Biotin and Hair, Skin, and Nail Health
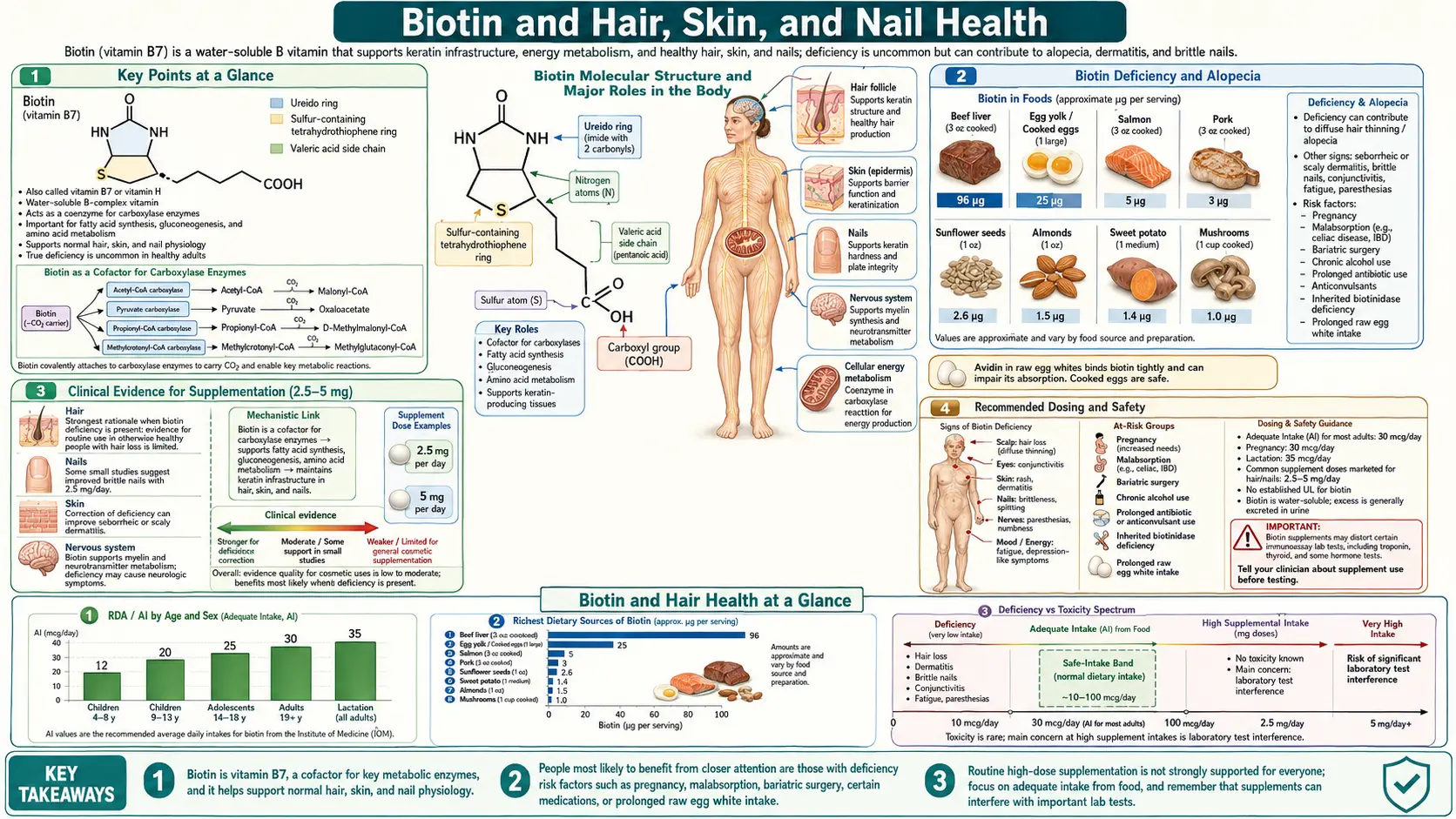
Biotin (vitamin B7) is one of the most widely marketed supplements for cosmetic health, appearing in countless hair growth formulas, nail strengtheners, and skin care products. As a coenzyme for carboxylase enzymes, biotin plays a genuine role in the metabolic pathways that support keratin infrastructure, but the gap between its biochemical function and the claims made by the supplement industry deserves careful examination.
