Biotin and Blood Sugar Regulation
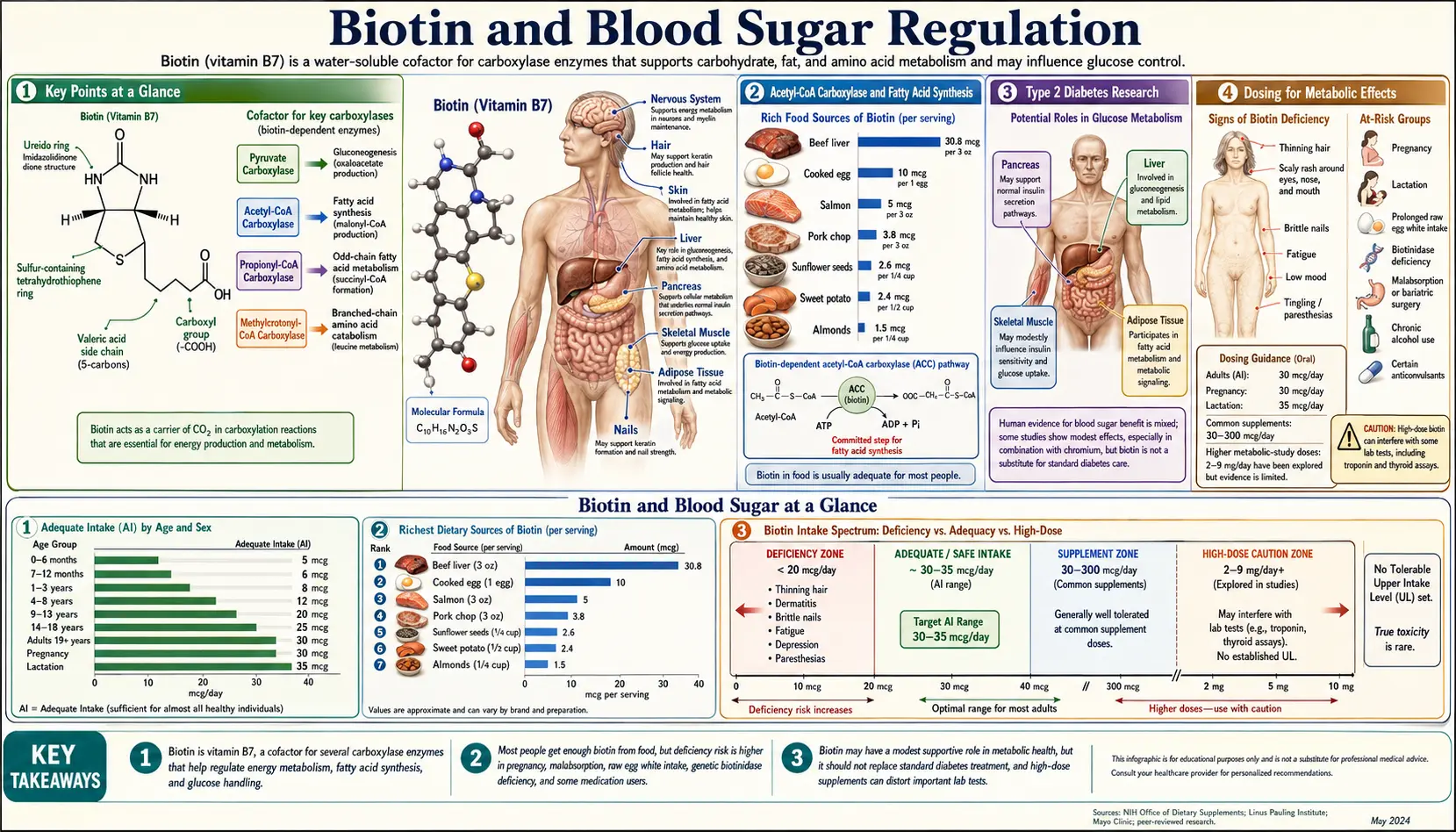
Beyond its well-known role in hair and nail health, biotin (vitamin B7) is an essential coenzyme in several metabolic pathways that directly govern glucose and lipid metabolism. Accumulating research over the past two decades has explored the therapeutic potential of biotin supplementation in improving glycemic control, particularly in individuals with type 2 diabetes mellitus. These effects involve biotin's influence on key regulatory enzymes in gluconeogenesis, fatty acid synthesis, and insulin secretion.
Table of Contents
- Key Points at a Glance
- Glucokinase Gene Expression
- Pyruvate Carboxylase and Gluconeogenesis
- Acetyl-CoA Carboxylase and Fatty Acid Synthesis
- Insulin Secretion Enhancement
- Chromium-Biotin Synergy Studies
- Type 2 Diabetes Research
- Glycemic Control Mechanisms
- Clinical Trial Evidence
- Dosing for Metabolic Effects
- Research Papers
- Connections
- Featured Videos
Key Points at a Glance
- Upregulates hepatic glucokinase, enhancing glucose uptake and disposal.
