Methylene Blue Dosing Guide: mg/kg, Oral, Sublingual, IV
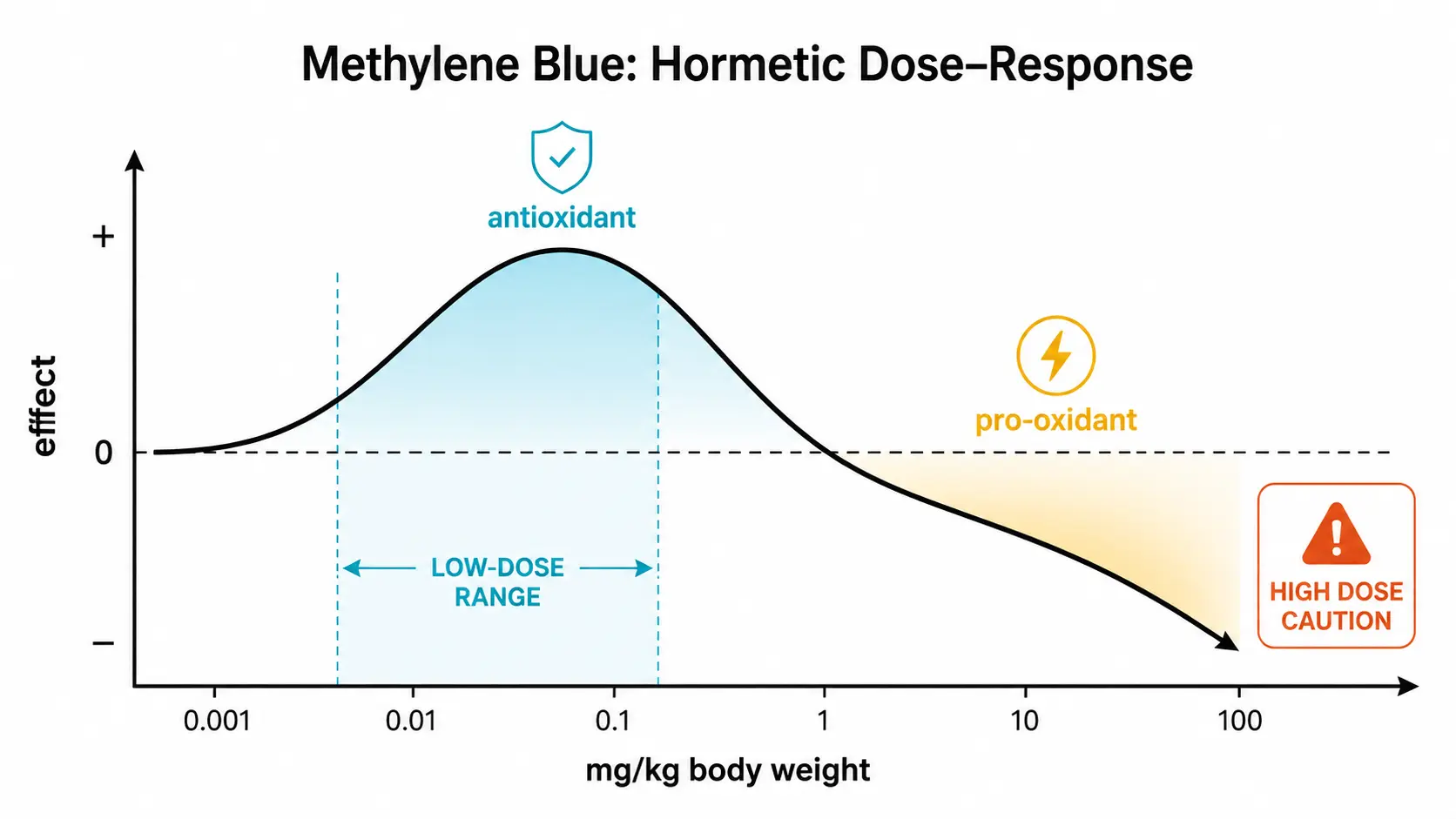

Dosing methylene blue correctly is the difference between a useful nootropic, a clinical antidote, and a serious adverse event. The hormetic bell curve described in the mitochondrial mechanism page means that doubling a dose can flip the molecule from antioxidant to pro-oxidant, restore vs raise methemoglobin, and tolerable vs dangerous. This guide gathers what's known about dosing across indications, routes of administration, timing, and the practical considerations that determine whether a given protocol is safe.
This page is informational, not a prescription. Off-label methylene blue use should be discussed with a clinician who can review your medication list, screen for G6PD deficiency, and confirm that the formulation you have access to is pharmaceutical grade.
