Riboflavin and Migraine Prevention
Migraine is a complex neurovascular disorder affecting approximately 12% of the global population, with attacks characterized by recurrent episodes of moderate to severe headache, often accompanied by nausea, photophobia, and phonophobia. Prophylactic treatment aims to reduce the frequency, severity, and duration of attacks. Among nutritional approaches to migraine prevention, riboflavin (vitamin B2) has emerged as one of the most well-studied and promising options, supported by clinical trial evidence and a compelling mechanistic rationale rooted in mitochondrial bioenergetics.
Table of Contents
- Key Benefits at a Glance
- Mitochondrial Energy Dysfunction in Migraine
- FAD and FMN Cofactor Roles
- Clinical Trials: The 400 mg/day Evidence
- Mechanism of Action
- Riboflavin vs. Conventional Prophylaxis
- Safety Profile
- Dosing Recommendations
- Combined Nutrient Approaches
- Research Papers
- Connections
- Featured Videos
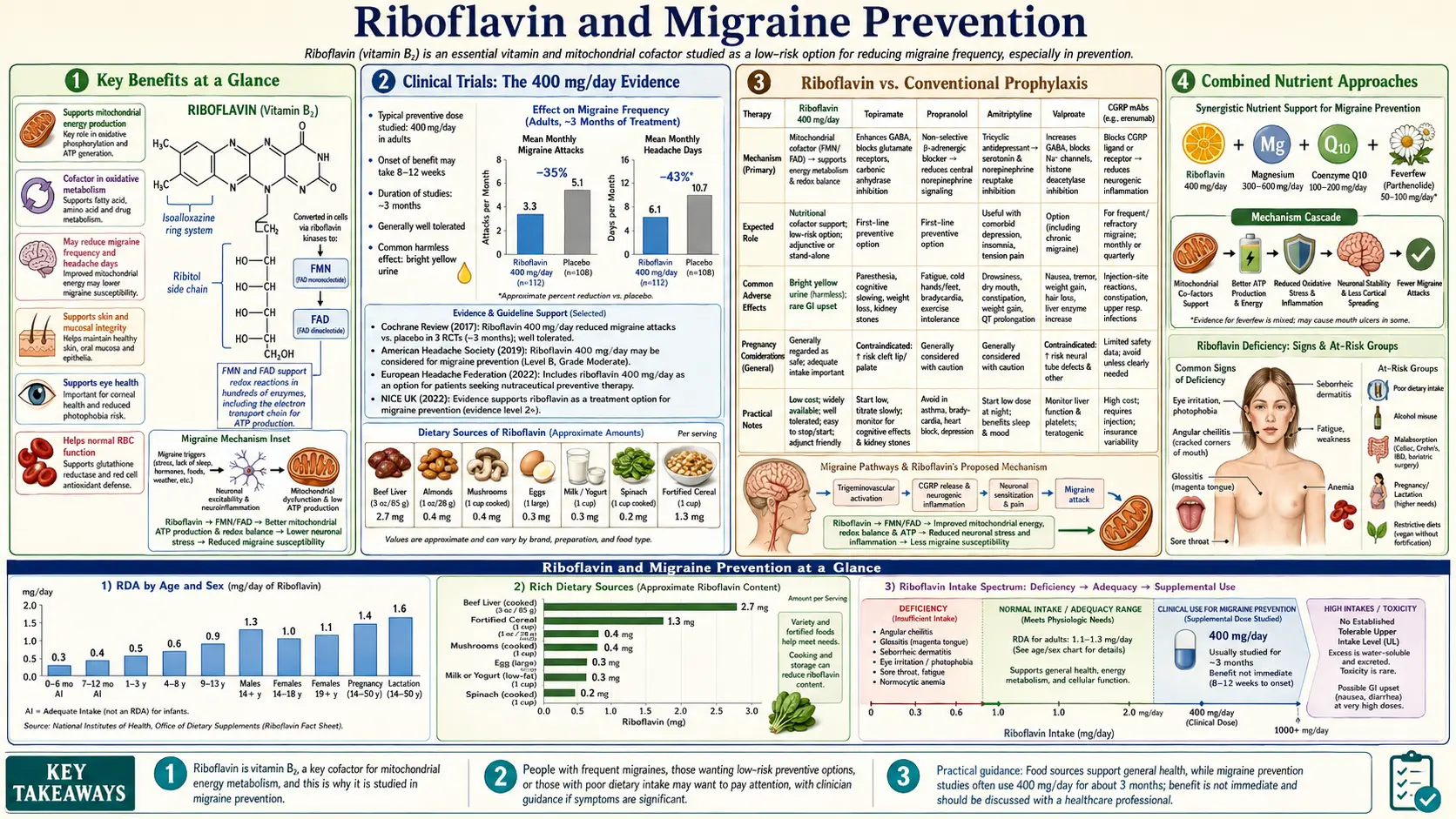
Key Benefits at a Glance
- ~50% reduction in migraine frequency at 400 mg/day (Schoenen 1998 RCT).
