Natamycin (E235): The Antibiotic Preservative on Your Cheese
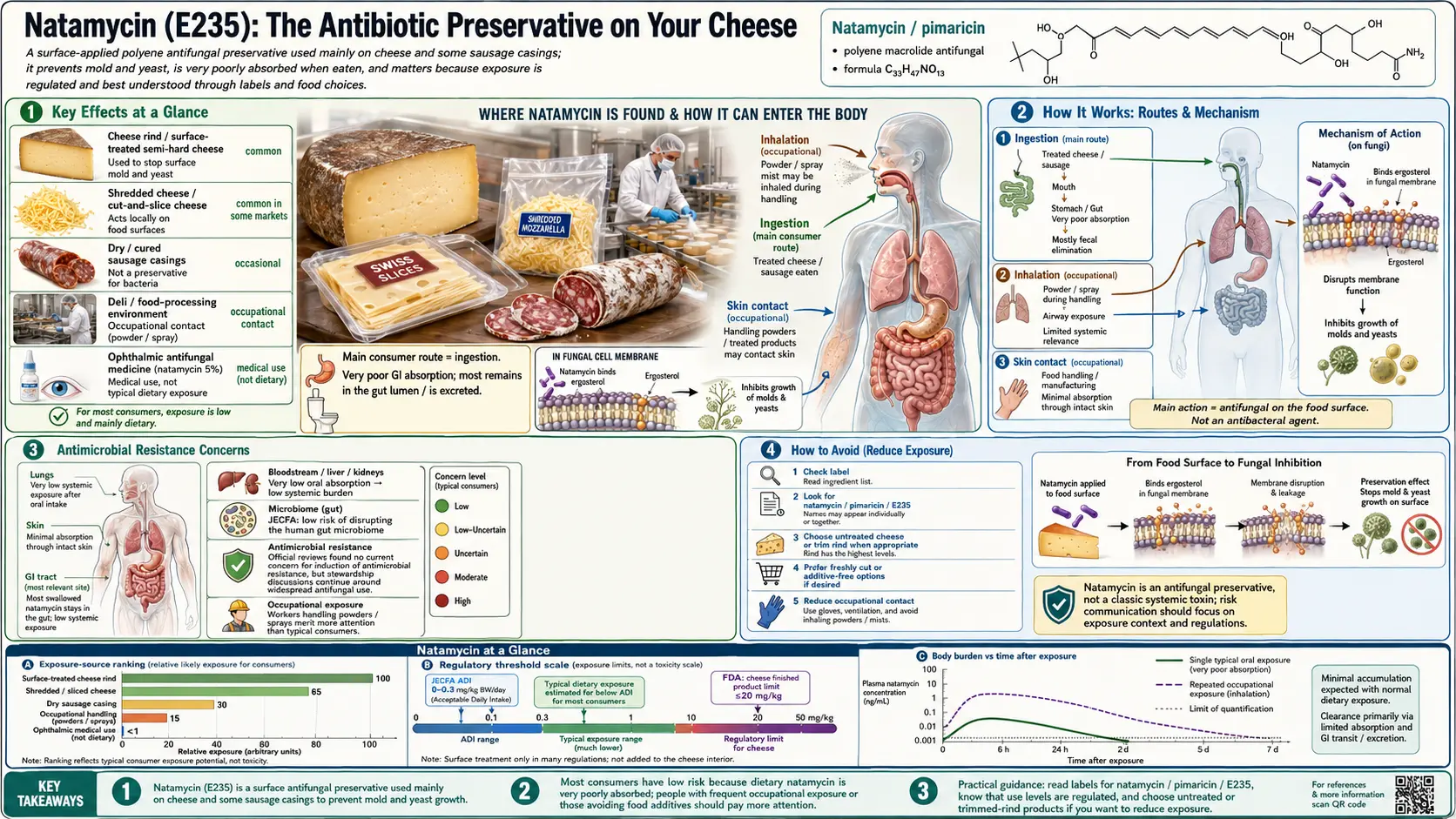
Table of Contents
- Key Effects at a Glance
- Overview
- Where It's Found (Exposure Routes)
- How It Works
- Health Effects
- Antimicrobial Resistance Concerns
- Regulatory Status & Safety Thresholds
- Organic Food Controversy
- How to Avoid
- Research Papers
- Connections
- Featured Videos
Key Effects at a Glance
- Antifungal antibiotic food additive – Polyene macrolide from Streptomyces natalensis; ergosterol-binding mechanism shared with amphotericin B.
