Riboflavin and Cellular Energy Production
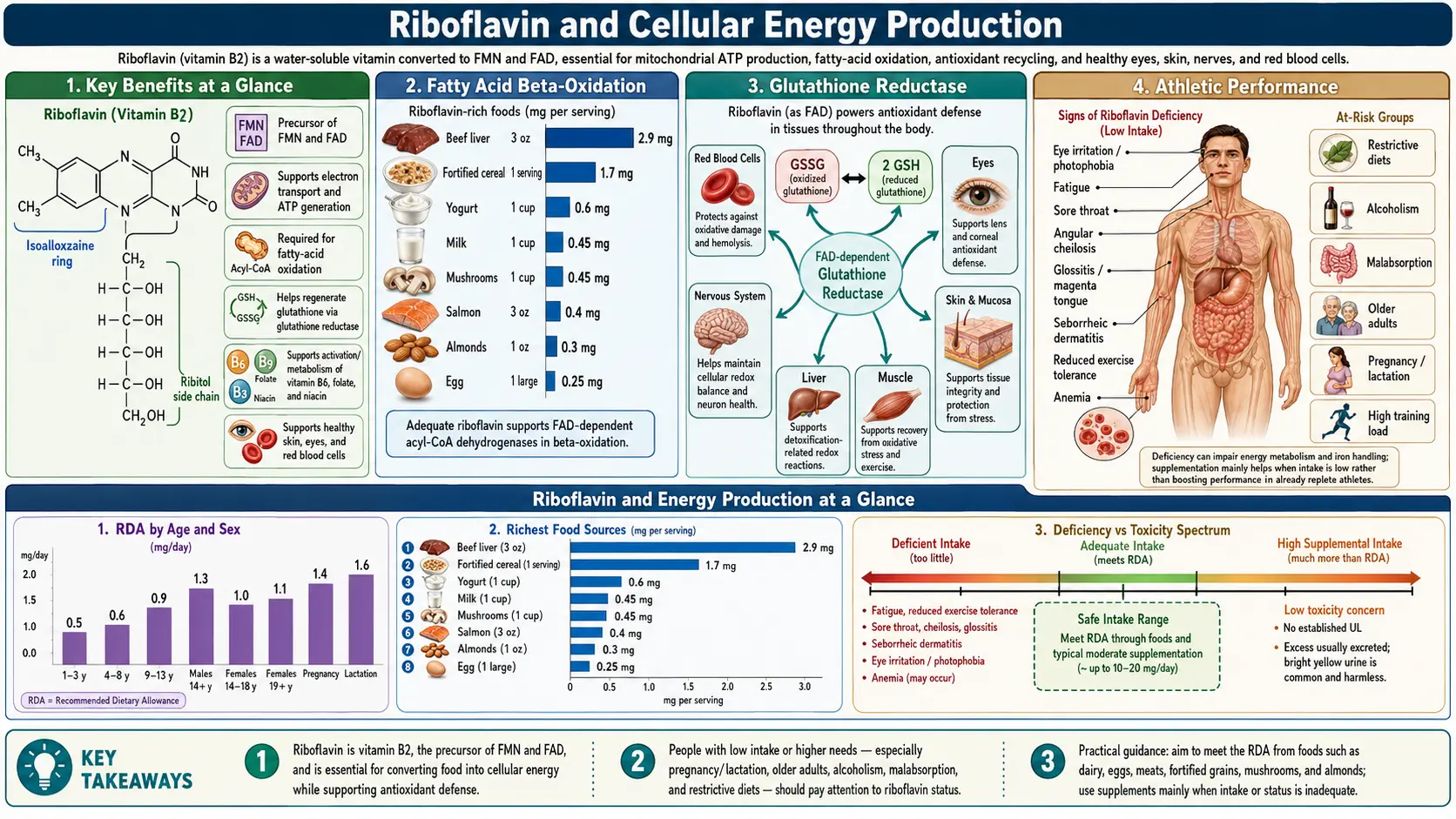
Riboflavin (vitamin B2) occupies a central position in cellular energy metabolism as the precursor to two essential coenzymes: flavin adenine dinucleotide (FAD) and flavin mononucleotide (FMN). These flavocoenzymes participate in a vast array of oxidation-reduction reactions throughout the cell, serving as electron carriers in pathways that ultimately generate adenosine triphosphate (ATP), the universal currency of cellular energy. Without adequate riboflavin, the fundamental metabolic machinery that powers every cell in the body would grind to a halt.
