Nisin (E234): The Bacterial Weapon in Your Food
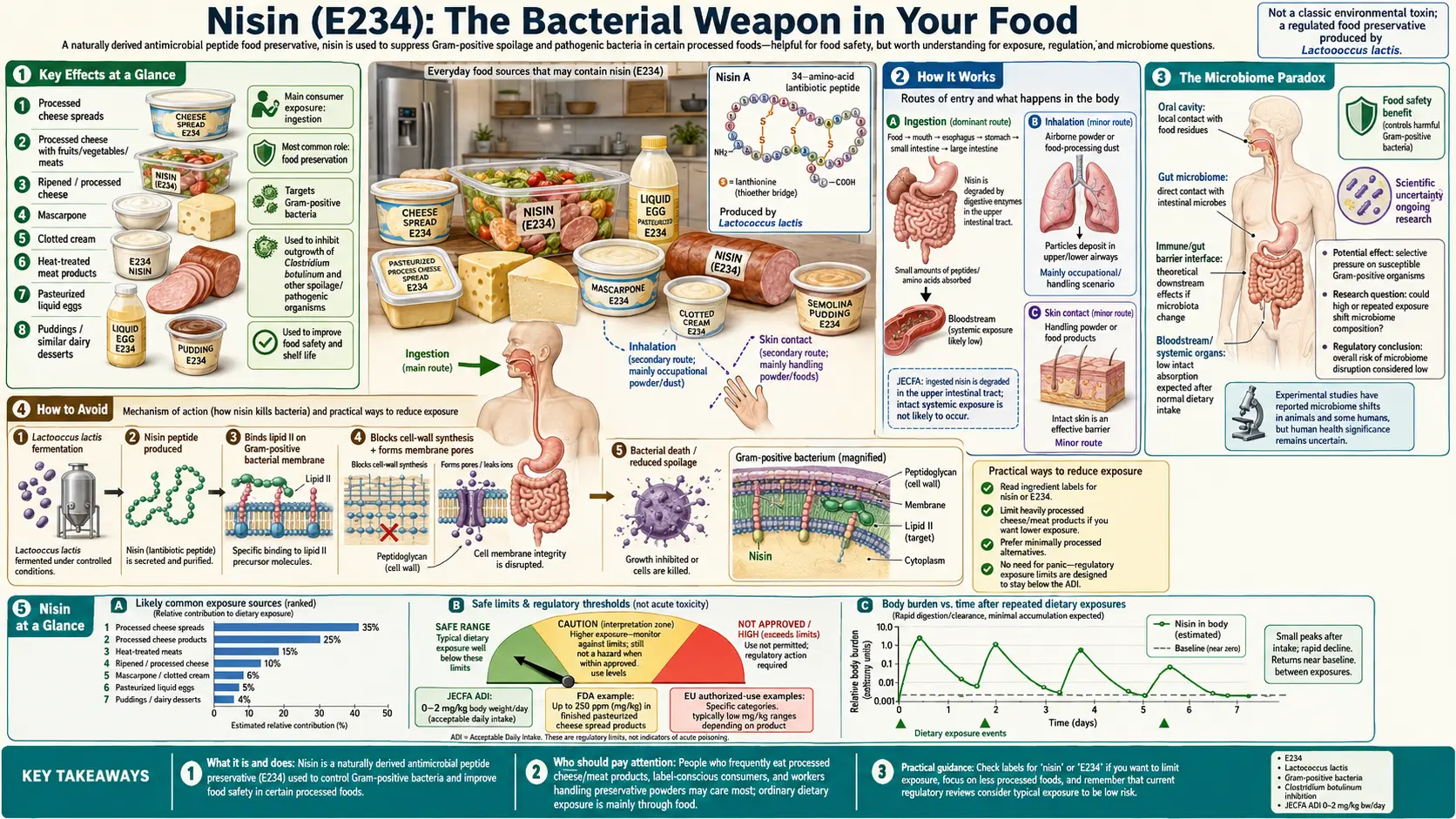
Table of Contents
- Key Effects at a Glance
- Overview
- Where It's Found (Exposure Routes)
- How It Works
- Health Effects
- Antimicrobial Resistance
- The Microbiome Paradox
- Regulatory Status & Safety Thresholds
- Emerging Research
- How to Avoid
- Research Papers
- Connections
- Featured Videos
Key Effects at a Glance
- Antibiotic food additive – A lantibiotic that targets Lipid II in Gram-positive bacteria.
- Digestive inactivation – Rapidly hydrolyzed by chymotrypsin; negligible systemic absorption.