Table of Contents
- Key Points at a Glance
- Keratin Infrastructure and Biotin's Role
- Hair Follicle Cell Proliferation
- Biotin Deficiency and Alopecia
- Nail Brittleness and Onychorrhexis
- Dermatitis Connection
- Clinical Evidence for Supplementation (2.5-5 mg)
- Biotin and Thyroid/Lab Test Interference
- Popular Use vs. Scientific Evidence
- Recommended Dosing and Safety
- Research Papers
- Connections
- Featured Videos
Key Points at a Glance
- Cofactor for five carboxylases essential for fatty acid, amino acid, and glucose metabolism.
- Brittle nail syndrome — 2.5 mg/day thickens nail plates in the best-supported cosmetic indication.
- Deficiency alopecia resolves fully with repletion but primary deficiency is rare in well-fed adults.
- No proven benefit for hair growth in biotin-replete individuals despite popular marketing.
- Lab assay interference — high-dose biotin causes spuriously high or low thyroid, troponin, and hormone results.
- Raw egg whites contain avidin, which binds biotin; cooking denatures avidin.
- Adequate Intake (AI) 30 mcg/day; water-soluble with no established toxicity threshold.
- Stop biotin ≥72 hours before blood work that involves biotin-streptavidin immunoassays.
Keratin Infrastructure and Biotin's Role
Keratin is the primary structural protein of hair, skin, and nails. Its production depends on a complex series of metabolic processes in which biotin serves as an essential cofactor.
- Keratin synthesis: Biotin supports the activity of carboxylase enzymes involved in amino acid metabolism, particularly the catabolism of leucine, isoleucine, and valine. These branched-chain amino acids contribute building blocks for keratin protein assembly in hair follicle keratinocytes and nail matrix cells.
- Acetyl-CoA carboxylase: This biotin-dependent enzyme catalyzes the first committed step of fatty acid synthesis. Adequate fatty acid production is essential for the lipid-rich intercellular cement that holds keratinocytes together in the nail plate and skin barrier.
- Propionyl-CoA carboxylase: This enzyme metabolizes odd-chain fatty acids and certain amino acids. Disruptions in its activity can impair the structural integrity of keratinized tissues.
- Gene expression regulation: Emerging research suggests biotin may influence the expression of genes encoding keratin and keratin-associated proteins through biotinylation of histones, a form of epigenetic modification.
Hair Follicle Cell Proliferation
The hair follicle is one of the most metabolically active structures in the human body, with matrix cells in the hair bulb dividing approximately every 23 to 72 hours during the anagen (growth) phase of the hair cycle.
- Energy metabolism: Rapidly dividing follicle cells require constant ATP production. Biotin-dependent carboxylases in the citric acid cycle and gluconeogenesis pathways ensure that hair matrix cells have adequate energy substrates to sustain high mitotic rates.
- Cell signaling: Biotin participates in cell signaling pathways that regulate keratinocyte differentiation. The Wnt/beta-catenin pathway, critical for hair follicle morphogenesis and cycling, may be indirectly influenced by biotin status through its effects on cellular metabolism.
- Dermal papilla function: The dermal papilla at the base of the hair follicle acts as a signaling center that instructs matrix cells. Nutritional deficiencies, including biotin deficiency, can impair dermal papilla signaling and shorten the anagen phase, leading to increased hair shedding.
Biotin Deficiency and Alopecia
True biotin deficiency produces a well-documented pattern of hair loss, though it is uncommon in the general population.
- Diffuse alopecia: Biotin deficiency causes a characteristic diffuse, non-scarring alopecia that typically affects the scalp, eyebrows, and eyelashes. Hair becomes progressively finer and more brittle before thinning becomes visually apparent.
- Biotinidase deficiency: This inherited enzyme deficiency prevents the recycling of biotin from biocytin, leading to profound biotin deficiency in infancy. Affected children develop alopecia totalis if left untreated, which is fully reversible with biotin supplementation.
- Acquired deficiency causes: Prolonged raw egg white consumption (avidin binds biotin), long-term anticonvulsant therapy (carbamazepine, phenytoin, phenobarbital), chronic alcoholism, and prolonged parenteral nutrition without biotin supplementation can all lead to acquired deficiency with hair loss as a prominent symptom.