Table of Contents
- Key Benefits at a Glance
- FAD and FMN Coenzymes
- Electron Transport Chain: Complex I and Complex II
- Fatty Acid Beta-Oxidation
- Krebs Cycle: Succinate Dehydrogenase
- Glutathione Reductase
- Drug Metabolism
- Deficiency Symptoms: Ariboflavinosis
- Athletic Performance
- Research Papers
- Connections
- Featured Videos
Key Benefits at a Glance
- Precursor to FAD and FMN, essential cofactors in 90+ flavoproteins.
- Central to oxidative phosphorylation via Complex I (FMN) and Complex II (FAD).
- Required for fatty acid beta-oxidation (acyl-CoA dehydrogenases, ETF, ETF-QO).
- Supports the citric acid cycle through succinate dehydrogenase and shared E3 subunits.
- Regenerates reduced glutathione via FAD-dependent glutathione reductase.
- Activates other B-vitamins (B6, B3, folate) through flavin-dependent enzymes.
- Supports drug metabolism via cytochrome P450 reductase and FMOs.
- Elevated exercise requirement — athletes need 20–60% more.
FAD and FMN Coenzymes
Riboflavin is converted to its active coenzyme forms through sequential enzymatic reactions:
- FMN synthesis – Riboflavin kinase (also called flavokinase) phosphorylates riboflavin at its ribityl side chain to produce flavin mononucleotide (FMN). This reaction requires ATP as the phosphate donor and is the rate-limiting step in flavocoenzyme synthesis. FMN retains the isoalloxazine ring system that is responsible for the electron-accepting properties of flavocoenzymes.
- FAD synthesis – FAD synthetase (also called FMN adenylyltransferase) catalyzes the adenylation of FMN with ATP to produce FAD. This reaction adds an adenosine monophosphate (AMP) moiety to FMN, creating the larger dinucleotide structure. FAD is the predominant flavocoenzyme in the body, accounting for approximately 75-90% of total tissue flavins.
- Redox chemistry – Both FAD and FMN function by accepting and donating electrons through their isoalloxazine ring. They can accept either one electron (forming a semiquinone radical intermediate) or two electrons (forming the fully reduced FADH2 or FMNH2). This versatility allows them to participate in both one-electron and two-electron transfer reactions, bridging different types of redox chemistry within the cell.
- Flavoprotein diversity – Over 90 flavoproteins have been identified in mammalian tissues, catalyzing reactions in energy metabolism, amino acid catabolism, fatty acid oxidation, purine degradation, vitamin metabolism (including activation of vitamins B6, B3, and folate), and xenobiotic detoxification.
Electron Transport Chain: Complex I and Complex II
The mitochondrial electron transport chain (ETC) is the final common pathway for aerobic ATP generation, producing approximately 90% of cellular ATP through oxidative phosphorylation. Riboflavin-derived coenzymes are essential components of the first two complexes:
- Complex I (NADH:ubiquinone oxidoreductase) – The largest complex of the ETC, containing 45 subunits in mammals with a total molecular mass of approximately 1,000 kDa. Complex I contains one molecule of FMN as its primary electron acceptor. NADH generated by the citric acid cycle and other metabolic pathways donates its electrons to FMN, which then passes them through a series of seven iron-sulfur clusters to ubiquinone (coenzyme Q10). This electron transfer is coupled to the translocation of four protons across the inner mitochondrial membrane, contributing to the proton motive force that drives ATP synthase.