- Low systemic toxicity – Oral bioavailability <1%; passes through GI tract largely intact.
- Antimicrobial resistance – Resistance documented in Aspergillus, Penicillium, and Fusarium isolates; potential cross-resistance with clinical amphotericin B.
- Gut mycobiome concern – Theoretical suppression of commensal yeasts, though clinical data are limited.
- Occupational allergy – Contact urticaria reported in cheese-industry workers.
- ADI – JECFA/EFSA 0–0.3 mg/kg body weight/day.
1. Overview
Natamycin (E235), also known as pimaricin, is a polyene macrolide antifungal antibiotic produced naturally by the soil bacterium Streptomyces natalensis. It occupies a unique and somewhat paradoxical position in the food additive landscape: it is simultaneously a medically important antibiotic used to treat human fungal eye infections, and a routine food preservative applied to the surfaces of cheeses and cured meats consumed daily by millions of people.
Natamycin was discovered in 1955 by researchers at the Gist-Brocades laboratory in the Netherlands, isolated from soil samples collected in the Natal province of South Africa — hence both its common names. Commercial production involves fermentation of S. natalensis in industrial-scale bioreactors, followed by purification. The compound has been used in food preservation since the 1960s.
What distinguishes natamycin from most food preservatives is its dual regulatory identity: it is approved as a food additive (E235) under food law, while simultaneously being classified as an antibiotic under pharmaceutical law. This duality has made it a focal point in global debates about antibiotic use in food systems and the risk of fostering antimicrobial resistance.
2. Where It's Found
Natamycin's food applications are concentrated in products where surface mold control is critical:
- Cheese (primary application): Applied to the surfaces of natural cheeses — including shredded, sliced, and block formats — and to cheese rinds to prevent mold growth during aging and retail storage. This is by far the largest volume use of natamycin in food.
- Cured and processed meats: Applied to sausage casings and the surfaces of cured meats such as dry-cured hams and salamis to inhibit surface mold during the drying and curing process.
- Yogurt and sour cream: Used in some markets to extend shelf life by preventing yeast and mold growth at the container surface.
- Beverages: Permitted in certain fruit-based beverages in some jurisdictions, though this is less common than cheese applications.
- Wine: Used in some wine-producing countries to prevent secondary fermentation and mold contamination, though this use is not universally approved.
In non-food applications, natamycin is a clinically important drug: Natacyn (natamycin ophthalmic suspension 5%) is the only FDA-approved topical antifungal for fungal keratitis (eye infections), prescribed for conditions caused by Fusarium and Candida species. This clinical use makes resistance development in food contexts a matter of direct medical concern.
3. How It Works
Natamycin's mechanism of action is shared with the medically critical antifungal amphotericin B — a fact that has major implications for antimicrobial resistance:
- Ergosterol binding: Natamycin specifically targets ergosterol, the principal sterol in fungal cell membranes (equivalent to cholesterol in mammalian cells). It binds with high affinity to ergosterol molecules, physically disrupting the membrane's structural integrity.
- Pore formation: The natamycin-ergosterol complexes aggregate to form transmembrane pores, causing leakage of essential ions and small molecules from fungal cells, ultimately leading to cell death.
- Fungal specificity: Because human cells use cholesterol rather than ergosterol in their membranes, natamycin exhibits very high selectivity for fungal cells. It does not inhibit bacteria at food-relevant concentrations, meaning it has no effect on bacterial spoilage or foodborne pathogens such as Salmonella or Listeria.
A critical feature of natamycin in food applications is its limited penetration depth. When applied to the surface of cheese or cured meat, natamycin diffuses only 1–2 mm into the food matrix. This surface-limited activity is why it is approved exclusively for surface treatments, not as a bulk food ingredient. It effectively creates an antifungal barrier at the product exterior while leaving the interior unaffected — a consideration relevant both to its technological function and to exposure assessment.
4. Health Effects
The direct toxicological profile of natamycin is considered favorable by regulatory agencies:
- Very low oral toxicity: The LD50 in rats exceeds 2,730 mg/kg (oral). Natamycin is poorly absorbed from the gastrointestinal tract, with bioavailability estimated at less than 1%. It passes through the GI tract largely intact and is excreted without significant systemic uptake.
- Carcinogenicity and mutagenicity: Standard battery toxicological tests have not identified carcinogenic or mutagenic potential. EFSA's 2009 re-evaluation found no genotoxic concern based on available data.
- Allergic reactions: Rare but documented. Hypersensitivity reactions, including contact urticaria, have been reported in occupational settings (workers in cheese manufacturing facilities) and in occasional food consumers. The clinical significance at typical dietary exposure is considered low.