- Gut microbiome disruption – Inhibits Lactobacillus and Bifidobacterium in the upper GI tract before full proteolytic inactivation.
- Antimicrobial resistance concerns – Nisin-resistant Listeria strains documented in food-processing environments; potential cross-resistance with vancomycin pathways.
- No known carcinogenicity – JECFA and EFSA evaluations found no chronic toxicity at relevant doses.
- ADI – JECFA 0–33,000 IU/kg body weight (~1.25 mg pure nisin/kg/day).
1. Overview
Nisin (E234) is a lantibiotic — a class I bacteriocin — produced naturally by the lactic acid bacterium Lactococcus lactis subsp. lactis. It is the only bacteriocin currently approved as a food preservative worldwide, a distinction it has held since the 1950s. The commercial product is sold under the trade name Nisaplin (a standardized preparation containing approximately 2.5% pure nisin in a sodium chloride and defatted milk solids matrix).
Nisin was first described in scientific literature in 1928 by Rogers and Whittier, who observed that certain strains of L. lactis — naturally present in dairy fermentation — produced a substance that inhibited the growth of competing bacteria during cheese making. The compound was named "nisin" from N-Inhibitory Substance. Systematic study of its structure and mechanism advanced considerably in the following decades, with its complete chemical characterization achieved in the 1970s and its three-dimensional structure resolved in the 1990s.
Nisin's approval as a food additive reflects its natural origin (produced by a bacterium used in fermentation for millennia), its peptide nature (making it susceptible to digestion), its narrow antibacterial spectrum, and its long history of incidental human consumption through fermented dairy products. However, like natamycin, its classification as both a food additive and an antibiotic creates inherent tensions with antimicrobial stewardship principles.
2. Where It's Found
Nisin is used across a diverse range of processed food categories, reflecting its particular effectiveness against the Gram-positive bacterial spoilage organisms and pathogens most prevalent in dairy, heat-processed, and fermented foods:
- Processed cheese and cheese spreads: The primary commercial application. Nisin prevents the growth of Clostridium species responsible for "blowing" (gas production) and textural defects in processed cheese during extended ambient storage.
- Canned foods: Nisin is used in heat-processed canned vegetables, soups, and ready meals to provide additional protection against thermophilic spore-formers that survive retort processing, allowing reduction in thermal treatment intensity and improved product quality.
- Pasteurized liquid eggs: Prevents post-pasteurization contamination with Gram-positive pathogens during storage.
- Beer: Nisin controls bacterial spoilage — particularly Lactobacillus and Pediococcus species — in brewery production, extending shelf life without affecting yeast-mediated fermentation (since nisin is ineffective against yeasts).
- Crumpets and other baked goods: Nisin is incorporated into the dough to extend mold-free shelf life by suppressing bacterial rope disease (Bacillus subtilis) and certain mold-associated bacterial flora.
- Reconstituted and flavored milk products: Used in some markets to extend the shelf life of dairy-based beverages susceptible to Gram-positive spoilage organisms.
- Canned soups and sauces: Applied as a supplement to thermal processing in ambient-stable products.
3. How It Works
Nisin operates through a dual mechanism that makes it one of the most potent and mechanistically sophisticated naturally occurring antibiotics known:
Mechanism 1: Lipid II Sequestration
Nisin binds with extremely high affinity (Kd in the nanomolar range) to Lipid II, the essential bactoprenol-anchored cell wall precursor that shuttles cell wall building blocks (N-acetylmuramic acid pentapeptide units) across the bacterial cell membrane. By capturing and sequestering Lipid II molecules, nisin physically blocks the transglycosylation and transpeptidation steps of cell wall synthesis — the same target exploited by vancomycin, a clinically critical antibiotic. This mechanism alone would be bacteriostatic, preventing new cell wall synthesis.
Mechanism 2: Pore Formation
Beyond sequestering Lipid II, nisin uses the captured Lipid II molecules as docking platforms to insert into the bacterial membrane and form transmembrane pores approximately 1–2 nm in diameter. These pores are large enough to allow passage of ions, amino acids, and small metabolites, rapidly dissipating the membrane potential and cellular energy charge. The pore-forming activity converts the bacteriostatic Lipid II inhibition into a bactericidal mechanism.
The synergy of these two mechanisms — using the same target molecule for both cell wall inhibition and membrane disruption — makes nisin exceptionally potent at very low concentrations (minimum inhibitory concentrations typically in the range of 0.01–10 μg/mL against susceptible organisms).
