Methylene Blue and Long COVID: Mitochondrial Dysfunction and Post-Viral Fatigue
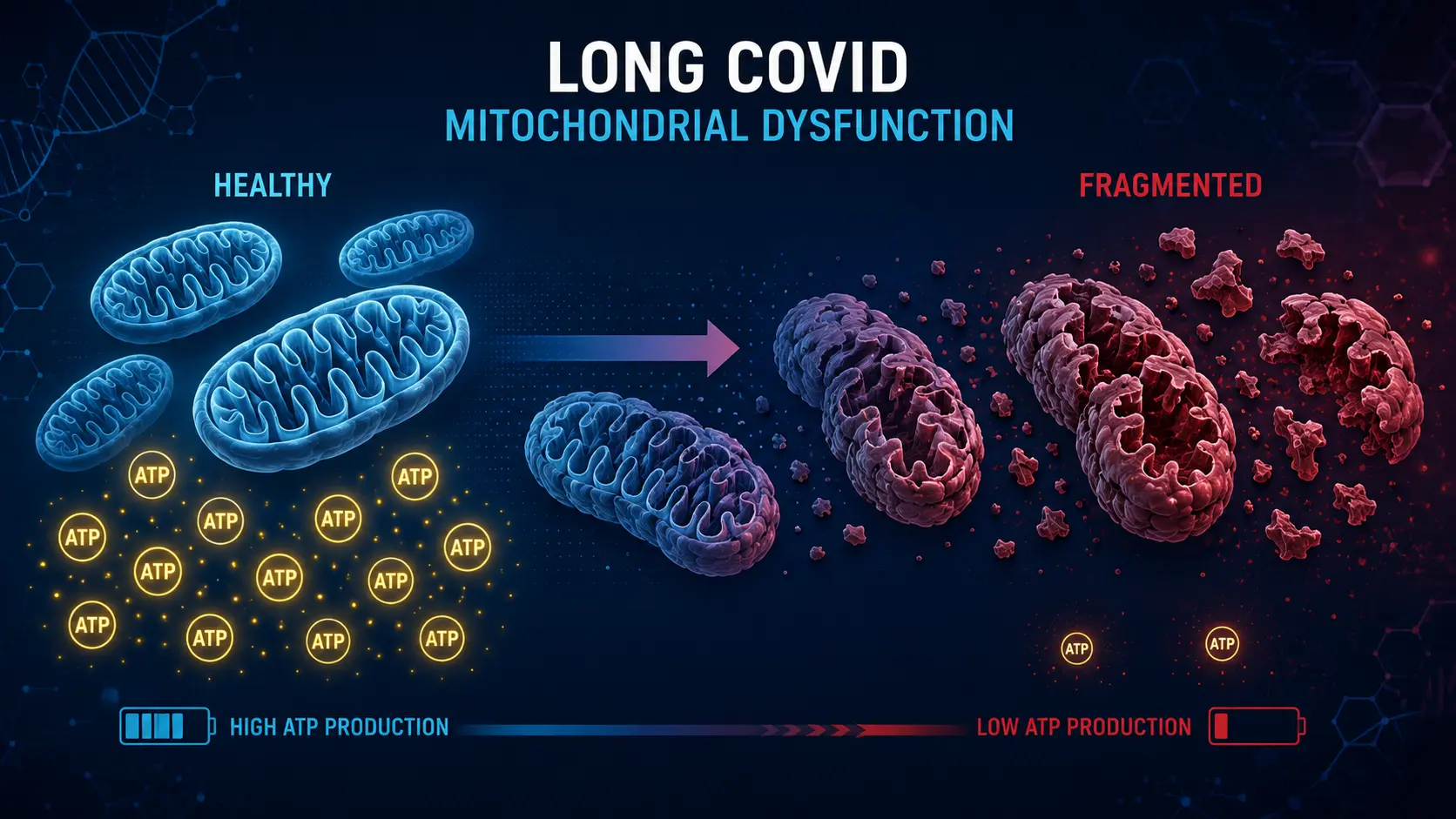
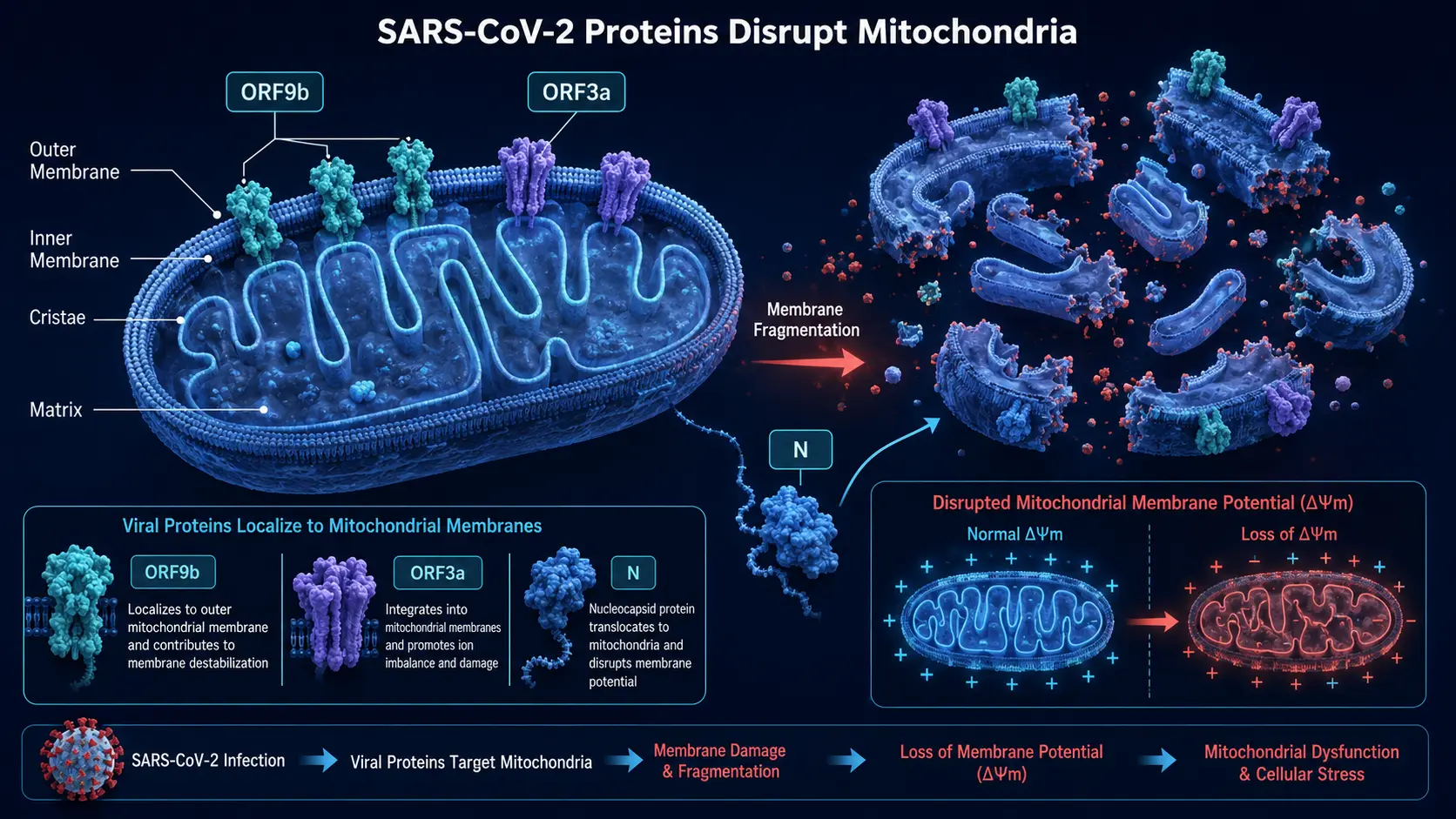
Long COVID is the umbrella term for the constellation of symptoms that persist for months or years after acute SARS-CoV-2 infection. Estimates of prevalence vary, but at least 5–10% of those infected develop persistent symptoms, with fatigue and cognitive impairment leading the list. The most consistent biological signature in long COVID research is mitochondrial dysfunction — reduced ATP production, elevated lactate, abnormal mitochondrial morphology on muscle biopsy, and impaired oxygen extraction during exercise. Because methylene blue's primary mechanism is mitochondrial support, it has become one of the most frequently discussed off-label interventions for post-COVID fatigue and cognitive impairment.