The more nuanced health concern relates to the gut mycobiome — the community of fungi residing in the human gastrointestinal tract:
- The gut mycobiome includes species such as Saccharomyces cerevisiae, Candida spp., and Malassezia spp. that play roles in gut homeostasis and interaction with the bacterial microbiome.
- Natamycin's antifungal activity, even at residual dietary concentrations, could theoretically disrupt the mycobiome by killing susceptible beneficial yeasts. However, given the very low oral bioavailability, significant luminal concentrations may not persist far into the GI tract.
- Controlled human studies on natamycin's effect on the gut mycobiome are lacking, representing a meaningful gap in the evidence base.
5. Antimicrobial Resistance Concerns
The antimicrobial resistance (AMR) implications of natamycin's use in food represent the most significant and contested dimension of its safety profile. The core concern is straightforward: natamycin is both a food preservative and a medically important antibiotic. Using antibiotics at sub-therapeutic concentrations in food — exactly what surface application to cheese represents — creates selection pressure for resistant organisms.
Documented Resistance
Natamycin resistance is no longer hypothetical. Resistance has been demonstrated in multiple clinically and agriculturally relevant fungal species:
- Aspergillus species resistant to natamycin have been isolated from cheese production environments, with resistance linked to mutations in the ergosterol biosynthesis pathway (Ballmann et al., 2015).
- Laboratory induction of natamycin resistance is achievable in multiple Aspergillus, Penicillium, and Fusarium species through serial passage in sub-inhibitory concentrations.
Cross-Resistance with Clinical Antifungals
Because natamycin and amphotericin B share the same primary mechanism — ergosterol binding — resistance mutations affecting the ergosterol biosynthesis pathway can confer cross-resistance to both compounds. Amphotericin B is a last-resort antifungal for life-threatening infections such as invasive aspergillosis and cryptococcal meningitis. Cross-resistance pathways therefore carry direct clinical implications.
WHO and FAO Positions
The World Health Organization's list of critically important antimicrobials for human medicine (CIA list, 6th revision, 2019) includes polyene antifungals. The WHO has expressed general concern about antibiotic use in food production, though natamycin as a food preservative has not been subject to a formal WHO restriction recommendation as of this writing.
Within the European Union, the broader debate over antibiotic use in animal agriculture and food production has intensified since the 2019 EU Regulation on Veterinary Medicinal Products (2019/6), which restricts prophylactic and metaphylactic antibiotic use. Some scientific commentators have argued that natamycin's food use should be brought under a similar framework, though it currently sits outside veterinary medicine regulation.
The Food-as-AMR-Vector Problem
Even if the absolute quantities of natamycin in food are small, the scale of exposure is enormous: hundreds of millions of people consume natamycin-treated cheese regularly. The selection pressure exerted on fungi in food environments — including ambient fungi that could enter human environments — is difficult to quantify but likely non-trivial. This systemic exposure differentiates food-additive antibiotic use from therapeutic use, where exposure is time-limited and in a clinical context.
6. Regulatory Status
Natamycin's regulatory treatment varies significantly between jurisdictions, reflecting differing risk tolerance regarding antibiotic food additives:
- European Union: Approved as E235 under Regulation (EC) No 1333/2008, but with strict use limitations. Permitted only on the surface of hard, semi-hard, and semi-soft cheeses, and on the surface of dried cured sausages. Maximum permitted level: 1 mg/dm² of surface area, with no detectable residue at a depth of 5 mm. Not permitted as a bulk food additive.
- United States (FDA): Approved as GRAS for use in or on specified foods. The FDA permits broader applications than the EU, including use in shredded cheese, cottage cheese, sour cream, and certain other dairy products. Listed in 21 CFR 172.155.
- Codex Alimentarius: The Codex General Standard for Food Additives sets a maximum residue limit of 40 mg/kg for natamycin on cheese surfaces (as a surface treatment). Codex standards influence national regulations in countries lacking independent review capacity.
- Banned or restricted uses: Several countries restrict natamycin use more stringently than the US or Codex. In Denmark and some other EU member states, historical national restrictions were harmonized upward following EU-wide approval, generating domestic controversy. China permits natamycin in specified fermented foods at defined limits.
7. Organic Food Controversy
Natamycin is not permitted in USDA certified organic foods, nor in EU organic production (Regulation (EC) No 834/2007 and implementing regulations). This stands in contrast to potassium sorbate, which is permitted in USDA organic products — an anomaly that some organic food advocates argue should be reversed given potassium sorbate's genotoxicity concerns.