- Modulates pyruvate carboxylase, a key gluconeogenic enzyme.
- Supports acetyl-CoA carboxylase, connecting lipogenesis to glucose metabolism.
- Chromium-biotin combination lowers A1c in inadequately controlled T2DM (Albarracin 2008).
- Pharmacological doses 2–15 mg/day are required for metabolic effects.
- Lab test interference at these doses — critical safety issue for diabetes monitoring.
- Water-soluble with good safety profile at high doses up to 24 months.
- Adjunctive only — not a replacement for standard diabetes therapy.
Glucokinase Gene Expression
One of the most significant mechanisms by which biotin influences blood sugar regulation is through its effect on glucokinase, the rate-limiting enzyme for glucose metabolism in both pancreatic beta cells and hepatocytes.
- Transcriptional regulation: Biotin stimulates the transcription of the glucokinase gene by enhancing the activity of its promoter region. In pancreatic beta cells, glucokinase serves as the glucose sensor that couples circulating glucose levels to insulin secretion. Increased glucokinase expression lowers the glucose threshold for insulin release, enabling more responsive glucose-stimulated insulin secretion.
- Hepatic glucose utilization: In the liver, glucokinase phosphorylates glucose to glucose-6-phosphate, trapping it within the hepatocyte for glycogen synthesis or glycolysis. Biotin-mediated upregulation of hepatic glucokinase promotes postprandial glucose clearance from the bloodstream and enhances glycogen storage capacity.
- Dose-response relationship: Animal studies have demonstrated that pharmacological doses of biotin (40 to 100 times the recommended intake) produce significant increases in glucokinase mRNA expression, with corresponding improvements in glucose tolerance. These effects appear to require supraphysiological biotin concentrations.
- Epigenetic mechanisms: Biotin may influence glucokinase expression through histone biotinylation, a post-translational modification of histone proteins that alters chromatin structure and gene accessibility. This epigenetic mechanism represents a distinct pathway through which biotin status can affect metabolic gene expression.
Pyruvate Carboxylase and Gluconeogenesis
Pyruvate carboxylase (PC) is a biotin-dependent enzyme that catalyzes the first committed step of gluconeogenesis, the metabolic pathway by which the liver produces glucose from non-carbohydrate precursors.
- Enzymatic function: PC converts pyruvate to oxaloacetate in the mitochondrial matrix, using biotin as a covalently bound prosthetic group that serves as a mobile carboxyl carrier. This reaction requires ATP and bicarbonate as co-substrates and is allosterically activated by acetyl-CoA.
- Gluconeogenesis regulation: By controlling the flux through this initial step, biotin availability can influence the rate of hepatic glucose production. In type 2 diabetes, excessive gluconeogenesis is a major contributor to fasting hyperglycemia. The relationship between biotin status and gluconeogenic rate is complex, as PC activity is essential for normal metabolism but dysregulated gluconeogenesis drives diabetic hyperglycemia.
- Anaplerotic function: PC also serves an anaplerotic role by replenishing oxaloacetate in the citric acid cycle. This dual function means that biotin's influence on PC activity affects both glucose production and overall energy metabolism in multiple tissues.
- Interaction with insulin signaling: Insulin normally suppresses hepatic gluconeogenesis by downregulating PC expression and activity. Biotin supplementation may enhance insulin-mediated suppression of gluconeogenesis, contributing to improved fasting glucose levels in insulin-resistant states.
Acetyl-CoA Carboxylase and Fatty Acid Synthesis
Acetyl-CoA carboxylase (ACC) is another biotin-dependent enzyme with profound implications for metabolic regulation and glucose homeostasis.
- Rate-limiting step: ACC catalyzes the carboxylation of acetyl-CoA to malonyl-CoA, the first and rate-limiting step in de novo fatty acid synthesis. This reaction directly links carbohydrate metabolism to lipid storage, as excess glucose is converted to fatty acids through this pathway.
- Malonyl-CoA as a metabolic signal: Beyond its role as a substrate for fatty acid synthase, malonyl-CoA inhibits carnitine palmitoyltransferase I (CPT-I), the enzyme that transports long-chain fatty acids into the mitochondria for beta-oxidation. Elevated malonyl-CoA levels therefore shift the metabolic balance from fat burning toward fat storage.
- ACC isoforms: Two isoforms exist: ACC1 (cytoplasmic, primarily in lipogenic tissues) and ACC2 (associated with the mitochondrial outer membrane, primarily in oxidative tissues). Biotin is required for both isoforms, and its effects on lipid metabolism vary by tissue and metabolic context.
