CAC, Statins, and the Density Paradox: How Calcium Reshapes Cholesterol Decisions
The single most consequential clinical use of the coronary calcium score is in reshaping the statin decision. CAC turns the abstract "10-year ASCVD risk percentage" into a concrete picture of accumulated plaque, and that visibility changes the conversation: it can confirm the need for therapy, defer it for years, or escalate it past guideline-default intensity. This page covers the guideline thresholds, the trial evidence behind them, and the most counterintuitive finding in the field: statins make your calcium score go up, even as they reduce your risk of dying from a heart attack.
Table of Contents
- Guideline Thresholds for Statin Recommendation
- Deferring Statins with CAC = 0
- The CAC ≥100 Threshold
- High CAC: Aggressive Therapy
- SCOT-HEART Trial
- ROBINSCA Trial
- The Density Paradox
- Progression on Statins
- Beyond Statins
- Research Papers and References
- Connections
Guideline Thresholds for Statin Recommendation
The 2018 ACC/AHA cholesterol guideline (and subsequent updates) explicitly incorporate CAC into the statin decision pathway for primary prevention. The relevant cutoffs:
- CAC = 0 — Reasonable to defer statin therapy in adults at intermediate ASCVD risk (5–19.9%) without diabetes, smoking, or family history of premature CAD
- CAC 1–99 — Statin reasonable, especially in those at higher percentiles for age
- CAC ≥100 OR ≥75th percentile — Statin recommended regardless of LDL-C level
- CAC ≥300 — High-intensity statin; consider aspirin if not contraindicated; aggressive risk-factor management
Different societies have slightly different thresholds. The European ESC guideline emphasizes percentile rather than absolute number. The American College of Cardiology favors the absolute ≥100 cutoff for simplicity.
Deferring Statins with CAC = 0
The deferral pathway is one of the strongest reasons to obtain a CAC. Patients with intermediate ASCVD risk but a calcium score of zero have 10-year event rates so low that the absolute benefit of statin therapy becomes very small — the number-needed-to-treat over a decade is in the 200–400 range, comparable to many other interventions whose risk/benefit balance favors deferral.
Practical interpretation: a 60-year-old with LDL-C 145, no diabetes, no smoking, no family history, and a CAC of 0 has <1% 10-year event risk. Standard pooled-cohort risk calculation might suggest 8–10% risk and trigger a statin recommendation. The CAC = 0 result reasonably defers that recommendation with a re-evaluation timeline of 5–10 years. See the CAC = 0 page.
The CAC ≥100 Threshold
A CAC score of 100 or above triggers statin recommendation in most current guidelines, regardless of LDL-C level or pooled-cohort risk score. The reasoning:
- 10-year event rates approach or exceed 7.5% at CAC ≥100, meeting threshold for statin benefit
- The presence of mature plaque means the lipoprotein-driven atherogenic process has advanced; further LDL exposure compounds the existing burden
- Statin therapy reduces 10-year MI and CV death by 25–35% in this population
- The number-needed-to-treat over a decade falls to ~30–50, a clearly favorable risk/benefit balance
This threshold has shifted clinical practice meaningfully — many patients who would not have qualified for statins under older LDL-C-only criteria now qualify based on calcium burden, and many patients who would have been pressured into therapy on borderline calculator results are now reasonably watched.
High CAC: Aggressive Therapy
For CAC ≥300, especially ≥1000 or top-percentile-for-age:
- High-intensity statin (atorvastatin 40–80 mg or rosuvastatin 20–40 mg) — aim for LDL-C <70 mg/dL or 50% reduction
- Consider ezetimibe if LDL-C target not met on statin alone
- Consider PCSK9 inhibitors in genetic dyslipidemia or persistent very-high LDL despite statin + ezetimibe
- Aspirin 81 mg daily often appropriate (after weighing bleeding risk), especially in high-risk patterns
- Tight blood pressure control — goal <130/80
- Diabetes screening and management — SGLT2 inhibitors and GLP-1 agonists if diabetic
- Stress test or CCTA if symptoms emerge or as a one-time anatomy/ischemia evaluation
- Cardiology consultation at very high scores
SCOT-HEART Trial
The SCOT-HEART trial (Lancet, 2018) randomized 4,146 Scottish patients with stable chest pain to standard care vs CCTA-guided care. Five-year results:
- CCTA-guided arm: 41% relative reduction in death from CHD or non-fatal MI (HR 0.59, 95% CI 0.41–0.84)
- The benefit was driven by more accurate diagnosis and earlier statin initiation in the CCTA arm
- The trial demonstrated that imaging-guided care, beyond just CAC, can reduce hard endpoints
SCOT-HEART used CCTA, not pure CAC, but the principle — that visualization of plaque changes management in a way that improves outcomes — underlies the use of CAC in primary prevention as well.