- Pregnancy-related depletion: Marginal biotin deficiency occurs in up to one-third of pregnancies due to increased biotin catabolism and reduced renal reabsorption. While frank alopecia is uncommon, some postpartum hair shedding may be partly attributable to suboptimal biotin status during pregnancy.
Nail Brittleness and Onychorrhexis
Brittle nail syndrome, characterized by onychorrhexis (longitudinal ridging and splitting) and onychoschizia (lamellar splitting of the nail plate), is one area where biotin supplementation has more supportive clinical evidence.
- Nail plate composition: The nail plate is composed of approximately 80% hard keratins cross-linked by disulfide bonds, embedded in a matrix of soft keratins and lipids. Biotin's role in both amino acid metabolism and fatty acid synthesis is relevant to maintaining this complex structure.
- Clinical studies on nail brittleness: A landmark 1993 study by Colombo and colleagues demonstrated a 25% increase in nail plate thickness in patients with brittle nails who took 2.5 mg of biotin daily for 6 to 15 months. Subsequent studies have corroborated improvements in nail firmness and reduced splitting.
- Veterinary precedent: Biotin supplementation has a long and well-established history in veterinary medicine for improving hoof quality in horses and claw integrity in swine, which provided the initial rationale for human nail studies.
Dermatitis Connection
Skin manifestations of biotin deficiency are among the earliest and most recognizable signs of inadequate biotin status.
- Periorificial dermatitis: Biotin deficiency produces a characteristic erythematous, scaly rash distributed around the eyes, nose, and mouth. This distribution pattern overlaps with seborrheic dermatitis and zinc deficiency, requiring careful differential diagnosis.
- Skin barrier function: Biotin supports the synthesis of ceramides and other lipid components of the stratum corneum, the outermost layer of the epidermis that serves as the primary barrier against transepidermal water loss and external irritants. Deficiency compromises this barrier, leading to dry, flaky, and irritation-prone skin.
- Seborrheic dermatitis in infants: Some cases of infantile seborrheic dermatitis (cradle cap) have been associated with marginal biotin status, though this relationship remains debated. The condition typically resolves spontaneously regardless of biotin supplementation.
Clinical Evidence for Supplementation (2.5-5 mg)
The evidence base for biotin supplementation in individuals without documented deficiency is considerably weaker than marketing claims suggest.
- Hair growth studies: A 2017 systematic review found that all published cases of biotin supplementation improving hair growth involved individuals with underlying pathology or documented biotin deficiency. No large-scale, randomized controlled trials have demonstrated that biotin supplementation promotes hair growth in healthy individuals with adequate biotin intake.
- Common dosing regimens: Supplement manufacturers typically recommend 2.5 mg (2,500 mcg) to 5 mg (5,000 mcg) daily for hair and nail benefits. These doses are 80 to 170 times the Adequate Intake (AI) of 30 mcg per day for adults. While biotin has no established tolerable upper intake level due to its low toxicity, the clinical justification for such high doses in non-deficient individuals is limited.
- Duration of treatment: In studies showing positive results for nail brittleness, improvements were typically observed after 3 to 6 months of consistent supplementation, reflecting the slow growth rate of nails (approximately 3 mm per month for fingernails).
- Combination supplements: Many hair growth supplements combine biotin with other nutrients such as zinc, iron, vitamin D, and marine collagen, making it difficult to attribute any observed benefits specifically to biotin.
Biotin and Thyroid/Lab Test Interference
One of the most clinically significant concerns regarding high-dose biotin supplementation is its well-documented interference with immunoassay laboratory tests.
- Mechanism of interference: Many modern immunoassays use streptavidin-biotin chemistry in their detection systems. Excess circulating biotin competes with the biotinylated reagents, causing spurious results. Depending on the assay format (competitive vs. sandwich), biotin can cause either falsely elevated or falsely decreased values.
- Thyroid function tests: High-dose biotin supplementation can produce laboratory results mimicking Graves' disease, with falsely elevated free T4 and free T3 and falsely suppressed TSH on streptavidin-biotin-based assays. This pattern has led to unnecessary treatment for hyperthyroidism in several documented case reports.