- Responder rate ~59% vs. 15% placebo — comparable to beta-blockers and topiramate.
- Restores cortical mitochondrial energy reserves documented on 31P-MRS imaging.
- No meaningful side effects apart from harmless yellow-fluorescent urine.
- Pregnancy Category A — safe for pregnant migraineurs.
- No drug interactions with triptans, contraceptives, or other common medications.
- Raises the cortical spreading depression (CSD) threshold, reducing aura frequency.
- Synergistic with CoQ10 and magnesium in multi-nutrient prophylaxis.
Mitochondrial Energy Dysfunction in Migraine
A growing body of evidence supports the hypothesis that impaired mitochondrial energy metabolism plays a central role in migraine pathophysiology:
- Brain energy reserve deficit – Phosphorus-31 magnetic resonance spectroscopy (31P-MRS) studies have consistently demonstrated reduced brain energy reserves in migraineurs between attacks. Specifically, decreased phosphocreatine and increased inorganic phosphate and ADP levels indicate a state of chronic cerebral energy deficit in the cortex and occipital regions.
- Mitochondrial enzyme deficits – Studies of platelet mitochondria and muscle biopsy specimens from migraineurs have revealed reduced activity of electron transport chain complexes, particularly Complex I (NADH:ubiquinone oxidoreductase) and Complex II (succinate dehydrogenase), both of which require riboflavin-derived cofactors for their function.
- Cortical spreading depression – The migraine aura is believed to be caused by cortical spreading depression (CSD), a slowly propagating wave of neuronal depolarization followed by suppression. CSD is an energy-intensive process that places enormous demands on mitochondrial ATP production. Neurons with impaired mitochondrial function have a lower threshold for CSD initiation.
- Trigeminovascular activation – Mitochondrial dysfunction may sensitize the trigeminovascular system, the pain-signaling pathway responsible for migraine headache. Energy-depleted neurons and trigeminal nerve endings release calcitonin gene-related peptide (CGRP) and other neuropeptides that promote neurogenic inflammation and pain transmission.
- Genetic predisposition – Certain mitochondrial DNA polymorphisms and mutations are overrepresented in migraine populations. Maternal inheritance patterns of migraine (consistent with mitochondrial DNA transmission) further support a mitochondrial contribution to disease susceptibility.
FAD and FMN Cofactor Roles
Riboflavin exerts its biological effects primarily through its two coenzyme forms, flavin adenine dinucleotide (FAD) and flavin mononucleotide (FMN). These cofactors are essential for mitochondrial energy production:
- Complex I (NADH dehydrogenase) – This is the largest complex of the electron transport chain and the entry point for electrons from NADH. Complex I contains FMN as a prosthetic group and multiple iron-sulfur clusters. It transfers electrons to ubiquinone while pumping protons across the inner mitochondrial membrane. FMN is absolutely required for Complex I activity.
- Complex II (succinate dehydrogenase) – This enzyme complex participates in both the citric acid cycle and the electron transport chain. It contains a covalently bound FAD molecule that accepts electrons from succinate, converting it to fumarate. Complex II transfers these electrons to ubiquinone via iron-sulfur clusters.
- Electron transfer flavoprotein (ETF) – FAD-dependent ETF and ETF-ubiquinone oxidoreductase channel electrons from fatty acid beta-oxidation and amino acid catabolism into the electron transport chain, contributing to overall mitochondrial energy output.
- Dihydrolipoamide dehydrogenase – This FAD-dependent enzyme is a shared component of the pyruvate dehydrogenase, alpha-ketoglutarate dehydrogenase, and branched-chain keto acid dehydrogenase complexes, linking riboflavin status to the efficiency of the citric acid cycle.
Clinical Trials: The 400 mg/day Evidence
Several well-designed clinical trials have investigated riboflavin supplementation for migraine prophylaxis, with the most consistent evidence supporting a dose of 400 mg per day:
- Schoenen et al. (1998) – This landmark randomized, double-blind, placebo-controlled trial enrolled 55 migraineurs and demonstrated that 400 mg/day of riboflavin for 3 months reduced migraine frequency by 50% compared to a 15% reduction with placebo (p = 0.005). The proportion of patients achieving a 50% or greater reduction in attack frequency (the "responder rate") was 59% for riboflavin versus 15% for placebo.