- Complex II (succinate:ubiquinone oxidoreductase) – Also known as succinate dehydrogenase (SDH), Complex II is unique among ETC complexes in that it participates in both the citric acid cycle and the electron transport chain. It contains one covalently bound FAD molecule that accepts electrons from succinate as it is oxidized to fumarate. These electrons are then transferred through three iron-sulfur clusters to ubiquinone. Unlike Complex I, Complex II does not pump protons across the membrane, which is why FADH2-linked substrates yield fewer ATP molecules than NADH-linked substrates.
- Overall ATP yield – The complete oxidation of one molecule of glucose through glycolysis, the citric acid cycle, and oxidative phosphorylation generates approximately 30-32 molecules of ATP in mammalian cells. Electrons entering the ETC through Complex I (via NADH) contribute approximately 2.5 ATP per pair, while those entering through Complex II (via FADH2) contribute approximately 1.5 ATP per pair.
Fatty Acid Beta-Oxidation
Fatty acid beta-oxidation is the primary pathway for catabolism of fatty acids, generating acetyl-CoA for the citric acid cycle and reduced coenzymes (NADH and FADH2) for the electron transport chain. Multiple FAD-dependent enzymes are required for this process:
- Acyl-CoA dehydrogenases – The first step of each beta-oxidation cycle is catalyzed by one of several chain-length-specific acyl-CoA dehydrogenases: very long-chain (VLCAD), long-chain (LCAD), medium-chain (MCAD), and short-chain (SCAD). All of these enzymes use FAD as a prosthetic group to catalyze the formation of a trans-double bond between the alpha and beta carbons of the fatty acyl-CoA substrate.
- Electron transfer flavoprotein (ETF) – The FADH2 generated by acyl-CoA dehydrogenases does not donate its electrons directly to ubiquinone. Instead, electrons are transferred to electron transfer flavoprotein (ETF), which itself contains FAD. ETF then passes the electrons to ETF-ubiquinone oxidoreductase (ETF-QO), another FAD-containing enzyme embedded in the inner mitochondrial membrane, which reduces ubiquinone and feeds electrons into the ETC.
- Energy yield from fatty acids – Complete oxidation of one molecule of palmitate (a 16-carbon saturated fatty acid) yields 106 ATP molecules, making fatty acids the most energy-dense macronutrient. This high yield depends on the proper functioning of multiple FAD-dependent enzymes at each cycle of beta-oxidation.
- Deficiency consequences – Impaired FAD availability due to riboflavin deficiency can result in decreased fatty acid oxidation capacity. This may manifest clinically as an inability to tolerate fasting, hypoglycemia, elevated plasma free fatty acids and acylcarnitines, and in severe cases, a metabolic phenotype resembling multiple acyl-CoA dehydrogenase deficiency (MADD or glutaric aciduria type II).
Krebs Cycle: Succinate Dehydrogenase
The citric acid cycle (Krebs cycle or tricarboxylic acid cycle) is the central metabolic hub that oxidizes acetyl-CoA derived from carbohydrates, fats, and proteins. Riboflavin participates in this pathway through several mechanisms:
- Succinate dehydrogenase (SDH) – As noted above, SDH (Complex II) is the only enzyme that participates in both the citric acid cycle and the electron transport chain. Its FAD prosthetic group is covalently linked to a histidine residue of the SDHA subunit, making it one of the few flavoproteins where FAD is permanently attached rather than dissociable. The oxidation of succinate to fumarate generates FADH2, whose electrons are channeled directly into the ETC.
- Alpha-ketoglutarate dehydrogenase – This multi-enzyme complex contains a FAD-dependent dihydrolipoamide dehydrogenase (E3) subunit that reoxidizes dihydrolipoamide, generating NADH in the process. The same E3 subunit is shared with pyruvate dehydrogenase and branched-chain keto acid dehydrogenase complexes.
- Regulation of cycle flux – The overall flux through the citric acid cycle is sensitive to the NAD+/NADH and FAD/FADH2 ratios, which reflect cellular energy charge. Adequate riboflavin status ensures sufficient FAD availability to maintain optimal cycle activity, particularly under conditions of high energy demand such as exercise, fever, or rapid growth.