The evidence base is early and mostly clinician-level (case series, observational reports, mechanistic plausibility). This page reviews what is and isn't established, the dosing patterns being used, and how methylene blue fits into broader long COVID protocols.
Table of Contents
- What Long COVID Is
- The Mitochondrial Signature
- Why Methylene Blue Specifically
- Dosing Protocols in Use
- Combination Approaches
- What the Evidence Shows (and Doesn't)
- Overlap with ME/CFS
- Caveats and Cautions
- Research Papers and References
- Connections
What Long COVID Is
Long COVID (also called post-acute sequelae of SARS-CoV-2, PASC) is defined by WHO as symptoms persisting at least 3 months after acute infection that cannot be explained by an alternative diagnosis. The most common symptoms:
- Fatigue (especially post-exertional malaise — symptoms worsen 24–72 hours after exertion)
- Cognitive impairment ("brain fog," word-finding difficulty, slow processing)
- Dysautonomia / POTS (orthostatic intolerance, racing heart on standing)
- Persistent shortness of breath
- Sleep disturbance
- Muscle and joint pain
- Loss of taste and smell
- Persistent or recurrent low-grade fever
- New-onset food sensitivities, mast-cell activation symptoms (hives, flushing)
The clinical heterogeneity has frustrated single-cause hypotheses. Current research suggests several overlapping mechanisms: persistent viral reservoirs, autoimmunity (against ACE2 and other targets), microclots, vagal nerve injury, gut microbiome disruption, and mitochondrial dysfunction. These are not mutually exclusive; most patients likely have several of them simultaneously.
The Mitochondrial Signature
Multiple studies have documented mitochondrial abnormalities in long COVID:
- Reduced ATP production capacity in peripheral blood mononuclear cells (PBMCs)
- Reduced cytochrome c oxidase activity in muscle biopsies
- Abnormal mitochondrial morphology (swelling, fragmentation) on electron microscopy
- Elevated lactate at rest and exaggerated lactate response to exercise
- Reduced oxygen extraction during cardiopulmonary exercise testing
- Persistent transcriptional signatures of mitochondrial stress in PBMCs
- Reduced NAD+ levels and altered NAD+/NADH ratios
Mechanistically, SARS-CoV-2 proteins directly interact with host mitochondrial machinery: ORF9b localizes to mitochondria; ORF3a fragments the mitochondrial network; nucleocapsid disrupts membrane potential. The persistence of these effects post-infection is thought to drive a substantial portion of long COVID symptoms.
Why Methylene Blue Specifically
If long COVID involves persistent mitochondrial dysfunction, then any intervention that can restore mitochondrial function should in principle help. Methylene blue's mechanism makes it an obvious candidate:
- The electron-transport-chain bypass restores ATP production in cells with impaired Complex I or III — precisely the defect documented in long COVID
- Brain penetration is high — relevant for cognitive symptoms
- NOS inhibition may help with the vasoplegic/POTS-like component in some patients
- Antioxidant effects at low doses may reduce the oxidative stress that perpetuates mitochondrial damage
- Anti-inflammatory effects at the cellular level
Other mitochondrial supports being tried in long COVID protocols include CoQ10, alpha-lipoic acid, NAD+ precursors (NMN, NR), creatine, L-carnitine, magnesium, and various B vitamins. Methylene blue is unique in directly bypassing impaired ETC complexes.
