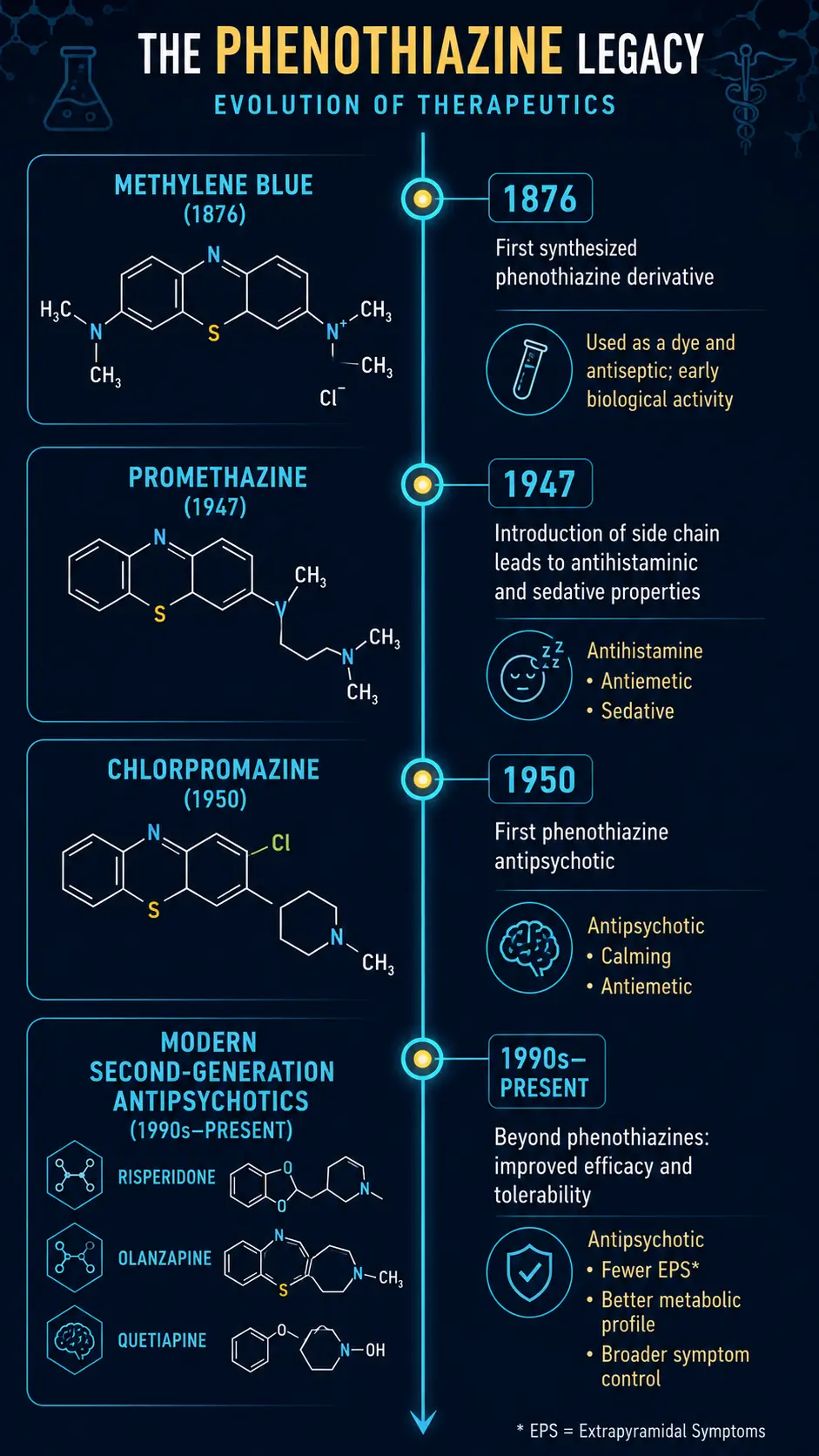
The Phenothiazine Revolution
The most consequential downstream effect of methylene blue's discovery came from chemists working on its core structure. The phenothiazine ring system is the template for an entire class of psychotropic drugs that emerged in the 1940s and 1950s. The story:
- 1883: Bernthsen synthesizes phenothiazine itself, derived from methylene blue chemistry
- 1934: Phenothiazines are tested as antimalarials and antihistamines
- 1947: French chemists at Rhone-Poulenc develop promethazine, a sedating antihistamine
- 1950: The structural modification that produces chlorpromazine
- 1952: French psychiatrists Jean Delay and Pierre Deniker publish the first clinical use of chlorpromazine for schizophrenia
- 1954: Thorazine (chlorpromazine) approved in the US, transforming psychiatric care
The line from a 19th-century textile dye to the deinstitutionalization of psychiatric patients in the mid-20th century is direct. Without methylene blue's tricyclic aromatic chemistry, the entire first generation of antipsychotics — chlorpromazine, fluphenazine, trifluoperazine, perphenazine, thioridazine — would not exist in their current form.