ROBINSCA Trial
ROBINSCA (Risk Or Benefit IN Screening for CArdiovascular disease) was a large Dutch trial randomizing asymptomatic adults at moderate baseline risk to no screening, ASCVD-risk-score-based management, or CAC-based management. Findings:
- CAC-based screening identified more high-risk patients earlier than ASCVD-risk-only management
- Patients with elevated CAC who started statins earlier had better long-term cardiovascular outcomes
- The trial supported population-level CAC screening for asymptomatic adults at intermediate baseline risk
ROBINSCA, alongside SCOT-HEART, has been influential in moving CAC from "interesting research test" into mainstream primary prevention practice.
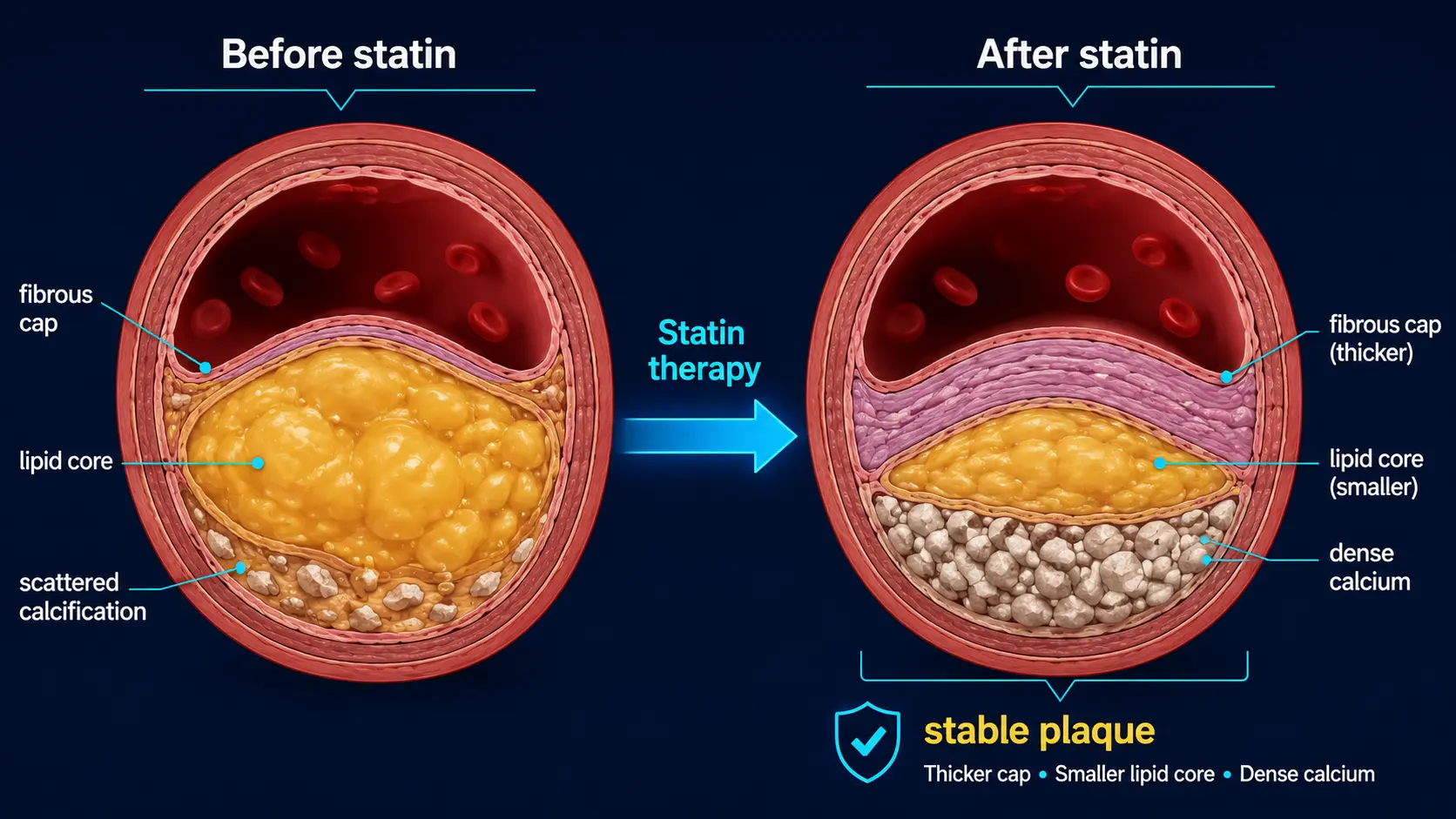
The Density Paradox
Here is the most counterintuitive finding in CAC research: statins make CAC scores go up.