- Boehnke et al. (2004) – An open-label study of 23 migraineurs treated with 400 mg/day riboflavin for 6 months showed significant reductions in headache frequency (from 4 to 2 days per month) and headache intensity. Notably, the study also demonstrated normalization of previously impaired cortical mitochondrial energy metabolism as measured by 31P-MRS, providing direct evidence for the bioenergetic mechanism of action.
- Condò et al. (2009) – A randomized trial comparing riboflavin (400 mg/day) to sodium valproate (500 mg twice daily) in 90 patients with migraine without aura found comparable efficacy between the two treatments in reducing migraine frequency and severity over 3 months, with riboflavin demonstrating significantly fewer adverse effects.
- Pediatric evidence – Studies in children and adolescents have yielded mixed results. MacLennan et al. (2008) found that 200 mg/day riboflavin was not superior to placebo in a pediatric RCT, though a high placebo response rate (typical of pediatric headache trials) may have obscured a treatment effect. Other open-label pediatric studies have reported positive outcomes at 200-400 mg/day.
- Onset of benefit – Across studies, the therapeutic benefit of riboflavin typically becomes apparent after 4-6 weeks of supplementation and reaches maximum effect by 3 months. This delayed onset is consistent with the time required for mitochondrial enzyme pools to reach new steady-state levels.
Mechanism of Action
The anti-migraine effect of riboflavin is believed to operate through several interconnected mechanisms:
- Enhanced mitochondrial electron transport – By increasing the availability of FAD and FMN, riboflavin supplementation at pharmacological doses (400 mg/day far exceeds the RDA of 1.1-1.3 mg) upregulates electron transport chain activity, increasing ATP production efficiency and restoring brain energy reserves.
- Increased CSD threshold – Improved mitochondrial function raises the threshold for cortical spreading depression, reducing the likelihood of migraine aura and the subsequent activation of pain pathways.
- Reduced oxidative stress – FAD is a cofactor for glutathione reductase, which regenerates reduced glutathione (GSH), the brain's primary intracellular antioxidant. Enhanced antioxidant defense protects neuronal mitochondria from oxidative damage that impairs their function.
- Anti-inflammatory effects – Improved mitochondrial function reduces the release of pro-inflammatory mediators from energy-stressed neurons and glial cells, potentially attenuating neurogenic inflammation in the trigeminovascular system.
Riboflavin vs. Conventional Prophylaxis
When compared to standard pharmacological migraine preventatives, riboflavin offers several distinct advantages and some limitations:
- Comparable efficacy – The responder rate for riboflavin (approximately 50-59% of patients achieving 50% reduction in attacks) is comparable to that of established prophylactic medications including beta-blockers (propranolol), anticonvulsants (topiramate, valproate), and tricyclic antidepressants (amitriptyline).
- Superior tolerability – Unlike conventional prophylactics, which commonly cause weight gain (valproate, amitriptyline), cognitive impairment (topiramate), fatigue (beta-blockers), or mood changes, riboflavin has virtually no significant side effects apart from harmless bright yellow discoloration of urine.
- No drug interactions – Riboflavin does not interact with triptans, analgesics, oral contraceptives, or other commonly used medications, making it suitable for patients on complex medication regimens.
- Pregnancy safety – Riboflavin is considered safe during pregnancy (Category A), unlike most conventional prophylactics, which are Category C or D. This makes it an attractive option for pregnant migraineurs who require preventive treatment.
- Limitations – The evidence base for riboflavin, while positive, is smaller than for conventional prophylactics. Riboflavin may be less effective for very high-frequency migraine or chronic daily headache, and the delayed onset of action (4-6 weeks) may be unsuitable for patients requiring rapid relief.
Safety Profile
Riboflavin has an excellent safety record, even at doses far exceeding the recommended dietary allowance:
- No established upper limit – The Institute of Medicine has not established a Tolerable Upper Intake Level (UL) for riboflavin due to the absence of evidence of toxicity at any dose studied. The limited intestinal absorption capacity for riboflavin and efficient renal excretion of excess amounts provide natural protection against accumulation.
- Bright yellow urine – The most noticeable effect of high-dose riboflavin supplementation is a vivid fluorescent yellow discoloration of urine, caused by the urinary excretion of unabsorbed and metabolized riboflavin. This is entirely harmless but should be explained to patients to prevent unnecessary concern.
- Gastrointestinal tolerance – Occasional mild gastrointestinal symptoms (diarrhea, increased urinary frequency) have been reported at high doses but are uncommon and generally self-limiting.