- Troponin assays: Biotin interference with high-sensitivity troponin assays can cause falsely low results, potentially masking an acute myocardial infarction. The FDA issued a safety communication in 2017 warning about this risk.
- Other affected tests: Parathyroid hormone, vitamin D (25-hydroxyvitamin D), cortisol, ferritin, testosterone, estradiol, progesterone, DHEA-sulfate, PSA, and many tumor markers can all be affected by biotin interference, depending on the specific assay platform used by the laboratory.
- Clinical recommendation: Patients should discontinue high-dose biotin supplements at least 72 hours before laboratory testing. Healthcare providers should inquire about biotin supplementation when laboratory results are discordant with clinical presentation.
Popular Use vs. Scientific Evidence
The disconnect between consumer enthusiasm and scientific evidence for biotin supplementation in cosmetic applications is striking.
- Market size: Biotin supplements represent a multi-billion-dollar global market, driven largely by social media marketing, influencer endorsements, and consumer testimonials rather than by clinical evidence from well-designed trials.
- Placebo effect and confounders: Individuals who begin taking biotin supplements often simultaneously adopt other hair-healthy behaviors such as improved diet, reduced heat styling, and gentler hair care practices, making it difficult to attribute improvements solely to biotin.
- Deficiency screening: Rather than empiric supplementation, a more evidence-based approach involves screening for biotin deficiency (via urinary 3-hydroxyisovaleric acid excretion or plasma biotinidase activity) in patients presenting with hair loss, and supplementing only those with demonstrated inadequacy.
- Other causes of hair loss: Androgenetic alopecia, telogen effluvium, iron deficiency, thyroid dysfunction, and autoimmune conditions are far more common causes of hair loss than biotin deficiency. Biotin supplementation is unlikely to address these underlying conditions.
Recommended Dosing and Safety
- Adequate Intake (AI): The established AI for biotin is 30 mcg per day for adults, an amount readily obtained from a varied diet including eggs, nuts, legumes, whole grains, and organ meats.
- Therapeutic dosing: For documented biotin deficiency, doses of 5 to 10 mg daily are typically used. For brittle nail syndrome, 2.5 mg daily has the most supporting evidence. For hair loss without documented deficiency, there is insufficient evidence to recommend a specific dose.
- Safety profile: Biotin is water-soluble and has no established upper limit of toxicity. Even at high doses (up to 200 mg daily in some multiple sclerosis trials), direct toxicity has not been observed. However, the laboratory test interference risk is a genuine safety concern at doses above 1 mg daily.
- Dietary sources: Egg yolks (cooked), liver, salmon, avocado, sweet potatoes, almonds, and sunflower seeds are excellent dietary sources of biotin. Intestinal bacteria also synthesize biotin, though the contribution of bacterial biotin to human nutritional status remains unclear.
Summary
Biotin plays a legitimate biochemical role in supporting keratin-producing tissues, and supplementation is clearly effective in reversing hair, skin, and nail abnormalities caused by biotin deficiency. However, evidence that supplementation benefits individuals with adequate biotin status is limited, particularly for hair growth. The strongest evidence for cosmetic supplementation exists for brittle nail syndrome, where 2.5 mg daily over several months has demonstrated measurable improvements. Patients and clinicians should be aware of the significant risk of laboratory test interference with high-dose biotin supplements.
Research Papers
- PubMed — Biotin for brittle nail syndrome (topic search)
- PubMed — Biotin deficiency and alopecia (topic search)
- PubMed — Biotin interference in immunoassays (topic search)
- PubMed — Biotinidase deficiency (topic search)
- PubMed — Keratin structure and cysteine disulfide bonds (topic search)
- PubMed — Biotin and hair growth systematic reviews (topic search)
- FDA Safety Communication — Biotin may interfere with lab tests
- PubMed — Avidin, raw egg whites, and biotin deficiency (topic search)
- NIH Office of Dietary Supplements — Biotin Fact Sheet for Health Professionals
- Linus Pauling Institute — Biotin
- Harvard T.H. Chan School of Public Health — Biotin (Vitamin B7)
Connections
- Vitamin B7
- Zinc
- Iron
- Biotin and Blood Sugar
- Vitamin B5
- Eggs
- Vitamin D3
- Alopecia
- Hair Loss
- Collagen
- Cysteine
- Testosterone
- Organ Meats