Multiple randomized trials and registries have shown that patients on statins have higher calcium scores at follow-up than statin-naïve controls, despite having lower rates of cardiovascular events. The Agatston score rises (or progresses faster) on statin therapy by an average of 10–20% per year, more than the natural progression rate.
The mechanism is plaque stabilization:
- Statins reduce lipid accumulation in plaques and reduce inflammatory drive
- Stable plaques are more calcified than unstable plaques
- The body deposits calcium into existing lipid-rich plaque cores as part of the fibro-calcific stabilization process
- The Agatston score, which weights heavily toward dense calcium, captures this stabilization as an apparent "progression"
- Fewer plaque ruptures occur because the stabilized, denser plaque is less vulnerable
This is why a CAC score that has risen on statin therapy is not a signal that the medication is failing — it is, in many cases, a signal that the medication is working. Calcium volume scoring (rather than Agatston) tracks burden more directly and is sometimes preferred for follow-up. Either way, treatment decisions should be based on event risk and clinical context, not on a rising follow-up Agatston score in a statin-treated patient.
Progression on Statins
Practical interpretation of follow-up scans on statin therapy:
- Modest annual rise in Agatston score (10–20% per year) is expected; not a failure of therapy
- Stable or slowly-rising volume score is more reassuring than rising Agatston score because it isn't density-weighted
- Rapid increase (>30–40% per year) warrants reassessment of LDL/ApoB targets, medication adherence, lifestyle factors
- New high-risk vessel patterns (Left Main, proximal LAD) at follow-up may warrant additional imaging or stress testing
- Symptom development always overrides imaging trends — new chest pain, dyspnea, or exertional intolerance warrants prompt evaluation
The main reason to obtain a follow-up CAC on statin therapy is for patient motivation and adherence, not for treatment titration. A patient seeing their score rise from 200 to 300 over five years on a statin can be counter-intuitively reassured that this represents stabilization — if the message is delivered correctly. A patient who never gets a follow-up scan misses the opportunity for that conversation.
Beyond Statins
For patients with high CAC who tolerate or fail statin therapy:
- Ezetimibe — reduces intestinal cholesterol absorption; modest LDL reduction; good safety profile
- PCSK9 inhibitors (alirocumab, evolocumab) — potent LDL reduction (~50% on top of statin); injectable
- Bempedoic acid — pre-statin pathway inhibitor; useful in statin-intolerant patients
- Inclisiran — siRNA against PCSK9; twice-yearly injection
- Lp(a)-targeted therapies — in trials; pelacarsen and others may be available in coming years
- Aspirin 81 mg — consider if no bleeding contraindication, especially at very high CAC scores
- Lifestyle: see Reversal & Lifestyle
Research Papers and References
- ACC/AHA cholesterol guideline & CAC — PubMed search
- SCOT-HEART — PubMed search
- ROBINSCA — PubMed search
- Statin density paradox — PubMed search
- PCSK9 inhibitor outcomes — PubMed search
- Lp(a)-targeted therapy — PubMed search
Connections
- Coronary Calcium Score Deep-Dive Articles:
- CAC Overview
- Agatston Calculation
- MESA Calculator
- CAC = 0
- CAC vs Other Tests
- Soft Plaque Limits
- Reversal & Lifestyle
- ApoB
- Lipoprotein(a)
- Lipid Panel
- Cardiology