- Photosensitivity – Riboflavin is a photosensitizer and can generate reactive oxygen species when exposed to UV light. However, at oral supplementation doses, clinically significant photosensitivity has not been reported.
Dosing Recommendations
Based on the available clinical evidence, the following dosing guidelines are recommended for riboflavin in migraine prophylaxis:
- Standard adult dose – 400 mg per day, taken as a single dose or divided into two doses of 200 mg. This is the dose supported by the strongest clinical trial evidence.
- Timing – Riboflavin can be taken with or without food. Taking it with a meal may improve absorption, as dietary fat and slowed gastric emptying allow more time for the saturable intestinal transport mechanism to absorb the vitamin.
- Duration of trial – Patients should continue supplementation for a minimum of 3 months before assessing efficacy, as the full benefit typically requires this duration to manifest. Premature discontinuation is a common reason for perceived treatment failure.
- Pediatric dosing – For children and adolescents, doses of 200-400 mg/day have been used in studies, though evidence is less robust. Consultation with a pediatric neurologist is advisable.
- Maintenance therapy – For patients who respond to riboflavin, continued supplementation is recommended. Discontinuation may result in a gradual return of previous migraine frequency over weeks to months.
Combined Nutrient Approaches
Riboflavin is often used in combination with other evidence-based nutrients for enhanced migraine prophylaxis:
- Coenzyme Q10 (CoQ10) – Like riboflavin, CoQ10 supports mitochondrial electron transport chain function. It acts as an electron carrier between Complexes I/II and Complex III. Doses of 100-300 mg/day have shown migraine-preventive efficacy in clinical trials. The combination of riboflavin and CoQ10 addresses multiple points in the electron transport chain simultaneously.
- Magnesium – Magnesium is involved in over 300 enzymatic reactions, including mitochondrial ATP production, and helps regulate neuronal excitability and neurotransmitter release. Supplementation with 400-600 mg/day of magnesium (as citrate, glycinate, or threonate) has demonstrated efficacy for migraine prevention and pairs well with riboflavin.
- Feverfew (Tanacetum parthenium) – This botanical agent has anti-inflammatory and serotonin-modulating properties. Combined with riboflavin and magnesium, it forms part of several commercially available migraine-prevention formulations.
- Multi-nutrient formulations – Several studies have evaluated combination products containing riboflavin, CoQ10, magnesium, and sometimes additional ingredients. While these combination studies make it difficult to attribute benefit to any single component, they generally show positive results for migraine frequency reduction with excellent tolerability.
Riboflavin represents a safe, well-tolerated, and evidence-based option for migraine prevention that is particularly suitable for patients who prefer non-pharmaceutical approaches, are intolerant of conventional prophylactics, or require treatment during pregnancy. Its mechanism of action through mitochondrial bioenergetic enhancement provides a sound scientific rationale, and the clinical trial evidence supports its inclusion in migraine management guidelines from major headache societies. Patients should be counseled about the expected 4-6 week delay before clinical benefit becomes apparent and encouraged to maintain supplementation for at least 3 months before evaluating efficacy.
Research Papers
- Schoenen J, Jacquy J, Lenaerts M. Effectiveness of high-dose riboflavin in migraine prophylaxis. A randomized controlled trial. Neurology. 1998;50(2):466-470.
- Boehnke C, et al. High-dose riboflavin treatment is efficacious in migraine prophylaxis: an open study in a tertiary care centre. Eur J Neurol. 2004;11(7):475-477.
- Condò M, et al. Riboflavin prophylaxis in pediatric and adolescent migraine. (PubMed topic search)
- MacLennan SC, et al. High-dose riboflavin for migraine prophylaxis in children: a double-blind, randomized, placebo-controlled trial. (PubMed topic search)
- PubMed — Coenzyme Q10 for migraine prophylaxis (topic search)
- PubMed — Magnesium in migraine prophylaxis (topic search)
- PubMed — Mitochondrial dysfunction in migraine (topic search)
- PubMed — Cortical spreading depression in migraine (topic search)
- PubMed — Riboflavin for migraine meta-analyses (topic search)
- NIH Office of Dietary Supplements — Riboflavin Fact Sheet for Health Professionals
- Linus Pauling Institute — Riboflavin
- Harvard T.H. Chan School of Public Health — Riboflavin (Vitamin B2)
Connections
- Vitamin B2
- Riboflavin and Energy Production
- Migraine
- Migraine (Pain & Allergy)
- Magnesium
- Magnesium and Heart Health
- Vitamin B6
- Vitamin B12
- Headache
- Magnesium Replenishment
- Depression
- Brain Fog
- Oxidative Stress
- Fatigue
- Magnesium Riboflavin and Supplements