Spectrum of Activity
Nisin's spectrum is restricted to Gram-positive bacteria. This specificity arises because:
- Gram-positive bacteria have cell walls with externally accessible Lipid II molecules, allowing nisin to reach its target.
- Gram-negative bacteria possess an outer membrane that excludes nisin (a polycationic peptide) from reaching the inner membrane and its Lipid II target.
Key susceptible organisms include: Listeria monocytogenes, Clostridium botulinum, Clostridium tyrobutyricum, Staphylococcus aureus (including some MRSA strains), Bacillus cereus, and numerous Lactobacillus and Streptococcus species — including many probiotic and beneficial gut bacteria.
4. Health Effects
Nisin's direct toxicological profile is among the most favorable of any food preservative:
- Rapid digestive inactivation: Nisin is a polypeptide of 34 amino acids (3,510 Da). In the gastrointestinal tract, it is rapidly hydrolyzed by pancreatic proteases — particularly chymotrypsin — into inactive amino acid fragments. This digestive inactivation means systemic absorption is negligible, and nisin is essentially treated by the body as a dietary protein source.
- Absence of carcinogenicity: No carcinogenic potential has been identified in animal studies. The Joint FAO/WHO Expert Committee on Food Additives (JECFA) evaluated nisin in 1969 and 1988 and found no evidence of chronic toxicity at relevant doses.
- Allergenicity: No credible reports of IgE-mediated allergic reactions to nisin in food have been documented in the peer-reviewed literature. The peptide's rapid digestion substantially reduces its allergenicity compared to intact dietary proteins.
- High acute oral tolerance: The LD50 in mice (oral) is approximately 6,950 mg/kg — extremely low toxicity. Rats fed 825 mg/kg/day for three generations showed no reproductive or developmental adverse effects.
Gut Microbiome Concerns
The less well-studied aspect of nisin consumption relates to its potential effects on the gut microbiome before digestive inactivation fully occurs:
- Nisin retains antibacterial activity in the upper GI tract, where pancreatic proteases have not yet fully degraded it. During this window, it encounters the Gram-positive bacteria colonizing the intestinal mucosa.
- Several studies using in vitro gut fermentation models have demonstrated that nisin at food-relevant concentrations significantly inhibits Lactobacillus and Bifidobacterium species — the cornerstone beneficial organisms of the human gut microbiome (Campion et al., 2013).
- Prolonged dietary exposure to nisin could theoretically shift the gut microbial community toward Gram-negative dominance, as Gram-negative organisms (Bacteroidetes, Proteobacteria) are intrinsically resistant. The consequences of such a shift for host metabolism, immune function, and disease risk are unknown but represent a meaningful research gap.
5. Antimicrobial Resistance
Despite nisin's favorable direct toxicology, its role as an antibiotic food additive raises parallel AMR concerns to those documented for natamycin:
Documented Nisin Resistance in Listeria
Nisin-resistant strains of Listeria monocytogenes — a priority foodborne pathogen — have been isolated from food processing environments including cheese production facilities (Gravesen et al., 2002). Resistance mechanisms include:
- Cell wall thickening: Increased D-alanylation of cell wall teichoic acids reduces the negative surface charge, decreasing nisin binding affinity.
- Membrane phospholipid modification: Incorporation of lysyl-phosphatidylglycerol into the membrane reduces nisin's ability to form stable pores.
- PrfA-regulated tolerance: Virulence regulator PrfA influences nisin tolerance through yet incompletely understood mechanisms.
Cross-Resistance with Clinical Antibiotics
The Lipid II-binding mechanism shared between nisin and vancomycin raises concern about cross-resistance. While the binding sites on Lipid II differ between nisin (pyrophosphate) and vancomycin (D-Ala-D-Ala), the D-alanylation resistance mechanisms that reduce nisin efficacy may also reduce susceptibility to other cell wall-active antibiotics. Cross-tolerance between nisin and clinical glycopeptide antibiotics has been demonstrated in laboratory conditions, though its clinical prevalence in food-associated strains requires further investigation.
Horizontal Gene Transfer of Resistance Determinants
Nisin resistance genes (particularly nsr encoding the nisin resistance protein, and operon components from nisRK regulatory pathways) reside on mobile genetic elements capable of horizontal transfer between bacterial species in food environments. Transfer of nisin immunity genes from nisin-producing L. lactis to other Gram-positive organisms — including potential pathogens — has been demonstrated in laboratory and food matrix models.