- Implications for insulin resistance: Dysregulation of ACC activity contributes to ectopic lipid accumulation in liver and skeletal muscle, a key driver of insulin resistance. Biotin's influence on ACC activity may modulate intracellular lipid partitioning in ways relevant to insulin sensitivity, though the direction and magnitude of these effects depend on overall metabolic status.
Insulin Secretion Enhancement
Biotin's effects on pancreatic beta cell function extend beyond glucokinase gene expression to encompass multiple aspects of insulin synthesis and secretion.
- Insulin gene transcription: Biotin upregulates the transcription of the insulin gene (INS) in pancreatic beta cells. Studies in isolated islets and beta cell lines have demonstrated increased insulin mRNA levels and protein content following biotin treatment at pharmacological concentrations.
- Calcium signaling: Glucose-stimulated insulin secretion depends on calcium influx through voltage-gated calcium channels. Biotin has been shown to enhance calcium channel activity in beta cells, potentially amplifying the insulin secretory response to glucose stimulation.
- Islet cGMP-dependent protein kinase: Biotin activates soluble guanylate cyclase in pancreatic islets, increasing intracellular cGMP levels. The cGMP/PKG signaling pathway potentiates insulin exocytosis and contributes to the insulin secretagogue effect observed with high-dose biotin.
- Beta cell preservation: Some animal studies suggest biotin may have protective effects against beta cell apoptosis induced by inflammatory cytokines and oxidative stress, though these findings require confirmation in human studies.
Chromium-Biotin Synergy Studies
The combination of chromium picolinate and biotin has been investigated as a synergistic approach to improving glycemic control in type 2 diabetes.
- Complementary mechanisms: Chromium enhances insulin receptor signaling by potentiating insulin receptor tyrosine kinase activity and increasing GLUT4 transporter translocation to the cell surface. Biotin's effects on glucokinase expression and insulin secretion operate through distinct pathways, providing the rationale for combination therapy.
- Clinical trial evidence: A randomized, double-blind, placebo-controlled trial (Singer and Geohas, 2006) investigated the combination of chromium picolinate (600 mcg) and biotin (2 mg) in 447 subjects with poorly controlled type 2 diabetes. The combination produced statistically significant reductions in HbA1c (-0.54%) and fasting glucose compared to placebo over 90 days.
- Lipid profile effects: The same trial reported improvements in the atherogenic lipid profile, with reductions in LDL cholesterol and the LDL/HDL ratio in the treatment group. These lipid effects suggest broader metabolic benefits beyond glycemic control.
- Mechanism of synergy: The proposed mechanism involves chromium improving peripheral insulin sensitivity (enhancing glucose uptake in muscle and adipose tissue) while biotin enhances hepatic glucose utilization and insulin secretion. Together, they address both the insulin resistance and beta cell dysfunction components of type 2 diabetes.
Type 2 Diabetes Research
The body of evidence examining biotin's role in type 2 diabetes management continues to evolve, with both promising findings and significant limitations.
- Animal model data: Studies in diabetic rodent models (db/db mice, Zucker Diabetic Fatty rats, streptozotocin-induced diabetic rats) have consistently demonstrated that pharmacological doses of biotin improve glucose tolerance, reduce fasting glucose, and enhance insulin sensitivity. Effective doses in these models typically range from 2 to 100 mg/kg body weight.
- Human observational data: Several cross-sectional studies have reported lower serum biotin levels in patients with type 2 diabetes compared to non-diabetic controls. However, whether reduced biotin levels are a cause or consequence of the metabolic derangements of diabetes remains unclear.
- Intervention trials: Beyond the chromium-biotin combination trials, smaller studies of biotin monotherapy (typically 5 to 15 mg daily) have reported improvements in fasting glucose and postprandial glucose excursions in type 2 diabetes patients. However, these studies are limited by small sample sizes, short durations, and methodological heterogeneity.
- Triglyceride reduction: Biotin supplementation has been associated with significant reductions in serum triglyceride levels in several human studies, an effect attributable to its role in ACC regulation and fatty acid metabolism. This lipid-lowering effect may have independent cardiovascular benefits in the diabetic population.
Glycemic Control Mechanisms
The multiple pathways through which biotin influences glucose homeostasis can be integrated into a comprehensive model of its metabolic effects.
- Hepatic glucose output: Biotin modulates both gluconeogenesis (via pyruvate carboxylase) and glycolysis/glycogen synthesis (via glucokinase), positioning it as a regulator of the hepatic glucose balance. In the fed state, biotin-mediated glucokinase upregulation promotes glucose disposal; in the fasting state, its role in PC activity supports appropriate glucose production.
- Pancreatic insulin response: Enhanced glucokinase expression in beta cells, coupled with effects on calcium signaling and cGMP pathways, improves the first-phase insulin secretory response that is characteristically blunted in type 2 diabetes.