Glutathione Reductase
Beyond energy production, FAD plays a critical role in cellular antioxidant defense through the glutathione system:
- Glutathione reductase (GR) – This FAD-dependent enzyme catalyzes the NADPH-dependent reduction of oxidized glutathione (GSSG) back to reduced glutathione (GSH). GSH is the most abundant intracellular thiol antioxidant, present at millimolar concentrations in most cells. It directly scavenges reactive oxygen species, detoxifies hydrogen peroxide (via glutathione peroxidase), and conjugates with xenobiotics and electrophilic metabolites (via glutathione S-transferases).
- Erythrocyte glutathione reductase (EGR) activity coefficient – The EGR activity coefficient is the standard functional biomarker for assessing riboflavin status. It measures the ratio of GR activity with and without added FAD in vitro. An activity coefficient above 1.2-1.4 indicates riboflavin deficiency, as it demonstrates that GR in the red blood cells is not fully saturated with its FAD cofactor.
- Oxidative stress protection – Adequate riboflavin status is essential for maintaining the GSH/GSSG ratio that protects cells from oxidative damage. Riboflavin deficiency leads to depletion of reduced glutathione, increased lipid peroxidation, protein oxidation, and DNA damage from unchecked reactive oxygen species.
- Interaction with selenium – Glutathione peroxidase, the selenium-dependent enzyme that uses GSH to detoxify hydrogen peroxide and lipid hydroperoxides, depends on continuous GSH regeneration by FAD-dependent glutathione reductase. Thus, riboflavin deficiency can functionally impair selenium-dependent antioxidant defense even when selenium status is adequate.
Drug Metabolism
Flavocoenzymes participate in the metabolism of drugs and xenobiotics through several enzyme systems:
- Cytochrome P450 reductase – This FAD- and FMN-containing enzyme is the obligate electron donor for all microsomal cytochrome P450 enzymes. It transfers electrons from NADPH, first to FAD, then to FMN, and finally to the heme iron of P450. The cytochrome P450 superfamily metabolizes the majority of pharmaceutical drugs, as well as environmental toxins, carcinogens, and endogenous compounds including steroid hormones and bile acids.
- Flavin-containing monooxygenases (FMOs) – These FAD-dependent enzymes catalyze the oxygenation of nucleophilic nitrogen and sulfur atoms in drugs and xenobiotics. FMO3, the predominant hepatic isoform, metabolizes drugs such as ranitidine, cimetidine, and tamoxifen, as well as dietary compounds like trimethylamine.
- Aldehyde oxidase and xanthine oxidase – These molybdoflavoproteins contain FAD and participate in the metabolism of various drugs (methotrexate, famciclovir) and endogenous purines (hypoxanthine, xanthine). Xanthine oxidase also generates superoxide radical as a byproduct, linking flavoprotein activity to oxidative stress.
- Clinical implications – Riboflavin deficiency may alter drug pharmacokinetics by impairing P450-mediated and FMO-mediated metabolism. This could lead to altered drug efficacy or toxicity, though clinical evidence for this effect in humans is still limited.
Deficiency Symptoms: Ariboflavinosis
Riboflavin deficiency (ariboflavinosis) produces a characteristic constellation of clinical signs affecting primarily the skin, mucous membranes, and eyes:
- Angular cheilitis (stomatitis) – Painful cracks, fissures, and ulceration at the corners of the mouth are among the earliest and most characteristic signs of riboflavin deficiency. The lesions result from impaired epithelial cell turnover and reduced antioxidant protection at these mechanically stressed sites.
- Glossitis – The tongue becomes swollen, red (magenta-colored), and painful. The filiform papillae atrophy, giving the tongue a smooth, shiny appearance. In chronic deficiency, the tongue may appear furrowed and fissured.
- Seborrheic dermatitis – Greasy, scaly, erythematous skin lesions develop in the nasolabial folds, on the alae nasi, behind the ears, and on the scrotum or vulva. These lesions reflect impaired fatty acid metabolism and epithelial maintenance in sebaceous gland-rich areas.