6. The Microbiome Paradox
Perhaps the most conceptually striking concern raised by dietary nisin is what might be called the microbiome paradox: nisin kills the very bacteria that the probiotic industry promotes as essential for health.
The probiotics market — now worth tens of billions of dollars annually — is anchored around oral supplementation with Lactobacillus and Bifidobacterium species. These genera are precisely the Gram-positive bacteria most susceptible to nisin at the concentrations achievable in the upper gastrointestinal tract before full proteolytic inactivation. Simultaneously, millions of people consuming probiotic supplements are regularly eating processed cheeses, canned soups, and other foods preserved with nisin.
The practical implications of this parallel are underexplored in published research:
- Probiotic viability reduction: Nisin present in the stomach and small intestine at the time of probiotic capsule transit could reduce the viable count of probiotic organisms reaching the large intestine, partially or fully negating the intended probiotic dose.
- Endogenous microbiome suppression: Beyond supplemented probiotics, nisin could suppress endogenous Lactobacillus populations in the small intestinal microbiome, where these organisms play roles in colonization resistance and mucosal immune modulation.
- Ecological shift toward Gram-negative dominance: Regular dietary nisin could exert selective pressure favoring Gram-negative bacteria in the gut, potentially contributing to dysbiosis characterized by increased lipopolysaccharide (LPS) burden from Gram-negative organisms — a state associated with systemic low-grade inflammation and metabolic disease risk.
Controlled human trials examining the gut microbiome composition of individuals with high versus low dietary nisin exposure — ideally using shotgun metagenomic sequencing to characterize community composition — are urgently needed to determine whether these theoretical concerns translate to measurable effects.
7. Regulatory Status
Nisin holds regulatory approval across more than 50 countries, making it one of the most broadly accepted natural antimicrobial food preservatives globally:
- United States (FDA): Approved as GRAS for use in pasteurized cheese spreads and pasteurized process cheese foods, at concentrations of up to 10,000 IU per gram of cheese. Listed in 21 CFR 184.1538. GRAS status has been extended by FDA notification for additional food categories.
- European Union: Approved as E234 under Regulation (EC) No 1333/2008 for use in clotted cream (maximum 10 mg/kg), mascarpone (maximum 10 mg/kg), and semi-hard and processed cheese (maximum 12.5 mg/kg). The EU imposes more restrictive permitted uses than the FDA.
- WHO/FAO (JECFA): An Acceptable Daily Intake (ADI) of 0–33,000 IU/kg body weight was established, reflecting extremely wide safety margins relative to typical dietary exposure. This ADI translates to approximately 1.25 mg/kg body weight/day of pure nisin — for a 70 kg adult, approximately 87.5 mg/day, vastly exceeding any realistic dietary intake.
- Codex Alimentarius: Nisin is listed in the Codex General Standard for Food Additives for use in specific food categories including heat-treated cheese and cream.
- Organic foods: Like natamycin, nisin is not permitted in certified organic foods under USDA NOP or EU organic regulations, due to its antibiotic classification.
8. Emerging Research
Beyond its established role as a food preservative, nisin has attracted substantial research interest in several emerging applications:
Anti-Cancer Activity
A series of studies beginning in the 2010s has investigated nisin's cytotoxic activity against cancer cells. Kamarajan et al. (2015) published significant findings demonstrating that nisin ZP (a variant form) exhibited potent cytotoxicity against head and neck squamous cell carcinoma (HNSCC) cells in vitro and in murine xenograft models. The proposed mechanism involves nisin-mediated disruption of cancer cell membranes through phosphatidylethanolamine enrichment at cancer cell surfaces — a lipid composition difference that may increase susceptibility to pore-forming peptides relative to normal cells. While these findings are early-stage, they have generated interest in nisin as an anti-cancer drug candidate. (DOI: 10.1371/journal.pone.0129519)
Nisin-Producing Probiotics
Research groups have explored the concept of engineering or selecting L. lactis nisin-producing strains as "therapeutic probiotics" — organisms that could colonize the gut and produce nisin in situ to suppress pathogenic Gram-positive bacteria including Clostridioides difficile and Listeria. This approach leverages nisin's natural origin but raises the same concern about suppression of beneficial Lactobacillus strains.
Bioengineered Nisin Variants
Through site-directed mutagenesis and combinatorial biosynthesis, researchers have generated hundreds of nisin analogs with modified activity spectra, enhanced stability, or improved activity against specific resistant pathogens. Nisin variants with activity against Gram-negative bacteria (achieved by combining nisin with membrane-permeabilizing agents such as EDTA) represent one avenue being explored for food safety applications against Salmonella and E. coli. Additionally, variants with increased activity against nisin-resistant Listeria isolates from food environments have been developed, addressing the practical resistance problem in food processing.