- Peripheral glucose uptake: While biotin's direct effects on skeletal muscle glucose uptake are less well characterized than its hepatic and pancreatic actions, some evidence suggests biotin may enhance insulin-stimulated glucose transport in muscle through effects on AMPK signaling.
- Inflammatory modulation: Chronic low-grade inflammation contributes to insulin resistance. Biotin has demonstrated anti-inflammatory properties, including suppression of NF-kB signaling and reduction of pro-inflammatory cytokine production, which may indirectly improve insulin sensitivity.
Clinical Trial Evidence
- Positive findings: Several clinical trials support biotin's glycemic benefits. Baez-Saldana et al. (2004) reported that 9 mg of biotin daily for one month reduced fasting plasma glucose by approximately 45% in type 2 diabetes patients. Revilla-Monsalve et al. (2006) demonstrated improvements in both fasting and postprandial glucose with 5 mg daily biotin supplementation.
- Limitations of existing evidence: The current evidence base suffers from several weaknesses: most studies are small (fewer than 100 participants), of short duration (less than 6 months), conducted in single centers with limited ethnic diversity, and often lack adequate blinding and placebo controls. Publication bias toward positive results is also a concern.
- Need for large-scale trials: Definitive determination of biotin's role in diabetes management will require large-scale, multi-center, randomized, double-blind, placebo-controlled trials with clinically meaningful endpoints (HbA1c reduction, cardiovascular outcomes) over extended durations. No such trial has been completed to date.
- Comparison with established therapies: The magnitude of glycemic improvement reported with biotin supplementation is generally modest compared to established antidiabetic medications such as metformin, sulfonylureas, or SGLT2 inhibitors. Biotin should not be considered a replacement for standard pharmacotherapy in diabetes management.
Dosing for Metabolic Effects
- Pharmacological vs. nutritional doses: The metabolic effects of biotin on blood sugar regulation require pharmacological doses (2 to 15 mg daily) that are 70 to 500 times the Adequate Intake of 30 mcg. These effects are not achievable through dietary biotin intake alone and represent a true pharmacological action distinct from correction of nutritional deficiency.
- Commonly studied doses: Most clinical trials have used doses in the range of 5 to 15 mg daily, with 9 mg being a frequently studied dose in monotherapy trials and 2 mg being typical in combination with chromium picolinate.
- Safety at pharmacological doses: Biotin is water-soluble and has demonstrated a favorable safety profile at pharmacological doses in clinical trials lasting up to 24 months. No significant adverse effects have been consistently attributed to high-dose biotin. However, the laboratory test interference issue is a critical safety concern at these doses and must be communicated to patients and their healthcare providers.
- Timing of administration: Limited pharmacokinetic data suggest that dividing the daily dose across meals may optimize absorption and maintain more consistent plasma biotin concentrations. However, no definitive dosing regimen has been established for metabolic indications.
- Clinical integration: Biotin supplementation for glycemic benefit should be viewed as a potential adjunctive therapy under medical supervision, not as a standalone treatment for diabetes. Patients should inform all healthcare providers of biotin supplementation to ensure appropriate interpretation of laboratory tests.
Summary
Biotin influences blood sugar regulation through multiple biochemically plausible mechanisms, including glucokinase gene upregulation, modulation of gluconeogenesis via pyruvate carboxylase, regulation of fatty acid synthesis through acetyl-CoA carboxylase, and enhancement of insulin secretion. Clinical trial data, while promising, remain insufficient to establish definitive clinical recommendations. The combination of chromium and biotin has the strongest clinical evidence to date. Pharmacological doses of 2 to 15 mg daily are required for metabolic effects, and patients must be counseled about laboratory test interference at these doses.
Research Papers
- Albarracin CA, et al. Chromium picolinate and biotin combination improves glucose metabolism in treated, uncontrolled type 2 diabetes. (PubMed topic search)
- PubMed — Biotin and glucokinase gene expression (topic search)
- PubMed — Biotin and pyruvate carboxylase (topic search)
- PubMed — Biotin and insulin secretion (topic search)
- PubMed — Biotin and plasma lipids (topic search)
- PubMed — Biotin and HbA1c in type 2 diabetes (topic search)
- PubMed — Chromium picolinate and glucose control (topic search)
- PubMed — Biotin assay interference in endocrine testing (topic search)
- NIH Office of Dietary Supplements — Biotin Fact Sheet for Health Professionals
- Linus Pauling Institute — Biotin
- Harvard T.H. Chan School of Public Health — Biotin (Vitamin B7)
Connections
- Vitamin B7
- Diabetes
- Chromium
- Biotin and Hair Health
- Magnesium
- Zinc
- Hemoglobin A1C
- Vitamin D3
- Blood Sugar
- Insulin Resistance
- Fasting
- Metabolic Syndrome
- Calcium