Chlorpromazine and Modern Psychiatry
Chlorpromazine's clinical effects were revolutionary. Patients with schizophrenia who had been confined to asylums for decades could, on chlorpromazine, function in the community. The state psychiatric hospital census in the US peaked at over 500,000 in 1955; by 1990 it had fallen below 100,000, driven primarily by the availability of antipsychotics. The biological basis — dopamine D2 receptor antagonism — would not be elucidated until the 1970s, but the clinical effects were unmistakable from the 1950s onward.
The atypical antipsychotics (clozapine, risperidone, olanzapine, quetiapine) developed since the 1980s have moved away from the pure phenothiazine structure but still trace their conceptual origin to chlorpromazine. The tricyclic antidepressants (imipramine, amitriptyline) likewise share the tricyclic-aromatic chemistry. The lineage of half of modern psychotropic medicine runs through methylene blue.
A Second Life as Antidote
By the 1970s, methylene blue's clinical use had narrowed to a few specific indications: methemoglobinemia, vasoplegic shock, surgical staining, and ifosfamide encephalopathy. The drug was inexpensive, off-patent, and reliably stocked in hospital pharmacies for emergency use. It looked like a relic — useful, but not the subject of active research.
Three developments changed that:
- The 1990s discovery of methylene blue's effect on tau aggregation, opening Alzheimer's research
- The 2000s research on methylene blue as a mitochondrial electron carrier, with implications for aging, neurodegeneration, and metabolic disease
- The 2010s emergence of the cognitive enhancement and longevity communities, who rediscovered methylene blue's nootropic potential
Modern Cognitive and Mitochondrial Research
The modern research arc began with David Liebowitz's group at the University of Texas demonstrating in the 2000s that methylene blue improves cognition in animal models of Alzheimer's, stroke, and aging. Phase 2 and phase 3 trials of LMTX (the leuco-MB derivative) by Wischik's group in Aberdeen produced mixed results in Alzheimer's but identified subgroups with potentially meaningful effects. The 2016 Rodriguez fMRI study (see Cognitive Enhancement page) provided the first rigorous human evidence of cognitive effects in healthy adults.
Concurrently, the post-COVID era brought renewed interest in mitochondrial support drugs as long COVID and ME/CFS research expanded. Methylene blue, with its cheap price, well-documented safety profile (within its dose ceiling), and brain penetration, has been one of the more commonly tried interventions in clinician-led protocols.
Active areas of investigation as of 2025–2026 include:
- Mitochondrial-targeted MB derivatives (MitoQ-style compounds)
- Combinations with NAD+ precursors
- Photodynamic cancer protocols
- Long COVID and post-viral fatigue trials
- Tauopathies beyond Alzheimer's (frontotemporal dementia, PSP)
- Antimicrobial photodynamic therapy
Legacy
Methylene blue's 150-year medical history offers a few observations:
- The molecule has been continuously used in human medicine for longer than aspirin (1899), longer than penicillin (1928), longer than virtually anything else still on shelves
- The same molecule has been an antimalarial, antiseptic, antidote, surgical dye, antipsychotic precursor, mitochondrial nootropic, and photodynamic agent — a versatility nearly unmatched in pharmacology
- Each generation has rediscovered methylene blue's possibilities, and each has contributed mechanism-of-action details
- The transition from textile dye to medical antidote to neuropharmacology to cognitive enhancement traces the broader history of how modern medicine has matured
For a single molecule synthesized as a fabric dye in 1876, that is an unusually full life.
Research Papers and References
- Ehrlich, MB, and the origin of chemotherapy — PubMed search
- Phenothiazine origins of antipsychotics — PubMed search
- MB in modern malaria treatment — PubMed search
- Wischik LMTX in Alzheimer's — PubMed search
- MB in mitochondrial aging research — PubMed search
Connections
- Methylene Blue Deep-Dive Articles:
- Methylene Blue Overview
- Mitochondrial Mechanism
- Cognitive Enhancement
- Photodynamic Therapy
- Psychiatry
- Depression
- Dementia
- Aspirin (another long-lived drug)