The exclusion of natamycin from organic certification stems from its classification as an antibiotic. Organic standards consistently prohibit routine antibiotic use to maintain the integrity of organic claims and avoid contributing to AMR from organic production systems.
However, a labeling ambiguity sometimes arises in practice: conventional cheeses treated with natamycin are sometimes marketed with terms like "naturally made" or "no artificial preservatives," since natamycin is derived from a natural microbial fermentation process. This marketing language can mislead consumers who associate "natural" with antibiotic-free. Regulatory bodies have not systematically addressed this labeling gap.
Consumers seeking cheese without natamycin should look for USDA Certified Organic or EU Organic labels, or purchase from producers who explicitly state their preservation methods — including artisanal and farmstead cheese producers who rely on controlled aging conditions rather than surface antimicrobials.
8. How to Avoid
- Choose USDA Organic or EU Organic cheeses – both standards prohibit natamycin.
- Read labels for “natamycin,” “pimaricin,” or “E235.”
- Purchase from farmstead/artisanal producers who rely on controlled aging and cave/cellar conditions rather than surface antimicrobials.
- Cut and discard the rind of non-organic hard cheeses where natamycin is surface-applied and penetrates only 1–2 mm.
- Avoid shredded/sliced factory cheese in shrink-wrap packaging, which frequently receives natamycin anti-caking and anti-mold treatments.
Connections
- Preservatives
- Potassium Sorbate
- Nisin
- Sodium Nitrite
- BHA and BHT
- US vs EU Food Safety
- Banned Additives
- Food Additives
- Food Dyes Banned Worldwide
- Cancer
- Gut-Brain Axis
- Probiotics
- Gut Microbiome
- Fermented Foods
- Yogurt
- Meningitis
- Staphylococcus Aureus
Research Papers
- te Welscher YM, ten Napel HH, Balague MM, Souza CM, Riezman H, de Kruijff B, Breukink E. Natamycin blocks fungal growth by binding specifically to ergosterol without permeabilizing the membrane. Journal of Biological Chemistry. 2008;283(10):6393–6401.
- EFSA Panel on Food Additives and Nutrient Sources. Scientific Opinion on the re-evaluation of natamycin (E 235) as a food additive. EFSA Journal. 2009;7(12):1412.
- Ballmann R, Vödisch M, Weiss E, Würfl E, Brakhage AA, Kniemeyer O. Natamycin resistance in food-contaminating Aspergillus species correlates with specific changes in ergosterol biosynthesis. International Journal of Food Microbiology. 2015;205:30–38.
- Raab WP. Natamycin (pimaricin). Its properties and possibilities in medicine. Georg Thieme Verlag. Stuttgart; 1972.
- WHO. Critically Important Antimicrobials for Human Medicine, 6th Revision. World Health Organization; 2019. ISBN 978-92-4-151552-8.
- van Rij G, Speekenbrink AB, Ringens PJ, Beekhuis WH, van Bijsterveld OP, Pels E, Eggink CA. Natamycin in the treatment of fungal keratitis. A retrospective study of 22 patients in the Netherlands. Documenta Ophthalmologica. 1993;83(3):181–191.
- Stark J. Permitted preservatives — natamycin. In: Russell NJ, Gould GW, eds. Food Preservatives. 2nd ed. Kluwer Academic/Plenum Publishers; 2003:91–102.
- Ghannoum MA, Rice LB. Antifungal agents: mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clinical Microbiology Reviews. 1999;12(4):501–517.
- European Commission. Regulation (EC) No 1333/2008 of the European Parliament and of the Council on food additives. Official Journal of the European Union. 2008;L354:16–33.
- FDA. Code of Federal Regulations, Title 21, Part 172.155: Natamycin. U.S. Food and Drug Administration. 2023.
- Codex Alimentarius Commission. General Standard for Food Additives (CODEX STAN 192-1995, amended 2023). FAO/WHO; 2023.
- Ellepola AN, Morrison CJ. Laboratory diagnosis of invasive candidiasis. Journal of Microbiology. 2005;43(Spec):65–84.
- Meis JF, Chowdhary A, Rhodes JL, Fisher MC, Verweij PE. Clinical implications of globally emerging azole resistance in Aspergillus fumigatus. Philosophical Transactions of the Royal Society B. 2016;371(1709):20150460.
- Lund F, Frisvad JC. Occurrence of natural mold inhibitors — a survey. Journal of Food Protection. 1994;57(5):440–446.