- Ocular symptoms – Photophobia (light sensitivity), excessive lacrimation, burning and itching of the eyes, and corneal vascularization (ingrowth of blood vessels into the normally avascular cornea) occur in riboflavin deficiency. Prolonged deficiency can lead to corneal opacification and visual impairment.
- Normochromic normocytic anemia – Riboflavin deficiency can cause a pure red cell aplasia or normocytic anemia through impaired iron mobilization and utilization. FAD-dependent ferredoxin reductase is involved in iron metabolism, and riboflavin deficiency impairs iron absorption and its incorporation into hemoglobin.
- Neuropathy – Peripheral neuropathy with paresthesias and diminished tactile sensitivity can occur in severe riboflavin deficiency, reflecting impaired myelin synthesis and maintenance due to inadequate fatty acid metabolism.
Athletic Performance
Given riboflavin's central role in energy production, its relationship to physical performance and exercise capacity has been an area of active investigation:
- Increased requirements in athletes – Exercise increases the metabolic demand for flavocoenzymes due to elevated mitochondrial electron transport activity, increased fatty acid oxidation, and greater antioxidant requirements to manage exercise-induced oxidative stress. Studies have shown that riboflavin requirements may increase by 20-60% in individuals engaged in regular vigorous exercise.
- Exercise performance studies – Controlled supplementation studies have demonstrated that correcting marginal riboflavin deficiency improves exercise tolerance, reduces perceived exertion, and enhances aerobic capacity as measured by VO2 max. However, supplementation beyond sufficiency does not appear to provide additional ergogenic benefits.
- Recovery and adaptation – Post-exercise recovery involves repair of oxidative damage to mitochondrial membranes and proteins. FAD-dependent glutathione reductase activity is critical for regenerating the reduced glutathione needed for this repair process. Athletes with suboptimal riboflavin status may experience prolonged recovery times and increased markers of oxidative stress.
- Female athletes – Several studies have identified female athletes, particularly those with restrictive dietary patterns or low energy availability, as being at heightened risk for marginal riboflavin deficiency. The combination of increased metabolic demands from training and potentially inadequate dietary intake creates a vulnerability that may impair both performance and health.
- Dietary sources for athletes – Athletes can meet their elevated riboflavin needs through dairy products (milk, yogurt, cheese), eggs, lean meats, green leafy vegetables, and fortified cereals. Vegans and those avoiding dairy should pay particular attention to riboflavin intake, as animal products are among the richest dietary sources.
Riboflavin's role as the precursor to FAD and FMN places it at the very heart of cellular energy metabolism. From the electron transport chain to fatty acid oxidation, from the citric acid cycle to antioxidant defense, flavocoenzymes are indispensable participants in the biochemical reactions that sustain life. Ensuring adequate riboflavin intake through a balanced diet or targeted supplementation supports optimal mitochondrial function, protects against oxidative damage, and maintains the metabolic capacity needed for health and physical performance across all stages of life.
Research Papers
- PubMed — Riboflavin, FAD, FMN, and mitochondrial function (topic search)
- PubMed — Complex I FMN and electron transport (topic search)
- PubMed — Succinate dehydrogenase FAD flavoprotein (topic search)
- PubMed — Acyl-CoA dehydrogenase and fatty acid oxidation (topic search)
- PubMed — Glutathione reductase and riboflavin status (topic search)
- PubMed — Riboflavin deficiency (ariboflavinosis) (topic search)
- PubMed — Riboflavin and exercise performance (topic search)
- PubMed — EGR activity coefficient riboflavin status (topic search)
- NIH Office of Dietary Supplements — Riboflavin Fact Sheet for Health Professionals
- Linus Pauling Institute — Riboflavin
- Harvard T.H. Chan School of Public Health — Riboflavin (Vitamin B2)
Connections
- Vitamin B2
- Riboflavin and Migraine Prevention
- Vitamin B1
- Vitamin B3
- Vitamin B6
- Vitamin B12
- Iron
- Selenium
- Glutathione
- Oxidative Stress
- Anemia
- Fatigue
- Peripheral Neuropathy
- Milk
- Eggs
- Pellagra and Niacin Deficiency