Table of Contents
- Three Dose Regimes
- Nootropic / Cognitive Dosing
- Therapeutic FDA-Approved Dosing
- Routes of Administration
- Oral Bioavailability and PK
- Timing and Co-Administration
- Vitamin C and Light Considerations
- Weight-Based Calculation Examples
- Frequency and Cycling
- Hard Contraindications Before Any Dose
- Research Papers and References
- Connections
Three Dose Regimes
Methylene blue is dosed in three distinct ranges, each with a different intent and a different safety profile:
- Nootropic / cognitive (0.5–4 mg/kg/day) — off-label; used for memory, focus, mood, post-viral fatigue, and as a longevity adjunct. Net antioxidant effect; well below the threshold for methemoglobin formation in healthy individuals.
- Therapeutic / clinical (1–2 mg/kg single IV dose) — FDA-approved for acute methemoglobinemia. Given as a 1% solution over 5 minutes. The same dose used in vasoplegic shock and ifosfamide encephalopathy.
- High-dose research (4–7 mg/kg) — investigational protocols for certain cancers, malaria, or refractory shock. Above this range adverse effects become predictable rather than rare.
Beyond about 7 mg/kg, methylene blue paradoxically causes methemoglobinemia, especially with repeat dosing — the very condition the FDA approval treats.
Nootropic / Cognitive Dosing
The most-cited cognitive trial is Rodriguez et al. (2016), which used a single 280 mg oral dose (about 4 mg/kg in a 70 kg adult) and reported improvements in attention and memory tasks 1 hour post-dose. Most contemporary nootropic protocols use lower doses, daily or every-other-day:
- Microdose — 0.5–1 mg/kg/day; 35–70 mg for a 70 kg adult. Often subjectively sufficient for focus and mild cognitive lift; minimal blue urine; lowest adverse effect profile.
- Standard nootropic — 1–2 mg/kg/day; 70–140 mg for a 70 kg adult. Most common protocol used by clinicians and the broader self-experimentation community. Blue urine is reliable.
- High nootropic — 2–4 mg/kg/day; 140–280 mg for a 70 kg adult. Used for more pronounced cognitive enhancement or for neurodegenerative protocols. Approaches the high end of the antioxidant window; some users report a mild flat or "off" sensation.
A common starting protocol: 1 mg/kg/day for the first week (assess tolerance and the colored-urine effect), then titrate to 1–2 mg/kg/day if useful. Many users find diminishing returns above 2 mg/kg.
Therapeutic FDA-Approved Dosing
- Acute methemoglobinemia — 1–2 mg/kg IV as a 1% solution over 5 minutes; may repeat in 1 hour if methemoglobin remains > 30% or symptoms persist. Maximum cumulative dose 7 mg/kg.
- Vasoplegic shock — 1–2 mg/kg IV bolus, sometimes followed by infusion at 0.25 mg/kg/hr.
- Ifosfamide encephalopathy — 50 mg IV every 4 hours.
- Cyanide poisoning — not a primary indication (sodium thiosulfate, hydroxocobalamin preferred), but historically used at 1–2 mg/kg IV.
These are inpatient indications. The relevance for off-label use is mostly to anchor the upper end of the safe single-dose range: 2 mg/kg IV is FDA-approved, 7 mg/kg is the cumulative ceiling, and methemoglobinemia begins to predominate above that.
Routes of Administration
Oral
The most common route for off-label/nootropic use. Bioavailability is roughly 50–70% with significant inter-individual variation. Effects begin within 30–60 minutes; peak around 1–2 hours; half-life around 5–6 hours. Blue urine appears within 1–2 hours and persists for 24–36 hours.
Sublingual
Drops or troches placed under the tongue and held for 60–90 seconds before swallowing. Absorbed partly through oral mucosa, bypassing some first-pass metabolism. Effects begin within 15–30 minutes. The major practical issue is taste (mild, slightly metallic) and tongue staining (blue tongue persists for 1–3 hours).
Intravenous
The reference route for FDA-approved indications. Provides 100% bioavailability and predictable peak concentrations. Inappropriate for outpatient nootropic use.
Topical
Used for photodynamic therapy of skin lesions, periodontal disease, and certain dermatologic conditions. Different pharmacology entirely — the mechanism is local singlet-oxygen generation under light exposure rather than systemic mitochondrial support.
Intranasal
Investigational; some practitioners use it for direct CNS delivery. Limited published data on bioavailability or safety in this route.
Oral Bioavailability and PK
Oral methylene blue is partly oxidized in the gut and during first-pass hepatic metabolism. Approximately 50–70% reaches systemic circulation; the rest appears as the leuco form in feces (which may explain occasional blue-green stools at higher doses). Peak plasma concentration occurs 1–2 hours after an oral dose. The plasma half-life is about 5–6 hours, but tissue half-life (especially in brain and kidney, which accumulate MB) is longer — up to 24 hours.