Dosing Protocols in Use
Several clinicians treating long COVID have published or shared protocols using methylene blue. Common patterns:
- Low-dose start: 0.5–1 mg/kg/day for 1–2 weeks, assessing tolerance and any subjective change
- Maintenance: 1–2 mg/kg/day for 4–12 weeks, often combined with vitamin C and other mitochondrial supports
- Cycling: 5 days on, 2 days off; or 3 weeks on, 1 week off, depending on practitioner preference
- Combination with red-light therapy: some protocols use 660 nm light therapy 1–2 hours after MB dosing, leveraging photodynamic effects
- Discontinuation criteria: no benefit by 8–12 weeks at therapeutic dose, intolerable side effects, or development of contraindicating condition
These are not standardized protocols backed by randomized trials — they are practitioner-level approaches that have spread through the long COVID treatment community.
Combination Approaches
Methylene blue is rarely used alone in long COVID protocols. Common companions:
- NAC (N-acetylcysteine) — glutathione precursor; addresses oxidative stress and microclot dissolution; see NAC page
- CoQ10 (ubiquinol) — supports the electron transport chain at Complex III
- NAD+ precursors — nicotinamide riboside or NMN; replenish the substrate methylene blue accepts electrons from
- Low-dose naltrexone (LDN) — addresses neuroinflammation; see LDN page
- Nattokinase — addresses microclot hypothesis; see Nattokinase page
- Vitamin D — immune modulation; see Vitamin D3 page
- Magnesium — cofactor for many mitochondrial enzymes
- Pacing and POTS management — non-pharmacologic foundations
What the Evidence Shows (and Doesn't)
What's established:
- Mitochondrial dysfunction is a documented feature of long COVID
- Methylene blue restores mitochondrial function in animal models of various impairments
- Methylene blue improves cognition and memory in healthy adults (Rodriguez 2016)
- Methylene blue is used safely in inpatient settings for FDA-approved indications
What's not established:
- No randomized controlled trial has tested methylene blue specifically for long COVID
- Optimal dose, duration, or patient selection criteria are unknown
- Whether methylene blue addresses other long COVID mechanisms (microclots, autoimmunity, viral persistence) is uncertain
- Long-term safety of months-long daily dosing in this population is undocumented
Several small clinical trials are reportedly in development. Until controlled data exist, methylene blue for long COVID is reasonable to discuss with a knowledgeable clinician but should not be self-administered without medical supervision.
Overlap with ME/CFS
Long COVID and Myalgic Encephalomyelitis / Chronic Fatigue Syndrome (ME/CFS) share many features — post-exertional malaise, cognitive impairment, autonomic dysfunction, sleep disturbance, and mitochondrial dysfunction on biopsy. Many long COVID patients meet ME/CFS diagnostic criteria. The interventions that have been tried in ME/CFS — including various mitochondrial supports — are largely the same ones now being tried in long COVID.
Methylene blue has been used in ME/CFS-treating clinics for years on similar mechanistic grounds. The evidence base for ME/CFS specifically is also limited but slightly longer in time horizon.
Caveats and Cautions
- SSRIs, SNRIs, MAOIs — many long COVID patients are on antidepressants for the depression and anxiety that often accompany the syndrome. These are absolute contraindications to therapeutic methylene blue. See Drug Interactions.
- Tramadol or fentanyl for post-COVID pain — same problem.
- Severe POTS or autonomic instability — methylene blue's vascular effects may not be predictable; start low.
- G6PD deficiency — screening is not routine and adds important safety information.
- Source quality — pharmaceutical USP grade only; see Pharmaceutical vs Industrial.
- Realistic timeline — subjective changes may take 4–8 weeks; long COVID itself often improves slowly with or without intervention
Research Papers and References
- Long COVID and mitochondrial dysfunction — PubMed search
- SARS-CoV-2 mitochondrial proteins — PubMed search
- Long COVID post-exertional malaise — PubMed search
- ME/CFS mitochondrial biopsies — PubMed search
- MB and post-viral fatigue — PubMed search
- Long COVID treatment reviews — PubMed search
- NAC and microclots in long COVID — PubMed search