Synergistic Combinations
Nisin combined with other hurdle technologies — high pressure processing, essential oils (thymol, carvacrol), organic acids, or bacteriophages — shows synergistic activity that allows reduction of nisin concentrations needed for equivalent preservation, potentially reducing selection pressure for resistance.
9. How to Avoid
- Read cheese labels for “nisin,” “E234,” or “Nisaplin.”
- Choose USDA Organic or EU Organic cheeses – both standards prohibit nisin as an antibiotic-category additive.
- Avoid shelf-stable processed cheese spreads and canned cheese products, which are the largest volume use of nisin.
- Artisanal and farmstead cheese producers typically rely on controlled aging rather than added bacteriocins.
- Take probiotics separately from meals containing nisin-preserved foods to reduce overlap of transit in the upper GI tract.
Connections
- Preservatives
- Natamycin
- Sodium Nitrite
- Potassium Sorbate
- BHA and BHT
- US vs EU Food Safety
- Banned Additives
- Food Additives
- Cancer
- Gut-Brain Axis
- Probiotics
- Gut Microbiome
- Fermented Foods
- Food Dyes Banned Worldwide
- Milk
- Staphylococcus Aureus
- Bacillus Subtilis
Research Papers
- Biswas SR, Ray P, Johnson MC, Ray B. Influence of growth conditions on the production of a bacteriocin, pediocin AcH, by Pediococcus acidilactici H. Applied and Environmental Microbiology. 1991;57(4):1265–1267.
- Breukink E, de Kruijff B. Lipid II as a target for antibiotics. Nature Reviews Drug Discovery. 2006;5(4):321–332.
- Campion A, Casey PG, Field D, Cotter PD, Hill C, Ross RP. In vivo activity of nisin against Listeria monocytogenes and the effects on the intestinal microbiota of mice. Applied and Environmental Microbiology. 2013;79(1):204–210.
- Gravesen A, Ramnath M, Rechinger KB, Andersen N, Jänsch L, Héchard Y, Hastings JW, Knøchel S. High-level resistance to class IIa bacteriocins is associated with one general mechanism in Listeria monocytogenes. Microbiology. 2002;148(8):2361–2369.
- Kamarajan P, Hayami T, Matte B, Liu Y, Danciu T, Ramamurthy NS, Bhatt DL, Bhatt DL, Kapila YL. Nisin ZP, a bacteriocin and food preservative, inhibits head and neck cancer tumorigenesis and prolongs survival. PLOS ONE. 2015;10(7):e0129519.
- Delves-Broughton J. Nisin and its uses as a food preservative. Food Technology. 1990;44(11):100–112.
- EFSA Panel on Food Additives and Nutrient Sources. Scientific Opinion on the re-evaluation of nisin (E 234) as a food additive. EFSA Journal. 2006;4(1):81.
- JECFA. Nisin preparation. In: WHO Food Additives Series 12. Joint FAO/WHO Expert Committee on Food Additives; 1978. WHO, Geneva.
- Cotter PD, Ross RP, Hill C. Bacteriocins — a viable alternative to antibiotics? Nature Reviews Microbiology. 2013;11(2):95–105.
- Rogers LA, Whittier EO. Limiting factors in the lactic fermentation. Journal of Bacteriology. 1928;16(4):211–229.
- Wiedemann I, Breukink E, van Kraaij C, Kuipers OP, Bierbaum G, de Kruijff B, Sahl HG. Specific binding of nisin to the peptidoglycan precursor lipid II combines pore formation and inhibition of cell wall biosynthesis for potent antibiotic activity. Journal of Biological Chemistry. 2001;276(3):1772–1779.
- Field D, Cotter PD, Hill C, Ross RP. Bioengineering of the model lantibiotic nisin. Bioengineered. 2015;6(4):187–192.
- Saarela M, Alakomi HL, Mättö J, Ahonen AM, Puhakka A, Tynkkynen S. Effect of nisin on the survival of Lactobacillus rhamnosus in the gastrointestinal tract and feces. Journal of Applied Microbiology. 2011;110(3):598–606.
- European Commission. Regulation (EC) No 1333/2008 of the European Parliament and of the Council on food additives. Official Journal of the European Union. 2008;L354:16–33.
- FDA. Code of Federal Regulations, Title 21, Section 184.1538: Nisin preparation. U.S. Food and Drug Administration. 2023.