Practical takeaway: a single morning dose provides cognitive effects throughout the working day; twice-daily dosing maintains higher steady-state concentrations but is rarely necessary at nootropic doses.
Timing and Co-Administration
- With or after food — reduces nausea; modest reduction in absorption rate but not in total bioavailability. Empty-stomach dosing produces faster, slightly higher peaks but more frequent GI upset.
- Morning dosing — aligns the cognitive effect with the working day; minimizes potential sleep disruption from MAO inhibition.
- Avoid evening dosing — some users report sleep disturbance, possibly from norepinephrine/serotonin elevation via MAO-A inhibition.
- With vitamin C — a common pairing; vitamin C reduces oxidized MB to leuco-MB, may improve tissue penetration and lower the effective oxidative burden. 500–1000 mg ascorbate is typical.
- Avoid concurrent serotonergics — mandatory; see Drug Interactions.
Vitamin C and Light Considerations
Vitamin C: co-administration of 500–1000 mg ascorbic acid is widely practiced. Mechanistically, ascorbate reduces blue oxidized MB to colorless leuco-MB, which appears to penetrate tissues more efficiently and is regenerated to MB intracellularly. Whether the synergy improves outcomes in humans is not yet rigorously demonstrated, but the pairing is biochemically plausible and well tolerated.
Light: visible red light (around 660 nm) excites MB to a triplet state that produces singlet oxygen. For purely cognitive use this is a downside — bright light during the peak window may shift MB toward pro-oxidant chemistry. Some practitioners pair MB with specific red-light therapy as a deliberate redox-cycling strategy; others recommend dosing in dim conditions and avoiding intense sun for an hour after a dose. The evidence base for either approach is anecdotal.
Weight-Based Calculation Examples
| Body Weight | Microdose (0.5 mg/kg) | Standard (1.5 mg/kg) | High Nootropic (3 mg/kg) |
|---|---|---|---|
| 50 kg (110 lb) | 25 mg | 75 mg | 150 mg |
| 60 kg (132 lb) | 30 mg | 90 mg | 180 mg |
| 70 kg (154 lb) | 35 mg | 105 mg | 210 mg |
| 80 kg (176 lb) | 40 mg | 120 mg | 240 mg |
| 90 kg (198 lb) | 45 mg | 135 mg | 270 mg |
| 100 kg (220 lb) | 50 mg | 150 mg | 300 mg |
USP-grade pharmaceutical methylene blue is most commonly available in 1% solutions (10 mg/mL) and 0.5% solutions (5 mg/mL), as well as in compounded capsules of 25 mg, 50 mg, or 100 mg. Drops are convenient for fine titration; capsules are convenient for daily-routine consistency.
Frequency and Cycling
The half-life is short enough that single daily morning dosing is the standard pattern. Several practical observations from clinical use:
- Five days on, two days off is a common cycling pattern; gives the body a rest from MAO inhibition
- Continuous daily dosing for > 2–4 weeks should prompt periodic reassessment of subjective benefit and any adverse effects
- Some users find an initial "novelty" phase of 2–3 weeks during which the cognitive lift is more pronounced; effects often plateau and may benefit from cycling
- Discontinuation does not require tapering; the half-life is short enough that any effects fade within days
Hard Contraindications Before Any Dose
Before any meaningful dose of methylene blue, particularly above microdose levels:
- Concurrent serotonergic medications — SSRIs, SNRIs, MAOIs, tramadol, fentanyl, dextromethorphan, lithium, triptans. See Drug Interactions. The washout requirement is at least 5 half-lives of the serotonergic drug (~5 weeks for fluoxetine) before initiating MB at any dose above ~1 mg/kg.
- Known or suspected G6PD deficiency — can precipitate severe hemolytic anemia. See G6PD page. Screening test is inexpensive; ask your doctor.
- Pregnancy — teratogenicity concerns; not recommended outside acute methemoglobinemia treatment.
- Severe renal impairment — methylene blue is partially renally cleared; accumulation possible.
- Significant hepatic impairment — MAO inhibition may be more pronounced.
- Industrial-grade or non-pharmaceutical source — see Pharmaceutical vs Industrial. Non-USP product can contain heavy metals and organic contaminants at levels unacceptable for human use.
Research Papers and References
- MB pharmacokinetics — PubMed search
- Rodriguez 2016 cognitive trial — PubMed search
- MB for methemoglobinemia — PubMed search
- MB in vasoplegic shock — PubMed search
- MB and vitamin C interaction — PubMed search
- MB in ifosfamide encephalopathy — PubMed search